Abstract
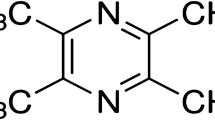
Emerging epidemiologic data indicates that diabetes is a potential predisposing factor for neuropsychiatric deficits as stroke, cerebrovascular diseases, diabetes-associated cognitive decline, depression and anxiety. Diabetes-associated cognitive decline, characterized by impaired cognitive functions and neurochemical and structural abnormalities, involves direct neuronal damage caused by intracellular glucose. The present study was designed to investigate the effect of sesamol (3,4-methylenedioxyphenol), a phenolic antioxidant and anti-inflammatory molecule, on cognitive functions, oxidative stress and inflammation in diabetic rats. Learning and memory behaviors were investigated using a spatial version of the Morris water maze test. Acetylcholinesterase activity, a marker of cholinergic dysfunction, was increased by 80% in the cerebral cortex of diabetic rats. There was 107 and 121% rise in thiobarbituric acid reactive substance levels in cerebral cortex and hippocampus of diabetic rats, respectively. Reduced glutathione levels and enzymatic activities of superoxide dismutase and catalase were decreased in both cerebral cortex and hippocampal regions of diabetic rat brain. Nitrite levels in cerebral cortex and hippocampus was increased by 138 and 109%, respectively. Serum tumor necrosis factor-alpha, a marker for inflammation, was found to increase by 1,100% in diabetic rats. Chronic treatment with sesamol (2, 4 and 8 mg/kg; p.o.) significantly and dose-dependently attenuated cognitive deficit, reduced acetylcholinesterase, oxidative stress and inflammation in diabetic rats. The results emphasize the involvement of oxidative stress and inflammation in the development of cognitive impairment in diabetic animals and point towards the therapeutic potential of sesamol in diabetes-associated cognitive decline.





Similar content being viewed by others
References
Anjaneyulu M, Chopra K (2004) Effect of irbesartan on the antioxidant defence system and nitric oxide release in diabetic rat kidney. Am J Nephrol 24:488–496
Arvanitakis Z, Wilson RS, Bienias JL, Evans DA, Bennett DA (2004) Diabetes mellitus and risk of Alzheimer disease and decline in cognitive function. Arch Neurol 61:661–666
Biessels GJ, Gispen WH (2005) The impact of diabetes on cognition: what can be learned from rodent models? Neurobiol Aging 26:36–41
Biessels GJ, Kamal A, Urban IJA, Spruijt BM, Erkelens DW, Gispen WH (1998) Water maze learning and hippocampal synaptic plasticity in streptozotocin-diabetic rats: effects of insulin treatment. Brain Res 800:125–135
Biessels GJ, Staekenborg S, Brunner E, Brayne C, Scheltens P (2006) Risk of dementia in diabetes mellitus: a systematic review. Lancet Neurol 5:64–74
Bloomgarden ZT (2007) Diabetic neuropathy. Diabetes Care 30:1027–1032
Bolanos JP, Cidad P, Garçia-Nogales P, Delgado-Esteban M, Fernandez E, Almeida A (2004) Regulation of glucose metabolism by nitrosative stress in neural cells. Mol Aspects Med 25:61–73
Bonnefont-Rousselot D (2002) Glucose and reactive oxygen species. Curr Opin Clin Nutr Metab Care 5:561–568
Brands AM, Kessels RP, de Haan EH, Kappelle LJ, Biessels GJ (2004) Cerebral dysfunction in type 1 diabetes: effects of insulin, vascular risk factors and blood-glucose levels. Eur J Pharmacol 490:159–168
Chavali SR, Utsunomiya T, Forse RA (2001) Increased survival after cecal ligation and puncture in mice consuming diets enriched with sesame seed oil. Crit Care Med 29:140–143
Claiborne A (1985) Catalase activity. In: Greenwald RA (ed) Handbook of methods for oxygen radical research. CRC, Boca Raton, pp 283–284
Clancy RM, Levartosky D, Leszczynska-Piziak J, Yegudin J, Abramson S (1994) Nitric oxide reacts with intracellular glutathione and activates the hexose monophosphate shunt in human neutrophils: evidence for S-nitrosoglutathione as a bioactive intermediary. PNAS 91:3680–3684
Cumiskey D, Butler MP, Moynagh PN, O’connor JJ (2007) Evidence for a role for the group I metabotropic glutamate receptor in the inhibitory effect of tumor necrosis factor-alpha on long-term potentiation. Brain Res 1136:13–19
Dobretsov M, Romanovsky D, Stimers JR (2007) Early diabetic neuropathy: triggers and mechanisms. World J Gastroenterol 13:175–191
Ellman GL, Courtney DK, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7:88–95
Evans JL, Goldfine ID, Maddux BA, Grodsky GM (2002) Oxidative stress and stress-activated signaling pathways: a unifying hypothesis of type 2 diabetes. Endocr Rev 23:599–622
Franceschi M, Cecchetto R, Minicucci F, Smizne S, Baio G, Canal N (1984) Cognitive processes in insulin-dependent diabetes. Diabetes Care 7:228–231
Friberg H, Wieloch T, Castilho RF (2002) Mitochondrial oxidative stress after global brain ischemia in rats. Neurosci Lett 334:111–114
Fukuhara M, Matsumura K, Wakisaka M, Takata Y, Sonoki K, Fujisawa K, Ansai T, Akifusa S, Fujii K, Iida M, Takehara T (2007) Hyperglycemia promotes microinflammation as evaluated by C-reactive protein in the very elderly. Intern Med 46:207–212
Fukui K, Onodera K, Shinkai T, Suzuki S, Urano S (2001) Impairment of learning and memory in rats caused by oxidative stress and aging, and changes in antioxidative defense systems. Ann NY Acad Sci 928:168–175
Gispen WH, Biessels GJ (2000) Cognition and synaptic plasticity in diabetes mellitus. Trends Neurosci 23:542–549
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N] nitrate in biological fluids. Anal Biochem 126:131–138
Hou RC, Chen YS, Chen CH, Chen YH, Jeng KC (2006) Protective effect of 1,2,4-benzenetriol on LPS-induced NO production by BV2 microglial cells. J Biomed Sci 13:89–99
Hirose M, Fukushima S, Shirai T, Hasegawa R, Kato T, Tanaka H, Asakawa E, Ito N (1990) Stomach carcinogenicity of caffeic acid, sesamol and catechol in rats and mice. Jpn J Cancer Res 81:207–212
Hoybergs YMJJ, Meert TF (2007) The effect of low-dose insulin on mechanical sensitivity and allodynia in type I diabetes neuropathy. Neurosci Lett 417:149–154
Hsu DZ, Chen KT, Li YH, Chuang YC, Liu MY (2006) Sesamol delays mortality and attenuates hepatic injury after cecal ligation and puncture in rats: role of oxidative stress. Shock 25:528–532
Hsu DZ, Chien SP, Chen KT, Liu MY (2007) The effect of sesamol on systemic oxidative stress and hepatic dysfunction in acutely iron-intoxicated mice. Shock
Jollow DJ, Mitchell JR, Zampaglione N, Gillette JR (1974) Bromobenze induced liver necrosis: protective role of glutathione and evidence for 3,4-bromobenzenoxide as the hepatotoxic intermediate. Pharmacol 11:151–169
Joshi R, Kumar MS, Satyamoorthy K, Unnikrisnan MK, Mukherjee T (2005) Free radical reactions and antioxidant activities of sesamol: pulse radiolytic and biochemical studies. J Agric Food Chem 53:2696–2703
Kapadia JG, Azuine M, Tokuda H, Takasaki M, Mukainaka T, Konoshima T, Nishino H (2002) Chemopreventive effect of resveratrol, sesamol, sesame oil and sunflower oil in the Epstein–Barr virus early antigen activation assay and the mouse skin two-stage carcinogenesis. Pharmacol Res 45:499–505
Konrad RJ, Mikolaenko I, Tolar JF, Liu K, Kudlow JE (2001) The potential mechanism of the diabetogenic action of streptozotocin: inhibition of pancreatic β-cell O-GlcNAc-selective-N-acetyl-β-d-glucosaminidase. Biochem J 356:31–41
Kono Y (1978) Generation of superoxide radical during autoxidation of hydroxylamine and an assay for superoxide dismutase. Arch Biochem Biophys 186:189–195
Kowluru RA, Kanwar M (2007) Effects of curcumin on retinal oxidative stress and inflammation in diabetes. Nutr Metab (Lond) 16:4–8
Layer PG, Alber R, Sporns O (1987) Quantitative development and molecular forms of acetylcholinesterase and butyrylcholinesterase during morphogenesi s and synaptogenesi s of chick brain and retina. J Neurochem 49:175–182
Li Z, Zhanga W, Sima AAF (2005) The role of impaired insulin/IGF action in primary diabetic encephalopathy. Brain Res 1037:12–24
Lobnig BM, Krömeke O, Optenhostert-Porst C, Wolf OT (2005) Hippocampal volume and cognitive performance in long-standing Type 1 diabetic patients without macrovascular complications. Diabetic Med 23:32–39
Mastrocola R, Restivo F, Vercellinatto I, Danni O, Brignardello E, Aragno M, Boccuzzi G (2005) Oxidative and nitrosative stress in brain mitochondria of diabetic rats. J Endocrinol 187:37–44
Mijnhout GS, Scheltens P, Diamant M, Biessels GJ, Wessels AM, Simsek S, Snoek FJ, Heine RJ (2006) Diabetic encephalopathy: a concept in need of a definition. Diabetologia 49:1447–1448
Miles WR, Root HF (1922) Psychologic tests applied to diabetic patients. Arch Int Med 30:767–777
Morris RG, Garrud P, Rawlins JN, O’Keefe J (1982) Place navigation impaired in rats with hippocampal lesions. Nature 297:681–683
Murray J, Taylor SW, Zhang B, Ghosh S (2003) Oxidative damage to mitochondrial complex I due to peroxynitrite. J Biol Chem 278:37223–37230
Nishikawa T, Edelstein D, Du XL, Yamagishi S, Matsumura T, Kaneda Y, Yorek A, Beebe D, Oates PJ, Hammes HP, Giardino I, Brownlee M (2000) Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 404:787–790
Parihar VK, Prabhakar KR, Veerapur VP, Kumar MS, Reddy YR, Joshi R, Unnikrishnan MK, Rao CM (2006) Effect of sesamol on radiation-induced cytotoxicity in Swiss albino mice. Mutat Res 611:9–16
Prasada NR, Mahesha T, Menona VP, Jeevanramb RK, Pugalendia KV (2005a) Photoprotective effect of sesamol on UVB-radiation induced oxidative stress in human blood lymphocytes in vitro. Environ Toxicol Pharmacol 20:1–5
Prasada NR, Menona VP, Vasudevb V, Pugalendia KV (2005b) Radioprotective effect of sesamol on radiation induced DNA damage, lipid peroxidation and antioxidants levels in cultured human lymphocytes. Toxicol 209:225–235
Reyes AE, Perez DR, Alvarez A, Garrido J, Gentry MK, Doctor BP, Inestrosa NC (1997) A monoclonal antibody against acetylcholinesterase inhibits the formation of amyloid fibrils induced by the enzyme. Biochem Biophys Res Commun 232:652–655
Ryan C, Vega A, Drash A (1985) Cognitive deficits in adolescents who developed diabetes early in life. Pediatrics 75:921–927
Ryan CM, Williams TM, Finegold DN, Orchard TJ (1993) Cognitive dysfunction in adults with type 1 (insulin-dependent) diabetes mellitus of long duration: effects of recurrent hypoglycaemia and other chronic complications. Diabetologia 36:329–334
Sanchez-Chavez G, Salceda R (2000) Effect of streptozotocin-induced diabetes on activities of cholinesterases in the rat retina. IUBMB Life 49:283–287
Saravia FE, Beauquis J, Revsin Y, Homo-Delarche F, Ronald de Kloet E, De Nicola AF (2006) Hippocampal neuropathology of diabetes mellitus is relieved by estrogen treatment. Cell Mol Neurobiol 26:943–957
Satoh J, Yagihashi S, Toyota T (2003) The possible role of tumor necrosis factor-alpha in diabetic polyneuropathy. Exp Diabesity Res 4:65–71
Sharma S, Anjaneyulu M, Kulkarni SK, Chopra K (2006a) Resveratrol, a polyphenolic phytoalexin, attenuates diabetic nephropathy in rats. Pharmacology 76:69–75
Sharma S, Chopra K, Kulkarni SK (2007) Effect of insulin and its combination with resveratrol or curcumin in attenuation of diabetic neuropathic pain: participation of nitric oxide and TNF-alpha. Phytother Res 21:278–283
Sharma S, Kaur IP (2006) Development and evaluation of sesamol as an antiaging agent. Int J Dermatol 45:200–208
Sharma S, Kulkarni SK, Chopra K (2006b) Curcumin, the active principle of turmeric (Curcuma longa), ameliorates diabetic nephropathy in rats. Clin Exp Pharmacol Physiol 33:940–945
Sima AAF, Kamiya H, Lia ZG (2004) Insulin, C-peptide, hyperglycemia, and central nervous system complications in diabetes. Eur J Pharmcol 490:187–197
Somfai GM, Knippel B, Ruzicska E, Stadler K, Toth M, Salacz G, Magyar K, Somogyi A (2006) Soluble semicarbazide-sensitive amine oxidase (SSAO) activity is related to oxidative stress and subchronic inflammation in streptozotocin-induced diabetic rats. Neurochem Int 48:746–752
Trudeaua F, Gagnonb S, Massicotte G (2004) Hippocampal synaptic plasticity and glutamate receptor regulation: influences of diabetes mellitus. Eur J Pharmacol 490:177–186
Tuzcu M, Baydas G (2006) Effect of melatonin and vitamin E on diabetes-induced learning and memory impairment in rats. Eur J Pharmacol 537:106–110
Wills ED (1965) Mechanism of lipid peroxide formation in animals. Biochem J 99:667–676
Uchida M, Nakajn S, Toyoshima M (1996) Antioxidative effect of sesamol and related compounds on lipid peroxidation. Biol Pharm Bull 19:623–626
Acknowledgments
The Senior Research Fellowship (Anurag Kuhad) of the Indian Council of Medical Research (ICMR), New Delhi, is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kuhad, A., Chopra, K. Effect of sesamol on diabetes-associated cognitive decline in rats. Exp Brain Res 185, 411–420 (2008). https://doi.org/10.1007/s00221-007-1166-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00221-007-1166-y