Abstract
Purpose
We evaluated the incidence, prevalence, and patterns of triptan use with a special focus on patients who also received cardiovascular drugs, considered as a deviation from appropriate triptan use.
Methods
We collected data on prescriptions reimbursed in between 2006 and 2008 in an Italian region. Patients receiving at least one prescription of triptans from January to December 2007 were divided in two populations: new users (without triptan prescriptions in 2006), and already in treatment. All patients were followed for 12 months in terms of both triptan use and cardiovascular coprescriptions.
Results
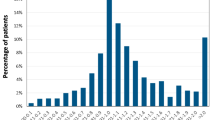
One-year prevalence of triptan use was 0.8%, whereas the incidence was 0.4%. New users accounted for 47.5% of total users (34,915). The percentages of very frequent users (>120 and >180 dosage unit per year) were about double among those already in treatment (26.6% and 15.3%, respectively) and new users (1% and 0.3%; p < 0.01). Patients starting with the lowest dose of tablet formulation were more likely to interrupt therapy after their first prescription (p < 0.01). Many users aged >65 received concomitant cardiovascular therapies: 36.6% among new users and 64.3% for already in treatment (p < 0.01). In both groups, about 5% of elderly patients received coprescriptions, suggesting a high deviation from appropriate triptan use.
Conclusions
A higher percentage of very frequent users was detected in patients already in treatment compared with new users. Moreover, the percentage of nonnegligible triptan recipients were in age groups for which the drug is not recommended by the product label (≤18 years and >65) and who had cardiovascular coprescriptions suggestive of vasoconstrictive risk.

Similar content being viewed by others
References
Kurth T, Kase CS, Schurks M, Tzourio C, Buring JE (2010) Migraine and risk of haemorrhagic stroke in women: prospective cohort study. BMJ 341:c3659
Stovner LJ, Andree C (2010) Prevalence of headache in Europe: a review for the Eurolight project. J Headache Pain 11:289–299
Duncan CW, Watson DP, Stein A (2008) Diagnosis and management of headache in adults: summary of SIGN guideline. BMJ 337:a2329
Pascual J, Mateos V, Roig C, Sanchez-Del-Rio M, Jimenez D (2007) Marketed oral triptans in the acute treatment of migraine: a systematic review on efficacy and tolerability. Headache 47:1152–1168
Loder E, Goldstein R, Biondi D (2005) Placebo effects in oral triptan trials: the scientific and ethical rationale for continued use of placebo controls. Cephalalgia 25:124–131
Pavone E, Banfi R, Vaiani M, Panconesi A (2007) Patterns of triptans use: a study based on the records of a community pharmaceutical department. Cephalalgia 27:1000–1004
Panconesi A, Pavone E, Vacca F, Vaiani M, Banfi R (2008) Triptans in the Italian population: a drug utilization study and a literature review. J Headache Pain 9:71–76
Panconesi A, Pavone E, Franchini M, Mennuti N, Bartolozzi M, Guidi L et al (2010) Triptans: low utilization and high turnover in the general population. Cephalalgia 30:576–581
Ifergane G, Wirguin I, Shvartzman P (2006) Triptans–why once? Headache 46:1261–1263
Hagen K, Jensen R, Boe MG, Stovner LJ (2010) Medication overuse headache: a critical review of end points in recent follow-up studies. J Headache Pain 11:373–377
Valguarnera F, Tanganelli P (2010) The efficacy of withdrawal therapy in patients with chronic daily headache and medication overuse following prophylaxis with topiramate and amitriptyline. Neurol Sci 31(Suppl 1):S175–S177
Pascual J, Colas R, Castillo J (2001) Epidemiology of chronic daily headache. Curr Pain Headache Rep 5:529–536
Gudmundsson LS, Scher AI, Aspelund T, Eliasson JH, Johannsson M, Thorgeirsson G et al (2010) Migraine with aura and risk of cardiovascular and all cause mortality in men and women: prospective cohort study. BMJ 341:c3966
Berger K, Evers S (2010) Migraine with aura and the risk of increased mortality. BMJ 341:c4410
Jamieson DG (2002) The safety of triptans in the treatment of patients with migraine. Am J Med 112:135–140
The International Classification of Headache Disorders: 2nd edition. Cephalalgia 2004; 24(Suppl 1):9–160
OsMed (Osservatorio Nazionale sull’Impiego dei Medicinali). L’uso dei farmaci in Italia. Rapporto Nazionale anno 2007. Roma, Il Pensiero Scientifico Editore, 2008
Becker C, Brobert GP, Almqvist PM, Johansson S, Jick SS, Meier CR (2008) Migraine incidence, comorbidity and health resource utilization in the UK. Cephalalgia 28:57–64
Lipton RB, Stewart WF, Simon D (1998) Medical consultation for migraine: results from the American Migraine Study. Headache 38:87–96
Stang PE, Osterhaus JT, Celentano DD (1994) Migraine. Patterns of healthcare use. Neurology 44:S47–S55
Steiner TJ, MacGregor EA, Davies PTG (2010) Guidelines for all healthcare professionals in the diagnosis and management of migraine, tension-type, cluster and medication-overuse headache. British Association for the Study of Headache
Ashkenazi A, Schwedt T (2011) Cluster headache–acute and prophylactic therapy. Headache 51:272–286
Lohman JJ, van der Kuy-de Ree MM (2005) Patterns of specific antimigraine drug use—a study based on the records of 18 community pharmacies. Cephalalgia 25:214–218
Ferrari MD, Roon KI, Lipton RB, Goadsby PJ (2001) Oral triptans (serotonin 5-HT(1B/1D) agonists) in acute migraine treatment: a meta-analysis of 53 trials. Lancet 358:1668–1675
Sondergaard J, Foged A, Kragstrup J, Gaist D, Gram LF, Sindrup SH et al (2006) Intensive community pharmacy intervention had little impact on triptan consumption: a randomized controlled trial. Scand J Prim Health Care 24:16–21
Acknowledgments
The study was supported by funds from the Emilia Romagna Region and the University of Bologna. The opinions expressed herein by AP do not necessarily reflect those of the Emilia Romagna Health Authority.
Competing interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Biagi, C., Poluzzi, E., Roberto, G. et al. Pattern of triptan use and cardiovascular coprescription: a pharmacoepidemiological study in Italy. Eur J Clin Pharmacol 67, 1283–1289 (2011). https://doi.org/10.1007/s00228-011-1076-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-011-1076-6