Abstract
Purpose
Cancer cachexia is characterized by hypoalbuminemia and with the hepatic production of acute-phase proteins in response to malignant growth. The aim of this study was to evaluate the influence of cachexia on the pharmacokinetic disposition of and clinical responses to oxycodone in cancer patients.
Methods
Forty-seven Japanese patients receiving oxycodone extended-release tablets as a starting opioid for cancer pain were enrolled in this study. Cachexia was evaluated using the Glasgow Prognostic Score (GPS). Predose plasma concentrations of oxycodone and noroxycodone were determined at the titration dose.
Results
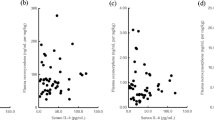
Seven patients had a GPS of 0, 21 a GPS of 1, and 19 had a GPS of 2. A higher GPS was significantly correlated with a higher oxycodone concentration and a lower concentration ratio of noroxycodone to oxycodone and significantly associated with a lower incidence of dose escalation and a higher incidence of central adverse reactions. Serum albumin, but not α1-acid glycoprotein and C-reactive protein, was inversely correlated with the free fraction of oxycodone. Serum albumin concentration was significantly associated with the incidence of dose escalation. In contrast, the free fraction of oxycodone and acute-phase proteins were not related to the clinical responses.
Conclusions
Cachexia had an effect on oxycodone metabolism and the clinical responses to oxycodone. The observed reduction in serum albumin concentration was associated with dose escalation. These findings suggest that cachexia affects the clinical responses to oxycodone through metabolic and nutritional disorders in cancer patients.


Similar content being viewed by others
References
Bosaeus I, Daneryd P, Svanberg E, Lundholm K (2001) Dietary intake and resting energy expenditure in relation to weight loss in unselected cancer patients. Int J Cancer 93:380–383
Fearon KC, Falconer JS, Slater C, McMillan DC, Ross JA, Preston T (1998) Albumin synthesis rates are not decreased in hypoalbuminemic cachectic cancer patients with an ongoing acute-phase protein response. Ann Surg 227:249–254
Fearon KC, Barber MD, Falconer JS, McMillan DC, Ross JA, Preston T (1999) Pancreatic cancer as a model: inflammatory mediators, acute-phase response, and cancer cachexia. World J Surg 23:584–588
McMillan DC (2008) An inflammation-based prognostic score and its role in the nutrition-based management of patients with cancer. Proc Nutr Soc 67:257–262
Lelli G, Montanari M, Gilli G, Scapoli D, Antonietti C, Scapoli D (2003) Treatment of the cancer anorexia-cachexia syndrome: a critical reappraisal. J Chemother 15:220–225
Lugo RA, Kern SE (2004) The pharmacokinetics of oxycodone. J Pain Palliat Care Pharmacother 18:17–30
Tien JH (1991) Transdermal-controlled administration of oxycodone. J Pharm Sci 80:741–743
Leow KP, Wright AW, Cramond T, Smith MT (1993) Determination of the serum protein binding of oxycodone and morphine using ultrafiltration. Ther Drug Monit 15:440–447
Pöyhiä R, Seppälä T (1994) Liposolubility and protein binding of oxycodone in vitro. Pharmacol Toxicol 74:23–27
Tisdale MJ (2005) Molecular pathways leading to cancer cachexia. Physiology 20:340–348
Slaviero KA, Clarke SJ, Rivory LP (2003) Inflammatory response: an unrecognised source of variability in the pharmacokinetics and pharmacodynamics of cancer chemotherapy. Lancet Oncol 4:224–232
Rivory LP, Slaviero KA, Clarke SJ (2002) Hepatic cytochrome P450 3A drug metabolism is reduced in cancer patients who have an acute-phase response. Br J Cancer 87:277–280
Lalovic B, Phillips B, Risler LL, Howald W, Shen DD (2004) Quantitative contribution of CYP2D6 and CYP3A to oxycodone metabolism in human liver and intestinal microsomes. Drug Metab Dispos 32:447–454
Forrest LM, McMillan DC, McArdle CS, Angerson WJ, Dunlop DJ (2003) Evaluation of cumulative prognostic scores based on the systemic inflammatory response in patients with inoperable non-small-cell lung cancer. Br J Cancer 89:1028–1030
Naito T, Takashina Y, Yamamoto K, Tashiro M, Ohnishi K, Kagawa Y, Kawakami J (2011) CYP3A5*3 affects plasma disposition of noroxycodone and dose escalation in cancer patients receiving oxycodone. J Clin Pharmacol 51:1529–1538
Mercadante S, Fulfaro F, Casuccio A, Barresi L (1999) Investigation of an opioid response categorization in advanced cancer patients. J Pain Symptom Manage 18:347–352
Pascussi JM, Gerbal-Chaloin S, Pichard-Garcia L, Daujat M, Fabre JM, Maurel P, Vilarem MJ (2000) Interleukin-6 negatively regulates the expression of pregnane X receptor and constitutively activated receptor in primary human hepatocytes. Biochem Biophys Res Commun 274:707–713
Muntané-Relat J, Ourlin JC, Domergue J, Maurel P (1995) Differential effects of cytokines on the inducible expression of CYP1A1, CYP1A2, and CYP3A4 in human hepatocytes in primary culture. Hepatology 22:1143–1153
Leow KP, Smith MT (1994) The antinociceptive potencies of oxycodone, noroxycodone and morphine after intracerebroventricular administration to rats. Life Sci 54:1229–1236
Liu JG, Anand KJ (2001) Protein kinases modulate the cellular adaptations associated with opioid tolerance and dependence. Brain Res Brain Res Rev 38:1–19
Kaiko RF, Benziger DP, Fitzmartin RD, Burke BE, Reder RF, Goldenheim PD (1996) Pharmacokinetic-pharmacodynamic relationships of controlled-release oxycodone. Clin Pharmacol Ther 59:52–61
Zwisler ST, Enggaard TP, Mikkelsen S, Brosen K, Sindrup SH (2010) Impact of the CYP2D6 genotype on post-operative intravenous oxycodone analgesia. Acta Anaesthesiol Scand 54:232–240
Samer CF, Daali Y, Wagner M, Hopfgartner G, Eap CB, Rebsamen MC, Rossier MF, Hochstrasser D, Dayer P, Desmeules JA (2010) Genetic polymorphisms and drug interactions modulating CYP2D6 and CYP3A activities have a major effect on oxycodone analgesic efficacy and safety. Br J Pharmacol 160:919–930
Wood LJ, Nail LM, Perrin NA, Elsea CR, Fischer A, Druker BJ (2006) The cancer chemotherapy drug etoposide (VP-16) induces proinflammatory cytokine production and sickness behavior-like symptoms in a mouse model of cancer chemotherapy-related symptoms. Biol Res Nurs 8:157–169
Myers JS (2008) Proinflammatory cytokines and sickness behavior: implications for depression and cancer-related symptoms. Oncol Nurs Forum 35:802–807
de Vries HE, Blom-Roosemalen MC, van Oosten M, de Boer AG, van Berkel TJ, Breimer DD, Kuiper J (1996) The influence of cytokines on the integrity of the blood–brain barrier in vitro. J Neuroimmunol 64:37–43
Saija A, Princi P, Lanza M, Scalese M, Aramnejad E, De Sarro A (1995) Systemic cytokine administration can affect blood–brain barrier permeability in the rat. Life Sci 56:775–784
Tallgren M, Olkkola KT, Seppälä T, Höckerstedt K, Lindgren L (1997) Pharmacokinetics and ventilatory effects of oxycodone before and after liver transplantation. Clin Pharmacol Ther 61:655–661
Andreassen TN, Eftedal I, Klepstad P, Davies A, Bjordal K, Lundström S, Kaasa S, Dale O (2012) Do CYP2D6 genotypes reflect oxycodone requirements for cancer patients treated for cancer pain? A cross-sectional multicentre study. Eur J Clin Pharmacol 68:55–64
Heiskanen T, Mätzke S, Haakana S, Gergov M, Vuori E, Kalso E (2009) Transdermal fentanyl in cachectic cancer patients. Pain 144:218–222
Labroo RB, Paine MF, Thummel KE, Kharasch ED (1997) Fentanyl metabolism by human hepatic and intestinal cytochrome P450 3A4: implications for interindividual variability in disposition, efficacy, and drug interactions. Drug Metab Dispos 25:1072–1080
Acknowledgments
This work was supported by a Grant-in-Aid for Scientific Research (C) from the Japanese Ministry of Education, Culture, Sports, Science and Technology (grant number 21590155).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Naito, T., Tashiro, M., Yamamoto, K. et al. Impact of cachexia on pharmacokinetic disposition of and clinical responses to oxycodone in cancer patients. Eur J Clin Pharmacol 68, 1411–1418 (2012). https://doi.org/10.1007/s00228-012-1266-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-012-1266-x