Abstract
Objective
The pharmacokinetics of lamotrigine (LTG) varies significantly among individuals and particularly among different ethnic groups. This is in part due to the presence of genetic polymorphisms affecting genes that metabolize LTG. UGT1A4 is a major metabolizing enzyme of LTG. The aim of this study was to investigate the effect of two UGT1A4 gene polymorphisms, UGT1A4 (70C > A) and UGT1A4 (142 T > G), on the levels and efficacy of LTG in Han Chinese patients with epilepsy.
Methods
The study cohort comprised 106 Han Chinese patients patients with epilepsy who were receiving LTG monotherapy. Blood samples were taken and LTG levels measured. The presence of UGT1A4 (70C > A) and UGT1A4 (142 T > G) was determined. The therapeutic efficacy of LTG at the 1-year time-point was assessed.
Results
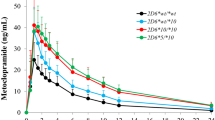
All patients were homozygous for the CC genotype of UGT1A4 (70C > A), while the distribution of UGT1A4 (142 T > G) varied among patients. Two patients had a single nucleotide deletion at position 127 (UGT1A4 127delA). To evaluate the effect of the UGT1A4 (142 T > G) polymorphism on LTG pharmacokinetics, patients were divided into two groups. Group A included patients with the 142TG or 142GG polymorphism and Group B patients had the 142TT polymorphism. The normalized blood concentration and the efficacy of LTG were higher in Group B patients than in Group A patients (P < 0.05). The two patients with UGT1A4 127delA genotype had extremely high blood levels of LTG, and treatment was discontinued in one of these patients due to a severe LTG-associated rash.
Conclusion
Patients with the UGT1A4 142TT polymorphism had a higher blood LTG concentration and better therapeutic efficacy, suggesting that this polymorphism influences LTG activity. The UGT1A4 127delA polymorphism significantly affected LTG levels and increased one of our patient's susceptibility to LTG-related adverse events.


Similar content being viewed by others
References
Pellock JM (2007) Overview of lamotrigine and the new antiepileptic drugs: the challenge. J Child Neurol 12(1):S48–S52
Johannessen SI, Tomson T (2006) Pharmacokinetic variability of newer antiepileptic drugs: when is monitoring needed? Clin Pharmacokinet 45:1061–1075
Milovanovic JR, Jankovic SM (2009) Population pharmacokinetics of lamotrigine in patients with epilepsy. Int J Clin Pharmacol Ther 47:752–760
Punyawudho B, Ramsay RE, Macias FM, Rowan AJ, Collins JF, Brundage RC, Birnbaum AK (2008) Population pharmacokinetics of lamotrigine in elderly patients. J Clin Pharmacol 48:455–463
Rivas N, Buelga DS, Elger CE, Santos-Borbujo J, Otero MJ, Dominguez-Gil A, Garcia MJ (2008) Population pharmacokinetics of lamotrigine with data from therapeutic drug monitoring in German and Spanish patients with epilepsy. Ther Drug Monit 30:483–489
Gulcebi MI, Ozkaynakci A, Goren MZ, Aker RG, Ozkara C, Onat FY (2011) The relationship between UGT1A4 polymorphism and serum concentration of lamotrigine in patients with epilepsy. Epilepsy Res 95:1–8
Franciotta D, Kwan P, Perucca E (2009) Genetic basis for idiosyncratic reactions to antiepileptic drugs. Curr Opin Neurol 2:144–149
Benoit-Binomics MO, Adam JP, Bernard O et al (2009) A pharmacogenetics study of the human glucuronosyltransferase UGT1A4. Pharmacogenet Genomics 19(12):945–954
Yea SS, Lee SS, Kim WY, Liu KH, Kim H, Shon JH, Cha IJ, Shin JG (2008) Genetic variations and haplotypes of UDP-glucuronosyltransferase 1A locus in a Korean population. Ther Drug Monit 30(1):23–34
Thomas SS, Li SS, Lampe JW, Potter JD, Bigler J (2006) Genetic variability, haplotypes, and htSNPs for exons 1 at the human UGT1A locus. Hum Mutat 27(7):717
Hakooz N, Alzubiedi S, Yousef AM, Arafat T, Dajani R, Ababneh N, Ismail S (2012) UDP-glucuronosyltransferase 1A4 (UGT1A4) polymorphisms in a Jordanian population. Mol Biol Rep 39(7):7763–7768
Engel J Jr (2001) A proposed diagnostic scheme for people with epileptic seizures and with epilepsy: report of the ILAE Task Force on Classification and Terminology. Epilepsia 42(6):796–803
Engel J Jr, Van Ness PC, Rasmussen TB, Ojemann LM (1993) Outcome with respect to epileptic seizures. In: Engel J Jr (ed) Surgical treatment of the epilepsies, 2nd edn. Raven Press, New York, pp 609–621
Rowland A, Elliot DJ, Williams JA et al (2006) In vitro characterization of lamotrigine N2-glucuronidation and the lamotrigine-valproic acid interation. Drug Metab Dispos 34:1055–1062
Hiller A, Nguyen N, Strassburg CP et al (1999) Retigabine N-glucuronidation and its potential role in enterohepatic circulation. Drug Metab Dispos 27(5):605–612
Posner J, Cohen AF, Land G et al (1989) The pharmacokinetics of lamotrigine (BW430C) in healthy subjects with unconjugated hyperbilirubinemia (Gilbert’s syndrome). Br J Clin Pharmacol 28:117–120
Kubota T, Lewis BC, Elliot DJ et al (2007) Critical roles of residues 36 and 40 in the phenol and tertiary amine aglycone substrate selectivities of UDP-glucuronosyltransferases 1A3 and 1A4. Mol Pharmacol 72(4):1054–1062
Singkham N, Towanabut S, Lertkachatarn S et al (2013) Influence of the UGT 2B7-161C > T polymorphism on the population pharmacokinetics of lamotrigine in Thai patients. Eur J Clin Pharmacol 69(6):1285–1291
Ehmer U, Vogel A, Schutte JK, Krone B, Manns MP, Sttassburg CP (2004) Variation of hepatic glucuronidation:novel functional polymorphisms of the UDP glucuronosyltransferase UGT1A4. Hepatology 39(4):970–977
Wiener D, Doerge DR, Fang JL et al (2004) Characterization of N-glucuronidation of the lung carcinogen 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanol (NNAL) in human liver:importance of UDP-glucuronosyltransferase 1A4. Drug Metab Dispos 32(1):72–79
Mori A, Maruo Y, Iwai M, Sato H, Takeuchi Y (2005) UDP-glucuronosyltransferase 1A4 polymorphisms in a Japanese population and kinetics of clozapine glucuronidation. Drug Metab Dispos 33(5):672–675
Saeki M, Saito Y, Jinno H et al (2005) Genetic variations and haplotypes of UGT1A4 in a Japanese population. Drug Metab Pharmacokinet 20(2):144–151
Erickson-Ridout KK, Sun D, Lazarus P (2012) Glucuronidation of the second-generation antipsychotic clozapine and its active metabolite N-desmethylclozapine potential importance of the UGT1A1 A(TA)7TAA and UGT1A4 L48V polymorphisms. Pharmacogenet Genomics 22:561–576
Ghotbi R, Mannheimer B, Aklillu E, Suda A et al (2010) Carriers of the UGT1A4 142 T > G gene variant are predisposed to reduced olanzapine exposure—an impact similar to male gender or smoking in schizophrenic patients. Eur J Pharmacol 66:465–474
Sun D, Chen G, Dellinger RW, Duncan K et al (2006) Characterization of tamoxifen and 4-hydroxytamoxifen glucuronidation by human UGT1A4 variants. Breast Can Res 8(R50):1–11
Saeki M, Saito Y, Jinno H et al (2005) Genetic variations and haplotypes of UGT1A4 in a Japanese population. Drug Metab Pharmacokinet 20(2):144–151
Duan JB, Wainwright MS, Comeron JM et al (2003) Synonymous mutations in the human dopamine receptor D2(DRD2) affect mRNA stability and synthesis of the receptor. Hum Mol Genet 12:205–216
Acknowledgments
This work is supported by grants from Jilin Province Science and Technology Program (3D511Z343430) and Basic Scientific Research Funds of Jilin University (450060491602).
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 13 kb)
Rights and permissions
About this article
Cite this article
Chang, Y., Yang, Ly., Zhang, Mc. et al. Correlation of the UGT1A4 gene polymorphism with serum concentration and therapeutic efficacy of lamotrigine in Han Chinese of Northern China. Eur J Clin Pharmacol 70, 941–946 (2014). https://doi.org/10.1007/s00228-014-1690-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-014-1690-1