Abstract
Purpose
In this study, the risk factors associated with sodium overcorrection were investigated with an optimal cutoff for baseline serum sodium for use in daily clinical practice.
Methods
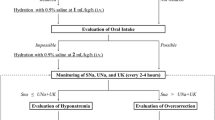
Electronic medical records of patients who received tolvaptan for non-hypovolemic hyponatremia were reviewed. Demographic and clinical data including age, sex, weight, height, comorbidity, cause of hyponatremia, hypertonic saline use, and comedication were collected. Baseline laboratory parameters measured included serum sodium, serum potassium, serum creatinine, blood urea nitrogen, serum tonicity, ALT, AST, and urine osmolality. The primary outcome was the overcorrection of serum sodium, which was defined as an increase in serum sodium by more than 10 mmol/L in 24 h.
Results
From a total of 77 patients included in the analysis, 24 (31.2%) showed sodium overcorrection (> 10 mmol/L/24 h); 2 (2.6%) in heart failure cohort, 17 (22.1%) in SIADH cohort, and 5 (6.5%) in unknown cause cohort. More than half of patients (51.9%) were administered hypertonic saline prior to tolvaptan. Hypertension, cancer, diuretics, baseline serum sodium, and SIADH were associated with the risk of overcorrection in the univariable analysis. Significant factors for the overcorrection from multivariable analysis were lower body mass index, presence of cancer (adjusted odds ratio, 10.87; 95% CI, 1.23–96.44), and lower serum sodium at baseline (adjusted odds ratio, 0.76 for every 1 mEq/L increase; 95% CI, 0.61–0.94).
Conclusion
The overcorrection of hyponatremia in non-hypovolemic patients treated with tolvaptan was significantly associated with lower body mass index, presence of cancer, and lower serum sodium at baseline. In subgroup analysis using SIADH patients, baseline sodium and cancer were found to be significant factors of overcorrection.


Similar content being viewed by others
References
Filippatos T, Elisaf M, Liamis G (2018) Pharmacological management of hyponatremia. Expert Opin Pharmacother 19(12):1337–1344. https://doi.org/10.1080/14656566.2018.1504920
Sterns RH, Silver SM (2016) Complications and management of hyponatremia. Curr Opin Nephrol Hypertens 25(2):114–119. https://doi.org/10.1097/mnh.0000000000000200
Sahay M, Sahay R (2014) Hyponatremia: a practical approach. Indian J Endocrinol Metab 18(6):760–771. https://doi.org/10.4103/2230-8210.141320
Adrogué HJ, Madias NE (2012) The challenge of hyponatremia, 10.1681/ASN.2012020128. J Am Soc Nephrol 23(7):1140–1148
Verbalis JG, Goldsmith SR, Greenberg A, Korzelius C, Schrier RW, Sterns RH, Thompson CJ (2013) Diagnosis, evaluation, and treatment of hyponatremia: expert panel recommendations. Am J Med 126(10 Suppl 1):S1–S42. https://doi.org/10.1016/j.amjmed.2013.07.006
Aratani S, Hara M, Nagahama M, Taki F, Futatsuyama M, Tsuruoka S, Komatsu Y (2017) A low initial serum sodium level is associated with an increased risk of overcorrection in patients with chronic profound hyponatremia: a retrospective cohort analysis. BMC Nephrol 18(1):316. https://doi.org/10.1186/s12882-017-0732-1
Spasovski G, Vanholder R, Allolio B, Annane D, Ball S, Bichet D, Decaux G, Fenske W, Hoorn EJ, Ichai C, Joannidis M, Soupart A, Zietse R, Haller M, van der Veer S, Van Biesen W, Nagler E, Group obotHGD (2014) Clinical practice guideline on diagnosis and treatment of hyponatraemia. Nephrol Dial Transplant 29(suppl_2):i1–i39. https://doi.org/10.1093/ndt/gfu0
Malhotra I, Gopinath S, Janga KC, Greenberg S, Sharma SK, Tarkovsky R (2014) Unpredictable nature of tolvaptan in treatment of hypervolemic hyponatremia: case review on role of vaptans. Case Rep Endocrinol 2014:807054–807054. https://doi.org/10.1155/2014/807054
Han SW, Yi JH, Kang KP, Kim HY, Kim SW, Choi HY, Ha SK, Kim GH, Kim YW, Jeong KH, Shin SK, Kim HJ (2018) Safety and efficacy of tolvaptan in Korean patients with hyponatremia caused by the syndrome of inappropriate antidiuretic hormone. J Korean Med Sci 33(15):e112. https://doi.org/10.3346/jkms.2018.33.e112
Lexicomp (2019) Tolvaptan. Wolters Kluwer Clinical Drug Information, Inc. https://online.lexi.com/lco/action/doc/retrieve/docid/multinat_f/4668778?searchUrl=%2Flco%2Faction%2Fsearch%3Fq%3Dtolvaptan%26t%3Dname%26va%3Dtolvaptan#cyt. Accessed July 24 2019
Park SH, Goo JM, Jo CH (2004) Receiver operating characteristic (ROC) curve: practical review for radiologists. Korean J Radiol 5(1):11–18. https://doi.org/10.3348/kjr.2004.5.1.11
Umbrello M, Mantovani ES, Formenti P, Casiraghi C, Ottolina D, Taverna M, Pezzi A, Mistraletti G, Iapichino G (2016) Tolvaptan for hyponatremia with preserved sodium pool in critically ill patients. Ann Intensive Care 6(1):1. https://doi.org/10.1186/s13613-015-0096-2
Albright RC (2015) Hyponatremia management: walking the tightrope without a net. Mayo Clin Proc 90(10):1320–1322. https://doi.org/10.1016/j.mayocp.2015.08.013
Shoaf SE, Bricmont P, Dandurand A (2017) Low-dose tolvaptan PK/PD: comparison of patients with hyponatremia due to syndrome of inappropriate antidiuretic hormone secretion to healthy adults. Eur J Clin Pharmacol 73(11):1399–1408. https://doi.org/10.1007/s00228-017-2302-7
Imamura T, Kinugawa K, Minatsuki S, Muraoka H, Kato N, Inaba T, Maki H, Hatano M, Yao A, Komuro I (2014) Urine sodium excretion after tolvaptan administration is dependent upon baseline serum sodium levels: a possible explanation for the improvement of hyponatremia with scarce chance of hypernatremia by a vasopressin receptor antagonist. Int Heart J 55(2):131–137
Velez JC, Dopson SJ, Sanders DS, Delay TA, Arthur JM (2010) Intravenous conivaptan for the treatment of hyponatraemia caused by the syndrome of inappropriate secretion of antidiuretic hormone in hospitalized patients: a single-centre experience. Nephrol Dialysis Transplant : official publication of the European Dialysis and Transplant Association - European Renal Association 25(5):1524–1531. https://doi.org/10.1093/ndt/gfp731
George JC, Zafar W, Bucaloiu ID, Chang AR (2018) Risk factors and outcomes of rapid correction of severe hyponatremia. Clin J Am Soc Nephrol 13(7):984–992. https://doi.org/10.2215/cjn.13061117
Hanley MJ, Abernethy DR, Greenblatt DJ (2010) Effect of obesity on the pharmacokinetics of drugs in humans. Clin Pharmacokinet 49(2):71–87. https://doi.org/10.2165/11318100-000000000-00000
Gharaibeh KA, Brewer JM, Agarwal M, Fulop T (2015) Risk factors, complication and measures to prevent or reverse catastrophic sodium overcorrection in chronic hyponatremia. Am J Med Sci 349(2):170–175. https://doi.org/10.1097/MAJ.0000000000000324
Humayun MA, Cranston IC (2017) In-patient tolvaptan use in SIADH: care audit, therapy observation and outcome analysis. BMC Endocr Disord 17(1):69. https://doi.org/10.1186/s12902-017-0214-2
Petereit C, Zaba O, Teber I, Lüders H, Grohé C (2013) A rapid and efficient way to manage hyponatremia in patients with SIADH and small cell lung cancer: treatment with tolvaptan. BMC Pulm Med 13(1):55. https://doi.org/10.1186/1471-2466-13-55
Schrier RW, Gross P, Gheorghiade M, Berl T, Verbalis JG, Czerwiec FS, Orlandi C (2006) Tolvaptan, a selective oral vasopressin V2-receptor antagonist, for hyponatremia 355 (20):2099–2112. https://doi.org/10.1056/NEJMoa065181
Funding
This research was supported by the Medical Research Center Program (2017R1A5A2015541) of the National Research Foundation of Korea (NRF) funded by the Korean government (MSIP).
Author information
Authors and Affiliations
Contributions
All authors have contributed significantly to the work and have read and approved the manuscript for publication. Yukyung Kim and Hye Sun Gwak were responsible for the study concept and design. Yukyung Kim and Nari Lee participated in data collection. Nari Lee and Kyung Eun Lee analyzed the data. Kyung Eun Lee and Hye Sun Gwak contributed to the manuscript writing and discussion.
Corresponding authors
Ethics declarations
This study was approved by the Institutional Review Board of the Catholic University of Korea Seoul St. Mary’s Hospital (IRB number KC19RESI0327).
Conflict of interest
The authors declare that there is no conflict of interest.
Data availability
The datasets during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 337 kb)
Rights and permissions
About this article
Cite this article
Kim, Y., Lee, N., Lee, K.E. et al. Risk factors for sodium overcorrection in non-hypovolemic hyponatremia patients treated with tolvaptan. Eur J Clin Pharmacol 76, 723–729 (2020). https://doi.org/10.1007/s00228-020-02848-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00228-020-02848-6