Abstract
Objective: With the aim to obtain a premixed rapid-acting insulin with a serum insulin profile more closely resembling the endogenous meal-stimulated serum insulin profiles, a 30/70 (rapid/intermediate-acting) premixed suspension of the rapid-acting insulin analogue insulin aspart (BIAsp30) was compared with a similar premixed suspension of biphasic human insulin 30/70 (BHI30) after a single subcutaneous injection.
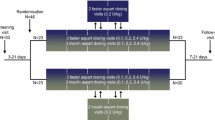
Methods: The study had a randomised, double-blind, two-period crossover design. Twenty-four healthy male subjects received a single subcutaneous dose of either 0.2 U · kg−1 bodyweight of BIAsp30 or BHI30 on two study days.
Results: BIAsp30 was absorbed faster than BHI30, as reflected in the area under the insulin concentration-time curve from 0 to 90 min after dosing [AUC(0–90 min)]. This was significantly larger for BIAsp30 than for BHI30 (1403 ± 372 versus 752 ± 191 mU · l−1 · min−1 [mean ± SD]; P < 0.0001). Furthermore, the time to maximum serum insulin concentration (tmax) of BIAsp30 was approximately half the tmax of BHI30 (60 [45–70] versus 110 [90–180] min [median, interquartile range]; P=0.0001) and the maximum insulin concentration (Cmax) was significantly higher for BIAsp30 than for BHI30 (23.4 ± 5.3 versus 15.5 ± 3.7 mU · l−1 [mean ± SD]; P < 0.0001). The serum glucose profiles showed a significantly earlier onset of the glucose-lowering effect following BIAsp30 than following BHI30.
Conclusions: The improved absorption properties of soluble insulin aspart in its premixed formulation provide a basis for a more efficient meal-related glucose control and immediate pre-meal delivery when compared with a similar human premixed insulin in the treatment of diabetes mellitus.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 22 November 1999 / Accepted in revised form: 7 April 2000
Rights and permissions
About this article
Cite this article
Jacobsen, L., Søgaard, B. & Riis, A. Pharmacokinetics and pharmacodynamics of a premixed formulation of soluble and protamine-retarded insulin aspart. E J Clin Pharmacol 56, 399–403 (2000). https://doi.org/10.1007/s002280000159
Issue Date:
DOI: https://doi.org/10.1007/s002280000159