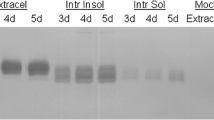
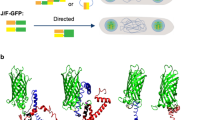
Abstract
Unraveling the characteristics and putative applications of naturally occurring protein aggregates has received an increasing interest during the last years. For example, the finding that the proteins embedded within bacterial inclusion bodies are, at least partially, biologically functional opened new opportunities for their rational design and application as naturally self-immobilized biocatalysts or as new drug delivery systems (“nanopills”). In another scenario, it is well established that “conformational diseases” are caused by misfolding and protein aggregation in different cells and tissues. The presence of such protein aggregates is a hallmark of these conditions, therefore becoming an excellent target for new therapeutic approaches for such devastating pathologies. Aggresomes are protein aggregates found in eukaryotic cells when the intracellular protein degradation machinery is overtitered. These protein-based nanoparticles are increasingly becoming excellent models in studies aimed to obtain a better understanding and control over protein aggregation processes in eukaryotic cells. In this work, we focus on some of the latest findings in the field of putative aggresome applications in biotechnology, as a new type of self-assembled immobilized biocatalysts, and in nanomedicine, mainly on their relationship with conformational diseases and the rational design of better therapeutics through a deeper understanding of protein aggregation processes.


Similar content being viewed by others
References
Askanas V, Engel WK (2001) Inclusion-body myositis: newest concepts of pathogenesis and relation to aging and Alzheimer disease. J Neuropathol Exp Neurol 60:1–14
Askanas V, Engel WK (2008) Inclusion-body myositis: muscle-fiber molecular pathology and possible pathogenic significance of its similarity to Alzheimer’s and Parkinson’s disease brains. Acta Neuropathol 116:583–595
Askanas V, Engel WK (2003) Proposed pathogenetic cascade of inclusion-body myositis: importance of amyloid-beta, misfolded proteins, predisposing genes, and aging. Curr Opin Rheumatol 15:737–744
Aston-Mourney K, Zraika S, Udayasankar J, Subramanian SL, Green PS, Kahn SE, Hull RL (2013) Matrix metalloproteinase-9 reduces islet amyloid formation by degrading islet amyloid polypeptide. J Biol Chem 288:3553–3559
Astruc D, Lu F, Aranzaes JR (2005) Nanoparticles as recyclable catalysts: the frontier between homogeneous and heterogeneous catalysis. Angew Chem Int Ed Engl 44:7852–7872
Bartolini M, Andrisano V (2010) Strategies for the inhibition of protein aggregation in human diseases. Chembiochem 11:1018–1035
Brown IR (2007) Heat shock proteins and protection of the nervous system. Ann N Y Acad Sci 1113:147–158
Cano-Garrido O, Rodriguez-Carmona E, Díez-Gil C, Vazquez E, Elizondo E, Cubarsi R, Seras-Franzoso J, Corchero JL, Rinas U, Ratera I, Ventosa N, Veciana J, Villaverde A, Garcia-Fruitos E (2013) Supramolecular organization of protein-releasing functional amyloids solved in bacterial inclusion bodies. Acta Biomater 9:6134–6142
Cao L, van Rantwijk F, Sheldon RA (2000) Cross-linked enzyme aggregates: a simple and effective method for the immobilization of penicillin acylase. Org Lett 2:1361–1364
Carrio M, Gonzalez-Montalban N, Vera A, Villaverde A, Ventura S (2005) Amyloid-like properties of bacterial inclusion bodies. J Mol Biol 347:1025–1037
Caughey B, Lansbury PT (2003) Protofibrils, pores, fibrils, and neurodegeneration: separating the responsible protein aggregates from the innocent bystanders. Annu Rev Neurosci 26:267–298
Chiba Y, Takei S, Kawamura N, Kawaguchi Y, Sasaki K, Hasegawa-Ishii S, Furukawa A, Hosokawa M, Shimada A (2012) Immunohistochemical localization of aggresomal proteins in glial cytoplasmic inclusions in multiple system atrophy. Neuropathol Appl Neurobiol 38:559–571
Dahl JU, Gray MJ, Jakob U (2015) Protein quality control under oxidative stress conditions. J Mol Biol 427:1549–1563
de Groot NS, Sabate R, Ventura S (2009) Amyloids in bacterial inclusion bodies. Trends Biochem Sci 34:408–416
de Groot NS, Ventura S (2006a) Effect of temperature on protein quality in bacterial inclusion bodies. FEBS Lett 580:6471–6476
de Groot NS, Ventura S (2006b) Protein activity in bacterial inclusion bodies correlates with predicted aggregation rates. J Biotechnol 125:110–113
Dehvari N, Mahmud T, Persson J, Bengtsson T, Graff C, Winblad B, Ronnback A, Behbahani H (2012) Amyloid precursor protein accumulates in aggresomes in response to proteasome inhibitor. Neurochem Int 60:533–542
Dickerson TJ, Reed NN, Janda KD (2002) Soluble polymers as scaffolds for recoverable catalysts and reagents. Chem Rev 102:3325–3344
Dobson CM (2003) Protein folding and misfolding. Nature 426:884–890
Doi H, Mitsui K, Kurosawa M, Machida Y, Kuroiwa Y, Nukina N (2004) Identification of ubiquitin-interacting proteins in purified polyglutamine aggregates. FEBS Lett 571:171–176
Ehrnhoefer DE, Bieschke J, Boeddrich A, Herbst M, Masino L, Lurz R, Engemann S, Pastore A, Wanker EE (2008) EGCG redirects amyloidogenic polypeptides into unstructured, off-pathway oligomers. Nat Struct Mol Biol 15:558–566
Francioso A, Punzi P, Boffi A, Lori C, Martire S, Giordano C, D’Erme M, Mosca L (2015) Beta-sheet interfering molecules acting against beta-amyloid aggregation and fibrillogenesis. Bioorg Med Chem 23:1671–1683
Fratta P, Engel WK, McFerrin J, Davies KJ, Lin SW, Askanas V (2005) Proteasome inhibition and aggresome formation in sporadic inclusion-body myositis and in amyloid-beta precursor protein-overexpressing cultured human muscle fibers. Am J Pathol 167:517–526
Garcia-Fruitos E, Aris A, Villaverde A (2007a) Localization of functional polypeptides in bacterial inclusion bodies. Appl Environ Microbiol 73:289–294
Garcia-Fruitos E, Gonzalez-Montalban N, Morell M, Vera A, Ferraz RM, Aris A, Ventura S, Villaverde A (2005) Aggregation as bacterial inclusion bodies does not imply inactivation of enzymes and fluorescent proteins. Microb Cell Factories 4:27
Garcia-Fruitos E, Martinez-Alonso M, Gonzalez-Montalban N, Valli M, Mattanovich D, Villaverde A (2007b) Divergent genetic control of protein solubility and conformational quality in Escherichia coli. J Mol Biol 374:195–205
Garcia-Fruitos E, Sabate R, de Groot NS, Villaverde A, Ventura S (2011) Biological role of bacterial inclusion bodies: a model for amyloid aggregation. FEBS J 278:2419–2427
Garcia-Mata R, Bebok Z, Sorscher EJ, Sztul ES (1999) Characterization and dynamics of aggresome formation by a cytosolic GFP-chimera. J Cell Biol 146:1239–1254
Garcia-Mata R, Gao YS, Sztul E (2002) Hassles with taking out the garbage: aggravating aggresomes. Traffic 3:388–396
Georgiou G, Valax P (1996) Expression of correctly folded proteins in Escherichia coli. Curr Opin Biotechnol 7:190–197
Gershon ND, Porter KR, Trus BL (1985) The cytoplasmic matrix: its volume and surface area and the diffusion of molecules through it. Proc Natl Acad Sci U S A 82:5030–5034
Ghosh AK, Gemma S, Tang J (2008) Beta-secretase as a therapeutic target for Alzheimer’s disease. Neurotherapeutics 5:399–408
Glabe CG (2006) Common mechanisms of amyloid oligomer pathogenesis in degenerative disease. Neurobiol Aging 27:570–575
Gonzalez-Montalban N, Garcia-Fruitos E, Villaverde A (2007) Recombinant protein solubility—does more mean better? Nat Biotechnol 25:718–720
Gonzalez-Montalban N, Natalello A, Garcia-Fruitos E, Villaverde A, Doglia SM (2008) In situ protein folding and activation in bacterial inclusion bodies. Biotechnol Bioeng 100:797–802
Guerrero-Munoz MJ, Castillo-Carranza DL, Kayed R (2014) Therapeutic approaches against common structural features of toxic oligomers shared by multiple amyloidogenic proteins. Biochem Pharmacol 88:468–478
Hartl FU, Hayer-Hartl M (2002) Molecular chaperones in the cytosol: from nascent chain to folded protein. Science 295:1852–1858
Hideshima T, Bradner JE, Wong J, Chauhan D, Richardson P, Schreiber SL, Anderson KC (2005) Small-molecule inhibition of proteasome and aggresome function induces synergistic antitumor activity in multiple myeloma. Proc Natl Acad Sci U S A 102:8567–8572
Invernizzi G, Papaleo E, Sabate R, Ventura S (2012) Protein aggregation: mechanisms and functional consequences. Int J Biochem Cell Biol 44:1541–1554
Ioannou YA, Bishop DF, Desnick RJ (1992) Overexpression of human alpha-galactosidase A results in its intracellular aggregation, crystallization in lysosomes, and selective secretion. J Cell Biol 119:1137–1150
Johnston JA, Dalton MJ, Gurney ME, Kopito RR (2000) Formation of high molecular weight complexes of mutant Cu, Zn-superoxide dismutase in a mouse model for familial amyotrophic lateral sclerosis. Proc Natl Acad Sci U S A 97:12571–12576
Johnston JA, Illing ME, Kopito RR (2002) Cytoplasmic dynein/dynactin mediates the assembly of aggresomes. Cell Motil Cytoskeleton 53:26–38
Johnston JA, Ward CL, Kopito RR (1998) Aggresomes: a cellular response to misfolded proteins. J Cell Biol 143:1883–1898
Junn E, Lee SS, Suhr UT, Mouradian MM (2002) Parkin accumulation in aggresomes due to proteasome impairment. J Biol Chem 277:47870–47877
Kalmar B, Novoselov S, Gray A, Cheetham ME, Margulis B, Greensmith L (2008) Late stage treatment with arimoclomol delays disease progression and prevents protein aggregation in the SOD1 mouse model of ALS. J Neurochem 107:339–350
Kastelic M, Kalyuzhnyi YV, Hribar-Lee B, Dill KA, Vlachy V (2015) Protein aggregation in salt solutions. Proc Natl Acad Sci U S A 112:6766–6770
Kieran D, Kalmar B, Dick JR, Riddoch-Contreras J, Burnstock G, Greensmith L (2004) Treatment with arimoclomol, a coinducer of heat shock proteins, delays disease progression in ALS mice. Nat Med 10:402–405
Kopito RR (1997) ER quality control: the cytoplasmic connection. Cell 88:427–430
Kopito RR (2000) Aggresomes, inclusion bodies and protein aggregation. Trends Cell Biol 10:524–530
Kopito RR, Sitia R (2000) Aggresomes and Russell bodies. Symptoms of cellular indigestion? EMBO Rep 1:225–231
Lashuel HA, Hartley D, Petre BM, Walz T, Lansbury PT Jr (2002) Neurodegenerative disease: amyloid pores from pathogenic mutations. Nature 418:291
Liu T, Bitan G (2012) Modulating self-assembly of amyloidogenic proteins as a therapeutic approach for neurodegenerative diseases: strategies and mechanisms. ChemMedChem 7:359–374
Luk KC, Mills IP, Trojanowski JQ, Lee VM (2008) Interactions between Hsp70 and the hydrophobic core of alpha-synuclein inhibit fibril assembly. Biochemistry 47:12614–12625
Maji SK, Perrin MH, Sawaya MR, Jessberger S, Vadodaria K, Rissman RA, Singru PS, Nilsson KP, Simon R, Schubert D, Eisenberg D, Rivier J, Sawchenko P, Vale W, Riek R (2009) Functional amyloids as natural storage of peptide hormones in pituitary secretory granules. Science 325:328–332
Maji SK, Schubert D, Rivier C, Lee S, Rivier JE, Riek R (2008) Amyloid as a depot for the formulation of long-acting drugs. PLoS Biol 6:e17
Marston FA (1986) The purification of eukaryotic polypeptides synthesized in Escherichia coli. Biochem J 240:1–12
Martinez-Alonso M, Garcia-Fruitos E, Villaverde A (2008a) Yield, solubility and conformational quality of soluble proteins are not simultaneously favored in recombinant Escherichia coli. Biotechnol Bioeng 101:1353–1358
Martinez-Alonso M, Gonzalez-Montalban N, Garcia-Fruitos E, Villaverde A (2009) Learning about protein solubility from bacterial inclusion bodies. Microb Cell Factories 8:4
Martinez-Alonso M, Gonzalez-Montalban N, Garcia-Fruitos E, Villaverde A (2008b) The functional quality of soluble recombinant polypeptides produced in Escherichia coli is defined by a wide conformational spectrum. Appl Environ Microbiol 74:7431–7433
Meng F, Abedini A, Plesner A, Verchere CB, Raleigh DP (2010) The flavanol (−)-epigallocatechin 3-gallate inhibits amyloid formation by islet amyloid polypeptide, disaggregates amyloid fibrils, and protects cultured cells against IAPP-induced toxicity. Biochemistry 49:8127–8133
Mitraki A (2010) Protein aggregation from inclusion bodies to amyloid and biomaterials. Adv Protein Chem Struct Biol 79:89–125
Morell M, Bravo R, Espargaro A, Sisquella X, Aviles FX, Fernandez-Busquets X, Ventura S (2008) Inclusion bodies: specificity in their aggregation process and amyloid-like structure. Biochim Biophys Acta 1783:1815–1825
Mukherjee A, Morales-Scheihing D, Butler PC, Soto C (2015) Type 2 diabetes as a protein misfolding disease. Trends Mol Med 21:439–449
Naeem A, Fazili NA (2011) Defective protein folding and aggregation as the basis of neurodegenerative diseases: the darker aspect of proteins. Cell Biochem Biophys 61:237–250
Nahalka J (2008) Physiological aggregation of maltodextrin phosphorylase from Pyrococcus furiosus and its application in a process of batch starch degradation to alpha-D-glucose-1-phosphate. J Ind Microbiol Biotechnol 35:219–223
Nahalka J, Dib I, Nidetzky B (2008a) Encapsulation of Trigonopsis variabilis D-amino acid oxidase and fast comparison of the operational stabilities of free and immobilized preparations of the enzyme. Biotechnol Bioeng 99:251–260
Nahalka J, Nidetzky B (2007) Fusion to a pull-down domain: a novel approach of producing Trigonopsis variabilis D-amino acid oxidase as insoluble enzyme aggregates. Biotechnol Bioeng 97:454–461
Nahalka J, Patoprsty V (2009) Enzymatic synthesis of sialylation substrates powered by a novel polyphosphate kinase (PPK3). Org Biomol Chem 7:1778–1780
Nahalka J, Vikartovska A, Hrabarova E (2008b) A crosslinked inclusion body process for sialic acid synthesis. J Biotechnol 134:146–153
Nawrocki ST, Carew JS, Pino MS, Highshaw RA, Andtbacka RH, Dunner K Jr, Pal A, Bornmann WG, Chiao PJ, Huang P, Xiong H, Abbruzzese JL, McConkey DJ (2006) Aggresome disruption: a novel strategy to enhance bortezomib-induced apoptosis in pancreatic cancer cells. Cancer Res 66:3773–3781
Necula M, Breydo L, Milton S, Kayed R, van der Veer WE, Tone P, Glabe CG (2007) Methylene blue inhibits amyloid Abeta oligomerization by promoting fibrillization. Biochemistry 46:8850–8860
Olzmann JA, Li L, Chin LS (2008) Aggresome formation and neurodegenerative diseases: therapeutic implications. Curr Med Chem 15:47–60
Ong DS, Kelly JW (2011) Chemical and/or biological therapeutic strategies to ameliorate protein misfolding diseases. Curr Opin Cell Biol 23:231–238
Pastor MT, Kummerer N, Schubert V, Esteras-Chopo A, Dotti CG, de la Lopez PM, Serrano L (2008) Amyloid toxicity is independent of polypeptide sequence, length and chirality. J Mol Biol 375:695–707
Pastore A, Temussi P (2012) Protein aggregation and misfolding: good or evil? J Phys Condens Matter 24:244101
Peternel S, Grdadolnik J, Gaberc-Porekar V, Komel R (2008) Engineering inclusion bodies for non denaturing extraction of functional proteins. Microb Cell Factories 7:34
Polizzi KM, Bommarius AS, Broering JM, Chaparro-Riggers JF (2007) Stability of biocatalysts. Curr Opin Chem Biol 11:220–225
Rodriguez-Carmona E, Mendoza R, Ruiz-Canovas E, Ferrer-Miralles N, Abasolo I, Schwartz S Jr, Villaverde A, Corchero JL (2015) A novel bio-functional material based on mammalian cell aggresomes. Appl Microbiol Biotechnol 99:7079–7088
Roessl U, Nahalka J, Nidetzky B (2010) Carrier-free immobilized enzymes for biocatalysis. Biotechnol Lett 32:341–350
Sah DW, Aronin N (2011) Oligonucleotide therapeutic approaches for Huntington disease. J Clin Invest 121:500–507
Schroder M, Kaufman RJ (2005) The mammalian unfolded protein response. Annu Rev Biochem 74:739–789
Seras-Franzoso J, Peebo K, Corchero JL, Tsimbouri PM, Unzueta U, Rinas U, Dalby MJ, Vazquez E, Garcia-Fruitos E, Villaverde A. (2013a). A nanostructured bacterial bioscaffold for the sustained bottom-up delivery of protein drugs. Nanomedicine (Lond)
Seras-Franzoso J, Peebo K, Garcia-Fruitos E, Vazquez E, Rinas U, Villaverde A (2014) Improving protein delivery of fibroblast growth factor-2 from bacterial inclusion bodies used as cell culture substrates. Acta Biomater 10:1354–1359
Seras-Franzoso J, Steurer C, Roldan M, Vendrell M, Vidaurre-Agut C, Tarruella A, Saldana L, Vilaboa N, Parera M, Elizondo E, Ratera I, Ventosa N, Veciana J, Campillo-Fernandez AJ, Garcia-Fruitos E, Vazquez E, Villaverde A (2013b) Functionalization of 3D scaffolds with protein-releasing biomaterials for intracellular delivery. J Control Release 171:63–72
Sgarbossa A (2012) Natural biomolecules and protein aggregation: emerging strategies against amyloidogenesis. Int J Mol Sci 13:17121–17137
Sheldon RA (2007) Cross-linked enzyme aggregates (CLEAs): stable and recyclable biocatalysts. Biochem Soc Trans 35:1583–1587
Shimohata T, Sato A, Burke JR, Strittmatter WJ, Tsuji S, Onodera O (2002) Expanded polyglutamine stretches form an “aggresome”. Neurosci Lett 323:215–218
Silva JL, De Moura Gallo CV, Costa DC, Rangel LP (2014) Prion-like aggregation of mutant p53 in cancer. Trends Biochem Sci 39:260–267
Silva JL, Rangel LP, Costa DC, Cordeiro Y, De Moura Gallo CV. (2013). Expanding the prion concept to cancer biology: dominant-negative effect of aggregates of mutant p53 tumour suppressor. Biosci Rep 33.
Silva JL, Vieira TC, Gomes MP, Bom AP, Lima LM, Freitas MS, Ishimaru D, Cordeiro Y, Foguel D (2010) Ligand binding and hydration in protein misfolding: insights from studies of prion and p53 tumor suppressor proteins. Acc Chem Res 43:271–279
Sirangelo I, Irace G (2010) Inhibition of aggregate formation as therapeutic target in protein misfolding diseases: effect of tetracycline and trehalose. Expert Opin Ther Targets 14:1311–1321
Suhr ST, Senut MC, Whitelegge JP, Faull KF, Cuizon DB, Gage FH (2001) Identities of sequestered proteins in aggregates from cells with induced polyglutamine expression. J Cell Biol 153:283–294
Tischer W, Kasche V (1999) Immobilized enzymes: crystals or carriers? Trends Biotechnol 17:326–335
Tokatlidis K, Dhurjati P, Millet J, Beguin P, Aubert JP (1991) High activity of inclusion bodies formed in Escherichia coli overproducing Clostridium thermocellum endoglucanase D. FEBS Lett 282:205–208
Tyedmers J, Mogk A, Bukau B (2010) Cellular strategies for controlling protein aggregation. Nat Rev Mol Cell Biol 11:777–788
Upadhyay AK, Murmu A, Singh A, Panda AK (2012) Kinetics of inclusion body formation and its correlation with the characteristics of protein aggregates in Escherichia coli. PLoS One 7:e33951
Vazquez E, Corchero JL, Burgueno JF, Seras-Franzoso J, Kosoy A, Bosser R, Mendoza R, Martinez-Lainez JM, Rinas U, Fernandez E, Ruiz-Avila L, Garcia-Fruitos E, Villaverde A (2012) Functional inclusion bodies produced in bacteria as naturally occurring nanopills for advanced cell therapies. Adv Mater 24:1742–1747
Vidair CA, Huang RN, Doxsey SJ (1996) Heat shock causes protein aggregation and reduced protein solubility at the centrosome and other cytoplasmic locations. Int J Hyperth 12:681–695
Vousden KH, Lane DP (2007) p53 in health and disease. Nat Rev Mol Cell Biol 8:275–283
Walker LC, Diamond MI, Duff KE, Hyman BT (2013) Mechanisms of protein seeding in neurodegenerative diseases. JAMA Neurol 70:304–310
Walsh DM, Selkoe DJ (2004) Oligomers on the brain: the emerging role of soluble protein aggregates in neurodegeneration. Protein Pept Lett 11:213–228
Walsh DM, Selkoe DJ (2007) A beta oligomers—a decade of discovery. J Neurochem 101:1172–1184
Wang H, Raleigh DP (2014) General amyloid inhibitors? A critical examination of the inhibition of IAPP amyloid formation by inositol stereoisomers. PLoS One 9:e104023
Wang Y, Meriin AB, Costello CE, Sherman MY (2007) Characterization of proteins associated with polyglutamine aggregates: a novel approach towards isolation of aggregates from protein conformation disorders. Prion 1:128–135
Wigley WC, Fabunmi RP, Lee MG, Marino CR, Muallem S, DeMartino GN, Thomas PJ (1999) Dynamic association of proteasomal machinery with the centrosome. J Cell Biol 145:481–490
Worrall DM, Goss NH (1989) The formation of biologically active beta-galactosidase inclusion bodies in Escherichia coli. Aust J Biotechnol 3:28–32
Yamamoto A, Lucas JJ, Hen R (2000) Reversal of neuropathology and motor dysfunction in a conditional model of Huntington’s disease. Cell 101:57–66
Yang F, Lim GP, Begum AN, Ubeda OJ, Simmons MR, Ambegaokar SS, Chen PP, Kayed R, Glabe CG, Frautschy SA, Cole GM (2005) Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J Biol Chem 280:5892–5901
Zerovnik E (2002) Amyloid-fibril formation. Proposed mechanisms and relevance to conformational disease. Eur J Biochem 269:3362–3371
Zhang X, Qian SB (2011) Chaperone-mediated hierarchical control in targeting misfolded proteins to aggresomes. Mol Biol Cell 22:3277–3288
Zu T, Duvick LA, Kaytor MD, Berlinger MS, Zoghbi HY, Clark HB, Orr HT (2004) Recovery from polyglutamine-induced neurodegeneration in conditional SCA1 transgenic mice. J Neurosci 24:8853–8861
Acknowledgments
The author acknowledges the financial support received for funding our research on protein-based nanotherapeutics and biomaterials from Agència de Gestió d’Ajuts Universitaris i de Recerca (AGAUR, 2014SGR-132), FIS (PS09-00165 and PI12/00327), Fundació La Marató de TV3 (TV32009-101235, TV32013-132031, and TV32013-133930), INIA (RTA2012-00028-C02-02), MINECO (BFU2010-17450, ACI2009-0919, IT2009-0021, BIO2013-41019-P, LIPOCELL TT02, and TERARMET RTC-2014-2207-1), and from the Centro de Investigación Biomédica en Red (CIBER) de Bioingeniería, Biomateriales y Nanomedicina (NANOLYSO, NANOPROTHER, and PENTRI projects). CIBER de Bioingeniería, Biomateriales y Nanomedicina (CIBER-BBN) is an initiative funded by the VI National R&D&i Plan 2008–2011, Iniciativa Ingenio 2010, Consolider Program, CIBER Actions, and financed by the Instituto de Salud Carlos III with assistance from the European Regional Development Fund. The author is also indebted to the Protein Production Platform (CIBER-BBN/UAB) for helpful technical assistance during recombinant protein production and purification (http://www.ciber-bbn.es/en/programas/89-plataforma-de-produccion-de-proteinas).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares no commercial or financial conflict of interest.
Rights and permissions
About this article
Cite this article
Corchero, J.L. Eukaryotic aggresomes: from a model of conformational diseases to an emerging type of immobilized biocatalyzers. Appl Microbiol Biotechnol 100, 559–569 (2016). https://doi.org/10.1007/s00253-015-7107-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-7107-y