Abstract
Objective
To determine which hydration (saline, saline + mannitol, or saline + furosemide) is associated with least cisplatin nephrotoxicity.
Methods
We randomized 49 women who received cisplatin (75 mg/m2 every 3 weeks) into one of the three hydration arms. The 24-h creatinine clearance was measured before and on day 6 after cisplatin infusion. The patients of each arm received 2 l of saline hydration. In the saline + furosemide arm, 40 mg of furosemide was given after hydration. In the saline + mannitol arm, 50 g of mannitol was mixed with the cisplatin.
Results
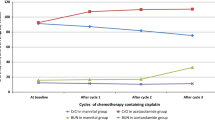
For the first cycle of chemotherapy, 15 women were randomized to saline, 17 to saline + furosemide, and 17 to saline + mannitol. For each group, the creatinine clearances before cisplatin infusion were (means±SD, milliliters per minute) 84.5±26.8, 82.5±24.0 and 87.4±25.6, and after cisplatin infusion were 79.1±31.9, 68.7±21.5, and 56.4±22.9, respectively. The decreases in creatinine clearance were similar between the saline group and the saline + furosemide group (P=0.66), but different between the saline + mannitol group and the saline group (P=0.02) or the saline + furosemide group (P=0.02). As each woman received multiple courses of cisplatin, 15 who received saline contributed 41 paired datasets, 17 who received saline + furosemide contributed 49 paired datasets, and 17 who received saline + mannitol contributed 36 paired datasets showed similar patterns.
Conclusions
Hydration with saline or saline + furosemide appears to be associated with less cisplatin nephrotoxicity than saline + mannitol.


Similar content being viewed by others
References
Whitney CW, Sause W, Bundy BN, Malfetano JH, Hannigan EV, Fowler WC Jr, Clarke-Pearson DL, Liao SY (1999) Randomized comparison of fluorouracil plus cisplatin versus hydroxyurea as an adjunct to radiation therapy in stage IIB-IVA carcinoma of the cervix with negative para-aortic lymph nodes: a Gynecologic Oncology Group and Southwest Oncology Group study. J Clin Oncol 17:1339–1348
Morris M, Eifel PJ, Lu J, Grigsby PW, Levenback C, Stevens RE, Rotman M, Gershenson DM, Mutch DG (1999) Pelvic radiation with concurrent chemotherapy compared with pelvic and para-aortic radiation for high-risk cervical cancer. N Engl J Med 340:1137–1143
Rose PG, Bundy BN, Watkins EB, Thigpen JT, Deppe G, Maiman MA, Clarke-Pearson DL, Insalaco S (1999) Concurrent cisplatin-based radiotherapy and chemotherapy for locally advanced cervical cancer. N Engl J Med 340:1144–1153
Keys HM, Bundy BN, Stehman FB, Muderspach LI, Chafe WE, Suggs CL 3rd, Walker JL, Gersell D (1999) Cisplatin, radiation, and adjuvant hysterectomy compared with radiation and adjuvant hysterectomy for bulky stage IB cervical carcinoma. N Engl J Med 340:1154–1161
Peters WA III, Liu PY, Barrett RJ, Stock RJ, Monk BJ, Berek JS, Souhami L, Grigsby P, Gordon W Jr, Alberts DS (2000) Concurrent chemotherapy and pelvic radiation therapy as adjunctive therapy after radical surgery in high risk early stage carcinoma of the cervix. J Clin Oncol 18:1606–1613
Ozols RF, Young RC (1985) High-dose cisplatin therapy in ovarian cancer. Semin Oncol 12:21–30
Cornelison TL, Reed E (1993) Nephrotoxicity and hydration management for cisplatin, carboplatin, and ormaplatin (review). Gynecol Oncol 50:147–158
Daugaard G, Abildgaard U, Larsen S (1987) Functional and histopathological changes in dog kidneys after administration of cisplatin. Renal Physiol 10:54–64
Clayton DA, Doda JN, Fiedberg ED (1974) The absence of a pyrimidine dimer repair mechanism in mammalian mitochondria. Proc Natl Acad Sci U S A 71:2777–2781
Gordon JA, Gattone VH (1986) Mitochondrial alteration in cisplatin-induced acute renal failure. Am J Physiol 250:F991–998
Tisher CA (1981) Anatomy of the kidney. In: Brenner Rectors (eds) The kidney. Saunders, Philadelphia, pp 28–35
Hayes DM, Cvitkovic E, Goldberg RB, Scheiner E, Helson L, Krakoff IH (1977) High dose cis-platinum diammine chloride. Cancer 39:1372–1381
Higby DJ, Wallace HJ Jr, Albert DJ, Holland JF (1974) Diaminodichloroplatinum: a phase-I study showing responses in testicular and other tumors. Cancer 33:1219–1225
Al-Sarraf M, Fletcher W, Oishi N (1982) Cisplatin hydration with and without mannitol diuresis in refractory disseminated malignant melanoma: a Southwest Oncology Group study. Cancer Treat Rep 66:31–35
Cvitkovic E, Spaulding J, Bethune V, Martin J, Whitmore WF (1977) Improvement of cis-dichlorodiammineplatinum (NSC 119875): therapeutic index in an animal model. Cancer 39:1357–1361
Ostrow O, Egorin MJ, Hahn D (1981) High dose cisplatin therapy using mannitol versus furosemide diuresis: comparative pharmacokinetics and toxicity. Cancer Treat Rep 65:73–78
Daugaard G, Abildgaard U, Holstein-Rathlou NH, Bruunshuus I, Bucher D, Leyssai PP (1988) Renal tubular function in patients treated with high-dose cisplatin. Clin Pharmacol Ther 44:164–172
Sobrero A, Guglemi A, Aschele C, Rosso R (1990) Current strategies to reduce cisplatin toxicity. J Chemother 2:3–7
Chary KK, Higby DJ, Henderson ES (1977) Phase I study of high-dose cis-dichlorodiammineplatinum(II) with forced diuresis. Cancer Treat Rep 61:367–370
Lehane D, Winston A, Gray R, Daskal Y (1979) The effect of diuretic pretreatment on clinical, morphological, and ultrastructural cis-platinum induced nephrotoxicity. Int J Radiat Oncol Biol Phys 5:1393–1399
Gehr M, Chopra S, Kaufman J, Chase D, Fjlamenbaum W (1981) The effect of furosemide on cis-diamminedichloroplatinum-induced acute renal failure. Kidney Int 19:201–204
Ward JM, Grabin ME, LeRoy AF, Young DM (1977) Modification of the renal toxicity of cis-diamminedichloroplatinum (II) with furosemide in male F344 rats. Cancer Treat Rep 61:375–379
Fisher DS, Knobf MT, Durivage HJ (1989) The cancer chemotherapy handbook, 4th edn. Mosby Yearbook, St. Louis
Anad AJ, Bashey B (1993) Newer insights into cisplatin nephrotoxicity. Ann Pharmacother 27:1519–1525
Alberts DS, Green S, Hannigan EV, O'Toole R, Stock-Novack D, Anderson P, Surwit EA, Malvlya VK, Nahhas WA, Jolles CJ (1992) Improved therapeutic index of carboplatin plus cyclophosphamide versus cisplatin plus cyclophosphamide: final report by the Southwest Oncology Group of a phase III randomized trial in stages III & IV ovarian cancer. J Clin Oncol 10:706–717
Kemp G, Rose P, Lurain J, Berman M, Manetta A, Roullet B, Homesley H, Belpomme D, Glick J (1996) Amifostine pretreatment for protection against cyclophosphamide-induced and cisplatin-induced toxicities: results of a randomized control trial in patients with advanced ovarian cancer. J Clin Oncol 14:2101–2112
Levey AS, Perrone RD, Madias NE (1988) Serum creatinine and renal function. Annu Rev Med 39:465–490
Daugaard G, Rossing N, Rorth M (1988) Effects of high dose cisplatin on glomerular function in the human kidney. Cancer Chemother Pharmacol 21:163–167
Acknowledgements
We wish to thank Robert Safirstein MD for his assistance in designing the study and James J. Grady DrPH for statistical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Santoso, J.T., Lucci, J.A., Coleman, R.L. et al. Saline, mannitol, and furosemide hydration in acute cisplatin nephrotoxicity: a randomized trial. Cancer Chemother Pharmacol 52, 13–18 (2003). https://doi.org/10.1007/s00280-003-0620-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-003-0620-1