Abstract
Introduction
An experimental study was conducted to investigate the effects of erythropoietin on the acute phase of esophageal burn damage induced by sodium hydroxide.
Materials and methods
A standard esophageal alkaline burn was produced by the application of 10% sodium hydroxide to the distal esophagus in an in vivo rat model. Fifty-six female rats were allocated into three groups: Group BC (baseline control, n = 8) rats were uninjured and untreated, Group PC (positive control, n = 24) rats were injured but untreated and Group EPO (erythropoietin-treated, n = 24) rats were injured and given subcutaneous erythropoietin (1,000 IU/kg per day), 15 min, 24, and 48 h after administration of the NaOH solution. Six animals from Group PC and six from Group EPO were killed at 4, 24, 48, and 72 h after application of NaOH to the esophagus. All of animals in Group BC were killed 4 h after exposure to 0.9% NaCl. Oxidative damage was assessed by measuring levels of malondialdehyde (MDA) and nitric oxide (NO), and activities of superoxide dismutase (SOD) and catalase (CAT) in homogenized samples of esophageal tissue. Histologic damage to esophageal tissue was scored by a single pathologist blind to groups.
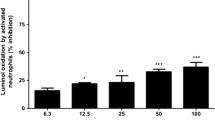
Results
MDA levels in the BC and EPO groups were significantly lower than those in the PC group (p < 0.05). CAT and SOD activities, and NO levels in the BC and EPO groups were significantly higher than in the PC group (p < 0.05). Esophageal tissue damage measured at 4, 24, 48, and 72 h after NaOH application was significantly less in the EPO group than in the PC group (p < 0.05).
Conclusions
When administered early after an esophageal burn induced by 10% sodium hydroxide in this rat model, erythropoietin significantly attenuated oxidative damage, as measured by biochemical markers and histologic scoring.



Similar content being viewed by others
References
Tanyel FC, Buyukpamukcu N, Hicsonmez A (1988) The predictability of esophageal burns after caustic ingestion in children. Turk J Pediatr 30:109–112
Turan C, Ozkan U, Ozokutan BH, Ozdemir M, Okur H, Küçükaydin M (2000) Corrosive injuries of the esophagus in newborns. Pediatr Surg Int 16:483–484
Karnak I, Tanyel FC, Buyukpamukcu N, Hiçsönmez A (1999) Combined use of steroid, antibiotics and early bougienage against stricture formation following caustic esophageal burns. J Cardiovasc Surg (Torino) 40:307–310
Keskin E, Okur H, Koltuksuz U, Zorludemir U, Olcay I (1991) The effect of steroid treatment on corrosive oesophageal burns in children. Eur J Pediatr Surg 1:335–338
Mutaf O, Ozok G, Avanoglu A (1995) Oesophagoplasty in the treatment of caustic oesophageal strictures in children. Br J Surg 82:644–646
Panieri E, Rode H, Millar AJ, Cywes S (1998) Oesophageal replacement in the management of corrosive strictures: when is surgery indicated? Pediatr Surg Int 13:336–340
Gupta DK, Kataria R, Bajpai M (1997) Gastric transposition for esophageal replacement in children-an Indian experience. Eur J Pediatr Surg 7:143–146
Demirbilek S, Bernay F, Rizalar R, Bariş S, Gürses N (1994) Effects of estradiol and progesterone on the synthesis of collagen in corrosive esophageal burns in rats. J Pediatr Surg 29:1426–1428
Koltuksuz U, Mutus HM, Kutlu R, Ozyurt H, Cetin S, Karaman A (2001) Effect of caffeic acid phenethyl ester andepidermal growth factor on the development of causticesophageal stricture in rats. J Pediatr Surg 36:1504–1509
Türkyilmaz Z, Sönmez K, Demirtola A, Karabulut R, Poyraz A, Gülen S, Dinçer S, Başaklar AC, Kale N (2005) Mitomycin C prevents strictures in caustic esophageal burns in rats. J Surg Res 123:182–187
Bingöl-Kologlu M, Tanyel FC, Müftüoğlu S, Renda N, Cakar N, Büyükpamukçu N, Hiçsönmez A (1999) The preventive effect of heparin on stricture formation after caustic esophageal burns. J Pediatr Surg 34:291–294
Topaloglu B, Bicakci U, Tander B, Ariturk E, Kilicoglu-Aydin B, Aydin O, Rizalar R, Ayyildiz SH, Bernay F (2008) Biochemical and histopathologic effects of omeprazole and vitamin E in rats with corrosive esophageal burns. Pediatr Surg Int 24:555–560
Günel E, Cağlayan F, Cağlayan O, Canbilen A, Tosun M (2002) Effect of antioxidant therapy on collagen synthesis in corrosive esophageal burns. Pediatr Surg Int 18:24–27
Yagmurlu A, Aksu B, Bingol-Kologlu M, Renda N, Altinok G, Fitoz S, Gokcora IH, Dindar H (2004) A novel approach for preventing esophageal stricture formation: sphingosylphosphorylcholine-enhanced tissue remodeling. Pediatr Surg Int 20:778–782
Li C, Jackson RM (2002) Reactive species mechanisms of cellular hypoxi-reoxygenation injury. Am J Physiol Cell Physiol 282:C227–C241
Gunel E, Caglayan F, Caglayan O, Akıllıoglu I (1999) Reactive oxygen radical levels in caustic esophageal burns. J Pediatr Surg 34:405–407
Soteras F, Lanas A, Fiteni I, Royo Y, Jimenez P, Iñarrea P, Ortego J, Esteva F (2000) Nitric oxide and superoxide anion in low-grade esophagitis induced by acid and pepsin in rabbits. Dig Dis Sci 45:1802–1809
Ozel SK, Dagli TE, Yuksel M, Kiyan G, Kotiloglu E (2004) The roles of free oxygen radicals, nitric oxide, and endothelin in caustic injury of rat esophagus. J Pediatr Surg 39:1381–1385
Mori S, Sawada T, Okada T, Kubota K (2008) Erythropoietin and its derivative protect the intestine from severe ischemia/reperfusion injury in the rat. Surgery 143:556–565
Spandou E, Tsouchnikas I, Karkavelas G, Dounousi E, Simeonidou C, Guiba-Tziampiri O, Tsakiris D (2006) Erythropoietin attenuates renal injury in experimental acute renal failure ischemic/reperfusion model. Nephrol Dial Transplant 21:330–336
Sepodes B, Maio R, Pinto R, Sharples E, Oliveira P, McDonald M, Yaqoob M, Thiemermann C, Mota-Filipe H (2006) Recombinant human erythropoietin protects the liver from hepatic ischemia–reperfusion injury in the rat. Transplant Int 19:919–926
Minamino T, Kitakaze M (2006) New therapeutic application of erythropoietin against ischemic heart diseases. J Pharmacol Sci 101:179–181
Ergur BU, Kiray M, Pekcetin C, Bagriyanik HA, Erbil G (2008) Protective effect of erythropoietin pretreatment in testicular ischemia–reperfusion injury in rats. J Pediatr Surg 43:722–728
Santhanam AV, Katusic ZS (2006) Erythropoietin and cerebral vascular protection: role of nitric oxide. Acta Pharmacol Sin 27:1389–1394
Jelkmann W (2007) Erythropoietin after a century of research: younger than ever. Eur J Haematol 78:183–205
Brines M, Cerami A (2006) Discovering erythropoietin’s extrahematopoietic functions: biology and clinical promise. Kidney Int 70:246–250
Liu AJ, Richardson MA (1985) Effects of N-acetylcysteine of experimentally induced esophageal lye injury. Ann Otol Rhinol Laryngol 94:477–482
Gehanno P, Guedon C (1981) Inhibition of experimental esophageal lye strictures by penicillamine. Arch Otolaryngol 107:145–147
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Lowry OH, Rosebrough NJ, Farr A (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Fridovich I (1974) Superoxide dismutase. Adv Enzymol 41:35–97
Beutler E (1975) Red cell metabolism, 2nd edn. Grune and Stratton Company, New York, pp 261–265
Moshage H, Kok B, Huizenga JR, Jansen PL (1995) Nitrite and nitrate determinations in plasma: a critical evaluation. Clin Chem 41:892–896
Cortas NK, Wakid NW (1990) Determination of inorganic nitratein serum and urine by akinetic cadmium reduction method. Clin Chem 36:1440–1443
Rothstein FC (1986) Caustic injuries to the oesophagus in children. Pediatr Clin North Am 33:665–674
Rui T, Feng Q, Lei M, Peng T, Zhang J, Xu M, Abel ED, Xenocostas A, Kvietys PR (2005) Erythropoietin prevents the acute myocardial inflammatory response induced by ischemia/reperfusion via induction of AP-1. Cardiovasc Res 65:719–727
Salmonson T, Danıelson BG, Wıkstrom B (1990) Pharmacokinetics of recombinant human erythropoietin after intravenous and subcutaneous administration to healthy subjects. Br J Clin Pharmacol 29:709–713
Singh AK, Szczech L, Tang KL et al (2006) Correction of anemia with epoetin alfa in chronic kidney disease. N Engl J Med 355:2085–2098
Lipsic E, Westenbrink BD, van der Meer P et al (2008) Low-dose erythropoietin improves cardiac function in experimental heart failure without increasing haematocrit. Eur J Heart Fail 10:22–29
Kumral A, Gonenc S, Acikgoz O, Sonmez A, Genc K, Yilmaz O, Gokmen N, Duman N, Ozkan H (2005) Erythropoietin increases glutathione peroxidase enzyme activity and decreases lipid peroxidation levels in hypoxic-ischemic brain injury in neonatal rats. Biol Neonate 87:15–18
Nagai A, Nakagawa E, Choi HB, Hatori K, Kobayashi S, Kim SU (2001) Erythropoietin and erythropoietin receptors in human CNS neurons, astrocytes, microglia, and oligodendrocytes grown in culture. J Neuropathol Exp Neurol 60:386–392
van der Vliet A, Eiserich JP, Halliwell B, Cross CE (1997) Formation of reactive nitrogen species during peroxidase-catalyzed oxidation of nitrite. J Biol Chem 272:7617–7625
Ferdinandy P (2006) Peroxynitrite: just an oxidative/nitrosative stressor or a physiological regulator as well? Br J Pharmacol 148:1–3
Ortega Mateo A, Amaya Aleixandre de Artinano (2000) Nitric oxide reactivity and mechanisms involved in its biological effects. Pharmacol Res 42:421–427
Makay O, Yukselen V, Vardar E, Yenisey C, Bicakci T, Ersin S, Ozutemiz O (2007) Role of allopurinol on oxidative stress in caustic burn: cure for stricture? Pediatr Surg Int 23:1105–1112
Marklund SL (1984) Extracellular superoxide dismutase and other superoxide dismutase isoenzymes in tissues from nine mammalian species. Biochem J 222:649–655
Beleslin-Cokic BB, Cokic VP, Yu X (2004) Erythropoietin and hypoxia stimulate erythropoietin receptor and nitric oxide production by endothelial cells. Blood 104:2073–2080
Basaran UN, Eskiocak S, Altaner S, Ture M, Yapar SB (2005) Inhibition of iNOS with S-methylisothiourea was impaired in wound healing in caustic esophageal burn. Int J Pediatr Otorhinolaryngol 69:471–477
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bakan, V., Garipardıc, M., Okumuş, M. et al. The protective effect of erythropoietin on the acute phase of corrosive esophageal burns in a rat model. Pediatr Surg Int 26, 195–201 (2010). https://doi.org/10.1007/s00383-009-2480-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-009-2480-1