Abstract
Primary progressive aphasia (PPA) may be the onset of several neurodegenerative diseases. This study evaluates a cohort of patients with PPA to assess their progression to different clinical syndromes, associated factors that modulate this progression, and patterns of cerebral metabolism linked to different clinical evolutionary forms. Thirty-five patients meeting PPA criteria underwent a clinical and neuroimaging 18F-Fluorodeoxyglucose PET evaluation. Survival analysis was performed using time from clinical onset to the development of a non-language symptom or deficit (PPA-plus). Cerebral metabolism was analyzed using Statistical Parametric Mapping. Patients classified into three PPA variants evolved to atypical parkinsonism, behavioral disorder and motor neuron disease in the agrammatic variant; to behavioral disorder in the semantic; and to memory impairment in the logopenic. Median time from the onset of symptoms to PPA-plus was 36 months (31–40, 95 % confidence interval). Right laterality, and years of education were associated to a lower risk of progression, while logopenic variant to a higher risk. Different regions of hypometabolism were identified in agrammatic PPA with parkinsonism, motor neuron disease and logopenic PPA-plus. Clinical course of PPA differs according to each variant. Left anterior temporal and frontal medial hypometabolism in agrammatic variant is linked to motor neuron disease and atypical parkinsonism, respectively. PPA variant, laterality and education may be associated to the risk of progression. These results suggest the possibility that clinical and imaging data could help to predict the clinical course of PPA.



Similar content being viewed by others
References
Mesulam MM (2013) Primary progressive aphasia and the language network: the 2013 H.Houston Merrit Lecture. Neurology 81:456–462
Rogalski EJ, Mesulam MM (2009) Clinical trajectories and biological features of primary progressive aphasia. Curr Alzheimer Res 6:331–336
Dickerson BC (2011) Quantitating severity and progression in primary progressive aphasia. J Mol Neurosci 45:618–628
Matías-Guiu JA, García-Ramos R (2013) Primary progressive aphasia: from syndrome to disease. Neurologia 28:366–374
Rogalski E, Cobia D, Martersteck A et al (2014) Asymmetry of cortical decline in subtypes of primary progressive aphasia. Neurology 83:1184–1191
Heim S, Pieperhoff P, Grande M et al (2014) Longitudinal changes in brains of patients with fluent primary progressive aphasia. Brain Lang 131:11–19
Rohrer JD, Caso F, Mahoney C et al (2013) Patterns of longitudinal brain atrophy in the logopenic variant of primary progressive aphasia. Brain Lang 127:121–126
Leyton CE, Hsieh S, Mioshi E, Hodges JR (2013) Cognitive decline in logopenic aphasia: more than losing words. Neurology 80:897–903
Hsieh S, Hodges JR, Leyton CE et al (2012) Longitudinal changes in primary progressive aphasias: differences in cognitive and dementia staging measures. Dement Geriatr Cogn Disord 34:135–141
Rogalski E, Cobia D, Harrison TM et al (2011) Progression of language decline and cortical atrophy in subtypes of primary progressive aphasia. Neurology 76:1804–1810
Lam BY, Halliday GM, Irish M et al (2014) Longitudinal white matter changes in frontotemporal dementia subtypes. Hum Brain Mapp 35:3547–3557
Wahlund LO, Barkoff F, Fazekas F et al (2001) A new rating scale for age-related white matter changes applicable to MRI and CT. Stroke 32:1318–1322
Gorno-Tempini ML, Hillis AE, Weintraub S et al (2011) Classification of primary progressive aphasia and its variants. Neurology 76:1006–1014
Lezak MD, Howieson DB, Bigler ED et al (2012) Neuropsychological assessment, 5th edn. Oxford University Press, Oxford
Pfeffer RI, Kurosaki TT, Harrah CH, Chance JM, Filos S (1982) Measurement of functional activities in older adults in the community. J Gerontol 3:323–329
Teunisse S, Derix MM, Crevel H (1991) Assessing the severity of dementia. Patient and caregiver. Arch Neurol 48:274–277
Quintana M, Peña-Casanova J, Sánchez-Benavides G, Neuronorma Study Team et al (2011) Spanish multicenter normative studies (Neuronorma project): norms for the abbreviated Barcelona test. Arch Clin Neuropsychol 26:144–157
Sapolsky D, Bakkour A, Negreira A et al (2010) Cortical neuroanatomic correlated of symptom severity in primary progressive aphasia. Neurology 75:358–366
Peña-Casanova J, Blesa R, Aguilar M, for the NEURONORMA Study Team et al (2009) Spanish Multicenter Normative Studies (NEURONORMA Project): methods and sample characteristics. Arch Clin Neuropsychol 24:307–319
Ashburner J, Friston KJ (2000) Voxel-based morphometry: the methods. Neuroimage 11:805–821
Tzourio-Mazoyer N, Landeau B, Papathanassiou D et al (2002) Automated anatomical labeling of activations in SPM using a macroscopic parcellation of the MNI MRI single-subject brain. Neuroimage 15:273–289
Lancaster JL, Woldorff MG, Parsons LM et al (2000) Automated Tailarach atlas labels for functional brain mapping. Hum Brain Mapp 10:120–131
Lacadie CM, Fulbright RK, Rajeevan N et al (2008) More accurate Talairach coordinates for neuroimaging using non-linear registration. Neuroimage 42:717–725
Kertesz A, Davidson W, McCabe P et al (2003) Primary progressive aphasia: diagnosis, varieties, evolution. J Int Neuropsychol Soc 9:710–719
Le Rhun E, Richard F, Pasquier F (2005) Natural history of primary progressive aphasia. Neurology 65:887–891
Rohrer JD, Rossor MN, Warren JD (2012) Alzheimer’s pathology in primary progressive aphasia. Neurobiol Aging 33:744–752
Hodges JR, Mitchell J, Dawson K et al (2010) Semantic dementia: demography, familial factors and survival in a consecutive series of 100 cases. Brain 133:300–306
Perneczky R, Diehl-Schmid J, Pohl C et al (2007) Non-fluent progressive aphasia: cerebral metabolic patterns and brain reserve. Brain Res 1133:178–185
Khedr EM, Hamed E, Said A et al (2002) Handedness and language cerebral lateralization. Eur J Appl Physiol 87:469–473
Josephs KA, Duffy JR, Strand EA et al (2012) Characterizing a neurodegenerative syndrome: primary progressive apraxia of speech. Brain 135:1522–1536
Gil-Navarro S, Lladó A, Rami L et al (2013) Neuroimaging and biochemical markers in the three variants of primary progressive aphasia. Dement Geriatr Cogn Disord 35:106–117
Fernández-Matarrubia M, Matias-Guiu JA, Moreno-Ramos T et al (2013) Biomarkers: a new approach to behavioural variant of frontotemporal dementia. Neurologia. doi:10.1016/j.nrl.2013.03.002
Chare L, Hodges JR, Leyton CE et al (2014) New criteria for frontotemporal dementia syndromes: clinical and pathological diagnostic limitations. J Neurol Neurosurg Psychiatry. doi:10.1136/jnnp-2013-306948
Gorno-Tempini ML, Dronkers NF, Rankin KP et al (2004) Cognition and anatomy in three variants of primary progressive aphasia. Ann Neurol 55:335–346
Rabinovici GD, Jagust WJ, Furst AJ et al (2008) Abeta amyloid and glucose metabolism in three variants of primary progressive aphasia. Ann Neurol 64:388–401
Conflicts of interest
The authors report no disclosures.
Ethical standard
The study was performed according to ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
415_2014_7608_MOESM4_ESM.pptx
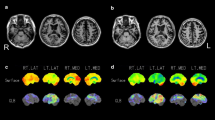
Figure A.3. PPA variants versus healthy controls. SPM map (neurological orientation). Regions with a lower metabolism in the group of gvPPA (blue), svPPA (green) and lvPPA (yellow) in comparison to healthy controls are shown (PPTX 1083 kb)
Rights and permissions
About this article
Cite this article
Matias-Guiu, J.A., Cabrera-Martín, M.N., Moreno-Ramos, T. et al. Clinical course of primary progressive aphasia: clinical and FDG-PET patterns. J Neurol 262, 570–577 (2015). https://doi.org/10.1007/s00415-014-7608-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-014-7608-0