Abstract
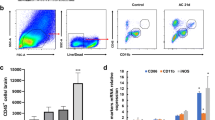
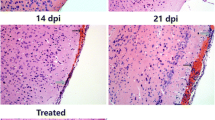
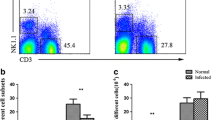
Angiostrongylus cantonensis is a neurotropic parasite which can cause injury to central nervous system and eosinophilic meningitis to human. Natural killer (NK) cells are specialized innate lymphocytes important in early defense against pathogens as in a variety of intracellular bacterial, viral, and protozoan infections. However, the number and function of NK cells in extracellular parasitic infection of A. cantonensis are unclear. In this study, on A. cantonensis infected mice which may mimic the human’s infection, we found that the percentage of splenic NK cells and the absolute number of peripheral blood NK cells were decreased at 21-day post infection compared with that of controls. When administrating with albendazole treatment at early stage of the infection, the changes of NK cells could be avoided. Further analysis confirmed that the reduction of NK cells was due to their apoptosis manifested as increased expressions of annexin V and activated caspase-3 after 16-day post infection. Moreover, both activated and inhibitory receptors such as CD16, CD69, NKG2D, and Ly49a on NK cells were down-regulated after 16-day post infection. Interestingly, NK cells isolated from mice of 21-day post infection showed enhanced IFN-γ production when stimulated with IL-12 for 24 h and cytotoxicity to YAC-1 cells, as well as elevated CD107a expression. It is evident that NK cell population and its function were changed in A. cantonensis infected mice, suggesting their involvement in pathogenesis of the infection.








Similar content being viewed by others
References
Aktas E, Kucuksezer UC, Bilgic S, Erten G, Deniz G (2009) Relationship between CD107a expression and cytotoxic activity. Cell Immunol 254(2):149–154
Alter G, Malenfant JM, Altfeld M (2004) CD107a as a functional marker for the identification of natural killer cell activity. J Immunol Methods 294(1–2):15–22
Andoniou CE et al (2005) Interaction between conventional dendritic cells and natural killer cells is integral to the activation of effective antiviral immunity. Nat Immunol 6(10):1011–1019
Bany J, Janiak MK, Budzynski W (1992) Activity of natural killer (NK) cells in the course of experimental trichinellosis in mice. Wiad Parazytol 38(3–4):117–126
Betts MR et al (2003) Sensitive and viable identification of antigen-specific CD8+ T cells by a flow cytometric assay for degranulation. J Immunol Methods 281(1–2):65–78
Brattig NW, Tischendorf FW, Albiez EJ, Buttner DW, Berger J (1987) Distribution pattern of peripheral lymphocyte subsets in localized and generalized form of onchocerciasis. Clin Immunol Immunopathol 44(2):149–159
Cardillo F, Voltarelli JC, Reed SG, Silva JS (1996) Regulation of Trypanosoma cruzi infection in mice by gamma interferon and interleukin 10: Role of NK cells. Infect Immun 64(1):128–134
Cerwenka A, Lanier LL (2001) Ligands for natural killer cell receptors: Redundancy or specificity. Immunol Rev 181:158–169
Chowdhury D, Lieberman J (2008) Death by a thousand cuts: Granzyme pathways of programmed cell death. Annu Rev Immunol 26:389–420
De La Garza R, 2nd, Asnis GM, Fabrizio KR, Pedrosa E (2005) Acute diclofenac treatment attenuates lipopolysaccharide-induced alterations to basic reward behavior and HPA axis activation in rats. Psychopharmacology (Berl) 179(2):356–65
Diefenbach A, Raulet DH (2001) Strategies for target cell recognition by natural killer cells. Immunol Rev 181:170–184
Elsner L et al (2007) The heat shock protein HSP70 promotes mouse NK cell activity against tumors that express inducible NKG2D ligands. J Immunol 179(8):5523–5533
Gelis S, Spratt DM, Raidal SR (2011) Neuroangiostrongyliasis and other parasites in tawny frogmouths (Podargus strigoides) in south-eastern Queensland. Aust Vet J 89(1–2):47–50
Grzywacz B et al (2006) Coordinated acquisition of inhibitory and activating receptors and functional properties by developing human natural killer cells. Blood 108(12):3824–3833
Guo PJ, et al. (2008) [Pathological change in the brain of mice infected with Angiostrongylus cantonensis]. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi 26(5):353–5
Hamerman JA, Ogasawara K, Lanier LL (2004) Cutting edge: Toll-like receptor signaling in macrophages induces ligands for the NKG2D receptor. J Immunol 172(4):2001–2005
Karlhofer FM, Ribaudo RK, Yokoyama WM (1992) The interaction of Ly-49 with H-2Dd globally inactivates natural killer cell cytolytic activity. Trans Assoc Am Physicians 105:72–85
Korten S et al (2002) Expansion of NK cells with reduction of their inhibitory Ly-49A, Ly-49C, and Ly-49G2 receptor-expressing subsets in a murine helminth infection: Contribution to parasite control. J Immunol 168(10):5199–5206
Lee JD, Wang JJ, Chang JH, Chung LY, Chen ER, Yen CM (1996) Role of T cell subpopulations in mice infected with Angiostrongylus cantonensis. J Helminthol 70(3):211–214
Lindo JF et al (2002) Enzootic Angiostrongylus cantonensis in rats and snails after an outbreak of human eosinophilic meningitis, Jamaica. Emerg Infect Dis 8(3):324–326
Liu H, Luo X, Shen E, Li H, Ding X, Chen D (2013) Alteration of T cell subtypes in spleen and antibodies of serum in mice infected with Angiostrongylus cantonensis. Parasitol Res 112(3):1255–1260
Lucas M, Schachterle W, Oberle K, Aichele P, Diefenbach A (2007) Dendritic cells prime natural killer cells by trans-presenting interleukin 15. Immunity 26(4):503–517
Lunemann A, Lunemann JD, Munz C (2009) Regulatory NK-cell functions in inflammation and autoimmunity. Mol Med 15(9–10):352–358
Maldonado A Jr et al (2010) First report of Angiostrongylus cantonensis (Nematoda: Metastrongylidae) in Achatina fulica (Mollusca: Gastropoda) from Southeast and South Brazil. Mem Inst Oswaldo Cruz 105(7):938–941
Marquez ME et al (2010) CD16 cross-linking induces increased expression of CD56 and production of IL-12 in peripheral NK cells. Cell Immunol 264(1):86–92
Mrakovcic-Sutic I et al (2010) Early changes in frequency of peripheral blood lymphocyte subpopulations in severe traumatic brain-injured patients. Scand J Immunol 72(1):57–65
Nakano Y, Onozuka K, Terada Y, Shinomiya H, Nakano M (1990) Protective effect of recombinant tumor necrosis factor-alpha in murine salmonellosis. J Immunol 144(5):1935–1941
Newman KC, Riley EM (2007) Whatever turns you on: Accessory-cell-dependent activation of NK cells by pathogens. Nat Rev Immunol 7(4):279–291
Newman KC, Korbel DS, Hafalla JC, Riley EM (2006) Cross-talk with myeloid accessory cells regulates human natural killer cell interferon-gamma responses to malaria. PLoS Pathog 2(12):e118
OuYang L et al (2012) Differences of larval development and pathological changes in permissive and nonpermissive rodent hosts for Angiostrongylus cantonensis infection. Parasitol Res 111(4):1547–1557
Pegram HJ, Andrews DM, Smyth MJ, Darcy PK, Kershaw MH (2011) Activating and inhibitory receptors of natural killer cells. Immunol Cell Biol 89(2):216–224
Perussia B (1998) Fc receptors on natural killer cells. Curr Top Microbiol Immunol 230:63–88
Prass K et al (2003) Stroke-induced immunodeficiency promotes spontaneous bacterial infections and is mediated by sympathetic activation reversal by poststroke T helper cell type 1-like immunostimulation. J Exp Med 198(5):725–736
Raulet DH, Gasser S, Gowen BG, Deng W, Jung H (2013) Regulation of ligands for the NKG2D activating receptor. Annu Rev Immunol 31:413–441
Schleicher U et al (2007) NK cell activation in visceral leishmaniasis requires TLR9, myeloid DCs, and IL-12, but is independent of plasmacytoid DCs. J Exp Med 204(4):893–906
Slom TJ et al (2002) An outbreak of eosinophilic meningitis caused by Angiostrongylus cantonensis in travelers returning from the Caribbean. N Engl J Med 346(9):668–675
Teixeira-Carvalho A et al (2008) Binding of excreted and/or secreted products of adult hookworms to human NK cells in Necator americanus-infected individuals from Brazil. Infect Immun 76(12):5810–5816
Trapani JA, Jans DA, Jans PJ, Smyth MJ, Browne KA, Sutton VR (1998) Efficient nuclear targeting of granzyme B and the nuclear consequences of apoptosis induced by granzyme B and perforin are caspase-dependent, but cell death is caspase-independent. J Biol Chem 273(43):27934–27938
Tseng RJ, Padgett DA, Dhabhar FS, Engler H, Sheridan JF (2005) Stress-induced modulation of NK activity during influenza viral infection: Role of glucocorticoids and opioids. Brain Behav Immun 19(2):153–164
Wang QP, Lai DH, Zhu XQ, Chen XG, Lun ZR (2008) Human angiostrongyliasis. Lancet Infect Dis 8(10):621–630
Wang J et al (2010) An outbreak of angiostrongyliasis cantonensis in Beijing. J Parasitol 96(2):377–381
Yokoyama WM, Kim S, French AR (2004) The dynamic life of natural killer cells. Annu Rev Immunol 22:405–429
Acknowledgments
This work was supported by National Basic Research Program of China (973 Program) (No. 2010CB530004), the Postgraduate Student Research Training Program of Jiangsu Province (CXZZ13-0566), and the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The authors have declared that no competing interests exist.
We thank Prof. Zhong-Dao Wu, Sun Yat-sen University, Guangzhou, China, for providing us with A. cantonensis infected snails and for his helpful suggestions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, Al., Qiu, Xy., Wang, W. et al. The quantitative and functional changes of NK cells in mice infected with Angiostrongylus cantonensis . Parasitol Res 113, 2087–2094 (2014). https://doi.org/10.1007/s00436-014-3858-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-014-3858-0