Abstract
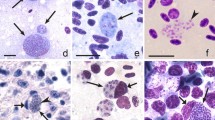
Avian haemosporidian parasites of the genus Haemoproteus (Haemoproteidae, Haemosporida) are widespread, and some species cause diseases both in vertebrate hosts and blood-sucking insects. Parasitemia of Haemoproteus species usually is long-lasting, with gametocytes present in the circulation for several months. However, the viability of gametocytes and their ability to produce sexual cells have been insufficiently understood in the course of parasitemia. We initiated the sexual development in vitro conditions and calculated proportions of normal and anomalous ookinetes, which developed in two species of Haemoproteus. Mature gametocytes of the parasites were obtained from naturally infected avian hosts at different days of parasitemia. Haemoproteus (Parahaemoproteus) lanii (cytochrome b lineage hRB1) was isolated from one red-backed shrike Lanius collurio. Two isolates of Haemoproteus (Parahaemoproteus) tartakovskyi (cytochrome b lineage hSISKIN1) were used: one was obtained from a siskin Carduelis spinus and one from a common crossbill Loxia curvirostra. The wild-caught birds were kept indoors under controlled conditions, and blood was taken from them every 1 or 2 days during 10–14 days. After each blood sampling, the sexual process and ookinete development were initiated in vitro by exposure of infected blood containing mature gametocytes to air. Smears were prepared at intervals of 15 min, 3 h, and 12 h after the exposure; they were examined microscopically. In all, 25 experiments were performed; each experiment was repeated two times. The ratios of macro- and microgametocytes did not change in all experimental infections during this study. Sexual process occurred, and both normal and anomalous ookinetes developed in all parasites. The proportion of normal ookinetes did not change significantly in both isolates of H. tartakovskyi. Between 8 and 10 days of observation, the proportion of normal ookinetes of H. lanii decreased 6 times compared to the beginning of the experiment. That was accompanied with the rapid decrease of parasitemia and the inability of the majority of mature gametocytes to escape from erythrocytes and produce gametes, indicating disorder of the gametogenesis. There was clear difference in the gametogenesis between H. tartakovskyi and H. lanii from this point of view. This study shows that the viability of Haemoproteus gametocytes might change dramatically in the course of parasitemia within 1–2 days, and the presence of mature gametocytes in the circulation does not necessarily indicate their ability to exflagellate and produce ookinetes. We predict that this finding is important epidemiologically due to relationship with sporogony success.


Similar content being viewed by others
References
Ahmed FE, Mohammed AH (1978) Haemoproteus columbae: course of infection, relapse and immunity to reinfection in the pigeon. Parasitol Res 57:229–236
Applegate JE (1971) Spring relapse of Plasmodium relictum infection in an experimental field population of English sparrows (Passer domesticus). J Wildl Dis 7:37–42
Atkinson CT, Thomas NJ, Hunter BC (2008) Parasitic diseases of wild birds. Wiley-Blackwell, Ames
Bensch S, Stjernman M, Hasselquist D, Östman Ö, Hansson B, Westerdahl H, Torres-Pinheiro R (2000) Host specificity in avian blood parasites: a study of Plasmodium and Haemoproteus mitochondrial DNA amplified from birds. Proc R Soc B 276:1583–1589
Bensch S, Hellgren O, Pérez-Tris J (2009) MalAvi: a public database of malaria parasites and related haemosporidians in avian hosts based on mitochondrial cytochrome b lineages. Mol Ecol Resour 9:1353–1358
Carter R, Graves PM (1988) Gametocytes. In: Wernsdorfer WH, McGregor I (eds) Malaria, vol 1, Principles and practice of malariology. Churchill Livingstone, Edinburgh, pp 253–305
Coatney GR (1933) Relapse and associated phenomena in the Haemoproteus infection of the pigeon. Am J Hyg 18:133–160
Desser SS (1972) Gametocyte maturation, exflagellation and fertilization in Parahaemoproteus (=Haemoproteus) velans (Coatney et Roudabush) (Haemosporidia: Haemoproteidae): an ultrastructural study. J Protozool 19:287–296
Fallis AM, Bennett GF (1960) Description of Haemoproteus canachites n. sp. (Sporozoa: Haemoproteidae) and sporogony in Culicoides (Diptera: Ceratopogonidae). Can J Zool 38:455–464
Ferrell ST, Snowden K, Marlar AB, Garner M, Lung NP (2007) Fatal hemoprotozoal infections in multiple avian species in a zoological park. J Zoo Wildl Med 38:309–316
Garnham PCC (1966) Malaria parasites and other haemosporidia. Blackwell, Oxford
Ghosh A, Edwards MJ, Jacobs-Lorena M (2000) The journey of the malaria parasite in the mosquito: hopes for the new century. Parasitol Today 16:196–201
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucleic Acids Symp Ser (Oxf) 41:95–98
Hellgren O, Waldenström J, Bensch S (2004) A new PCR assay for simultaneous studies of Leucocytozoon, Plasmodium, and Haemoproteus from avian blood. J Parasitol 90:797–802
Martínez J, Martínez-De La Puente J, Herrero J, Del Cerro S, Lobato E, Rivero-De Aguilar J, Vásquez RA, Merino S (2009) A restriction site to differentiate Plasmodium and Haemoproteus infections in birds: on the inefficiency of general primers for detection of mixed infections. Parasitology 136:713–722
Marzal A, de Lopes F, Navarro C, Møller AP (2005) Malarial parasites decrease reproductive success: an experimental study in a passerine bird. Oecologia 142:541–545
Pacheco MA, Escalante AA, Garner MM, Bradley GA, Aquilar RF (2011) Haemosporidian infection in captive masked bobwhite quail (Colinus virginianus ridgwayi), an endangered subspecies of the northern bobwhite quail. Vet Parasitol 182:113–120
Palinauskas V, Križanauskienė A, Iezhova TA, Bolshakov CV, Jönsson J, Bensch S, Valkiūnas G (2013) A new method for isolation of purified genomic DNA from haemosporidian parasites inhabiting nucleated red blood cells. Exp Parasitol 133:275–280
Perkins SL (2014) Malaria’s many mates: past, present and future of the systematics of the order Haemosporida. J Parasitol 100:11–25
Shurulinkov P, Chakarov N, Daskalova G (2012) Blood parasites, body condition, and wing length in two subspecies of yellow wagtail (Motacilla flava) during migration. Parasitol Res 110:2043–2051
Sibley LD, Werner JK (1984) Susceptibility of pekin and muscovy ducks to Haemoproteus nettionis. J Wildl Dis 20:108–113
Sinden RE (1998) Gametogenesis and sexual development. In: Sherman IW (ed) Malaria: parasite biology, pathogenesis, and protection. American Society for Microbiology Press, Washington, DC, pp 25–48
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Valkiūnas G (2005) Avian malaria parasites and other haemosporidia. CRC, Boca Raton
Valkiūnas G, Iezhova TA, Križanauskienė A, Palinauskas V, Bensch S (2008) In vitro hybridization of Haemoproteus spp.: an experimental approach for direct investigation of reproductive isolation of parasites. J Parasitol 94:1385–1394
Valkiūnas G, Palinauskas V, Križanauskienė A, Bernotienė R, Kazlauskienė R, Iezhova TA (2013) Further observations on in vitro hybridization of hemosporidian parasites: patterns of ookinete development in Haemoproteus spp. J Parasitol 99:124–136
Valkiūnas G, Kazlauskienė R, Bernotienė R, Bukauskaite D, Palinauskas V, Iezhova TA (2014a) Haemoproteus infections (Haemosporida, Haemoproteidae) kill bird-biting mosquitoes. Parasitol Res 113:1011–1018
Valkiūnas G, Palinauskas V, Ilgūnas M, Bernotienė R, Iezhova TA (2014b) In vitro development of Haemoproteus parasites: the efficiency of reproductive cells increase during simultaneous sexual process of different lineages. Parasitol Res 113:1417–1423
Valkiūnas G (2015) Haemoproteus. In: Mehlhorn H (ed) Encyclopedia of parasitology, 4th edn. Springer, Heidelberg
Acknowledgments
We would like to thank the staff of the Biological Station “Rybachy”, for assistance in the field. The director of the Biological Station “Rybachy”, Casimir V. Bolshakov, is acknowledged for generously providing facilities for the experimental research and Dovilė Bukauskaitė and Rita Žiegytė, for participation in fieldwork and assistance in the laboratory. This research was supported by the Open Access to research infrastructure of the Nature Research Centre under the Lithuanian open access network initiative.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Valkiūnas, G., Iezhova, T.A., Palinauskas, V. et al. The evidence for rapid gametocyte viability changes in the course of parasitemia in Haemoproteus parasites. Parasitol Res 114, 2903–2909 (2015). https://doi.org/10.1007/s00436-015-4491-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4491-2