Abstract
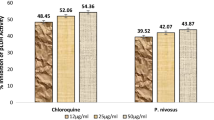
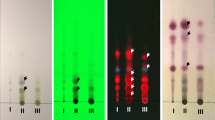
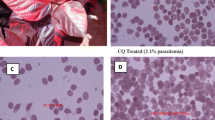
In an effort to identify novel therapeutic alternatives for the treatment of malaria, the present study evaluated the antimalarial effect of the crude hydroalcoholic extract (HCE) from the leaves of Chenopodium ambrosioides L. For this purpose, the molecular affinity between the total proteins from erythrocytes infected with Plasmodium falciparum and HCE or chloroquine was evaluated by surface plasmon resonance (SPR). Subsequently, the plasmodicidal potential of HCE was assessed in a P. falciparum culture. Using BALB/c mice infected with Plasmodium berghei intraperitoneally (ip.), we evaluated the effects of ip. treatment, for three consecutive days (day 7, 8, and 9 after infection), with chloroquine (45 mg/kg) or HCE (5 mg/kg), considering the survival index and the parasitaemia. The groups were compared to an untreated control group that receives only PBS at the same periods. The results indicated that HCE could bind to the total proteins of infected erythrocytes and could inhibit the parasite growth in vitro (IC50 = 25.4 g/mL). The in vivo therapeutic treatment with HCE increased the survival and decreased the parasitaemia in the infected animals. Therefore, the HCE treatment exhibited a significant antiplasmodial effect and may be considered as a potential candidate for the development of new antimalarial drugs.




Similar content being viewed by others
References
Bantie L, Assefa S, Teklehaimanot T, Engidawork E (2014) In vivo antimalarial activity of the crude leaf extract and solvent fractions of Croton macrostachyus Hocsht. (Euphorbiaceae) against Plasmodium berghei in mice. BMC Complement Altern Med 14:79. doi:10.1186/1472-6882-14-79
Basco LK, Mitaku S, Skaltsounis AL, Ravelomanantsoa N, Tillequin F, Koch M, Le Bras J (1994) In vitro activities of furoquinoline and acridone alkaloids against Plasmodium falciparum. Antimicrob Agents Chemother 38:1169–1171. doi:10.1128/AAC.38.5.1169
Batista R, Silva-Júnior AJ, Oliveira AB (2009) Plant-derived antimalarial agents: new leads and efficient phytomedicines. Part II. non-alkaloidal natural products. Molecules 14:3037–3072. doi:10.3390/molecules14083037
Bezerra JL, Costa GC, Lopes TC, Carvalho ICDS, Patrício FJ, Sousa SM, Amaral FMM, Rebelo JMM, Guerra RNM, Ribeiro MNS, Nascimento FRF (2006) Avaliação da atividade leishmanicida in vitro de plantas medicinais. Brazilian J Pharmacogn 16:631–637
Bienvenu AL, Picot S (2008) Statins alone are ineffective in cerebral malaria but potentiate artesunate. Antimicrob Agents Chemother 52:4203–4204. doi:10.1128/AAC.00513-08
Brasil (2008) Ministério Da Saúde, Secretaria de Ciência, Tecnologia e Insumos Estratégicos. Portaria Interministerial n° 2960 em 9 de dezembro de 2008. Programa de Nacional de Plantas Medicinais e Fitoterápicos. Diário Oficial da União, 10 de dezembro
Conway GA, Slocumb JC (1979) Plants used as abortifacients and emmenagogues by Spanish New Mexicans. J Ethnopharmacol 1:241–261. doi:10.1016/S0378-8741(79)80014-8
Cruz GVB, Pereira PVS, Patrício FJ, Costa GC, Sousa SM, Frazão JB, Aragão-Filho WC, Maciel MCG, Silva LA, Amaral FMM, Barroqueiro ESB, Guerra RNM, Nascimento FRF (2007) Increase of cellular recruitment, phagocytosis ability and nitric oxide production induced by hydroalcoholic extract from Chenopodium ambrosioides leaves. J Ethnopharmacol 111:148–154. doi:10.1016/j.jep.2006.11.006
Cui L, Su X (2009) Discovery, mechanisms of action and combination therapy of artemisinin. Expert Rev Anti Infect Ther 7:999–1013. doi:10.1586/eri.09.68
Deharo E, Bourdy G, Quenevo C, Muñoz V, Ruiz G, Sauvain M (2001) A search for natural bioactive compounds in Bolivia through a multidisciplinary approach. Part V. Evaluation of the antimalarial activity of plants used by the Tacana Indians. J Ethnopharmacol 77:91–98. doi:10.1016/S0378-8741(01)00270-7
Di Stasi L, Hiruma-Lima C (2002) Plantas medicinais na Amazônia e na Mata Atlântica. In: 2nd edn. UNESP, São Paulo, pp 163–164
Dolabela MF, Oliveira SG, Nascimento JM, Peres JM, Wagner H, Póvoa MM, de Oliveira AB (2008) In vitro antiplasmodial activity of extract and constituents from Esenbeckia febrifuga, a plant traditionally used to treat malaria in the Brazilian Amazon. Phytomedicine 15:367–372. doi:10.1016/j.phymed.2008.02.001
Fitch CD (2004) Ferriprotoporphyrin IX, phospholipids, and the antimalarial actions of quinoline drugs. Life Sci 74:1957–1972. doi:10.1016/j.lfs.2003.10.003
França F, Lago EL, Marsden PD (1996) Plants used in the treatment of leishmanial ulcers due to Leishmania (Viannia) braziliensis in an endemic area of Bahia, Brazil. Rev Soc Bras Med Trop 29:229–232
Gupta M, Mazumder UK, Rath N, Mukhopadhyay DK (2000) Antitumor activity of methanolic extract of Cassia fistula L. seed against Ehrlich ascites carcinoma. J Ethnopharmacol 72:151–156. doi:10.1016/S0378-8741(00)00227-0
Hanum SP, Hayano M, Kojima S (2003) Cytokine and chemokine responses in a cerebral malaria-susceptible or -resistant strain of mice to Plasmodium berghei ANKA infection: early chemokine expression in the brain. Int Immunol 15:633–640. doi:10.1093/intimm/dxg065
Hilou A, Nacoulma OG, Guiguemde TR (2006) In vivo antimalarial activities of extracts from Amaranthus spinosus L. and Boerhaavia erecta L. in mice. J Ethnopharmacol 103:236–240. doi:10.1016/j.jep.2005.08.006
Hyde JE (2002) Mechanisms of resistance of Plasmodium falciparum to antimalarial drugs. Microbes Infect 4:165–174. doi:10.1016/S1286-4579(01)01524-6
Janse CJ, Van Vianen PH (1994) Flow cytometry in malaria detection. Methods Cell Biol 42:295–318
Janse CJ, Franke-Fayard B, Mair GR, Ramesar J, Thiel C, Engelmann S, Matuschewski K, Van Gemert GJ, Sauerwein RW, Waters AP (2006a) High efficiency transfection of Plasmodium berghei facilitates novel selection procedures. Mol Biochem Parasitol 145:60–70. doi:10.1016/j.molbiopara.2005.09.007
Janse CJ, Franke-Fayard B, Waters AP (2006b) Selection by flow-sorting of genetically transformed, GFP-expressing blood stages of the rodent malaria parasite, Plasmodium berghei. Nat Protoc 1:614–623. doi:10.1038/nprot.2006.88
Jordão FM, Saito AY, Miguel DC, de Jesus PV, Kimura EA, Katzin AM (2011) In vitro and in vivo antiplasmodial activities of risedronate and its interference with protein prenylation in Plasmodium falciparum. Antimicrob Agents Chemother 55:2026–2031. doi:10.1128/AAC.01820-10
Juge N, Moriyama S, Miyaji T, Kawakami M, Iwai H, Fukui T, Nelson N, Omote H, Moriyama Y (2015) Plasmodium falciparum chloroquine resistance transporter is a H+ -coupled polyspecific nutrient and drug exporter. Proc Natl Acad Sci. doi:10.1073/pnas.1417102112
Kantele A, Jokiranta TS (2011) Review of cases with the emerging fifth human malaria parasite, Plasmodium knowlesi. Clin Infect Dis 52:1356–1362. doi:10.1093/cid/cir180
Kliks MM (1985) Studies on the traditional herbal anthelmintic Chenopodium ambrosioides L.: ethnopharmacological evaluation and clinical field trials
Lambros C, Vanderberg JP (1979) Synchronization of Plasmodium falciparum erythrocytic stages in culture. J Parasitol 65:418–420
Lima-Júnior JAC, Costa GC, Bezerra JL, Patrício FJB, Silva LA, Amaral FMM, Nascimento FRF (2014) Inibição da infecção in vitro de macrófagos por Leishmania amazonensis por extrato e frações de Chenopodium ambrosioides L. Rev Ciências da Saúde 16:46–53
Liu J, Modrek S, Gosling RD, Feachem RGA (2013) Malaria eradication: is it possible? Is it worth it? Should we do it? Lancet Glob Heal 1:e2–e3. doi:10.1016/S2214-109X(13)70002-0
MacDonald D, VanCrey K, Harrison P, Rangachari PK, Rosenfeld J, Warren C, Sorger G (2004) Ascaridole-less infusions of Chenopodium ambrosioides contain a nematocide(s) that is(are) not toxic to mammalian smooth muscle. J Ethnopharmacol 92:215–221. doi:10.1016/j.jep.2004.02.018
Monzote L, Montalvo AM, Almanonni S, Scull R, Miranda M, Abreu J (2006) Activity of the essential oil from Chenopodium ambrosioides grown in Cuba against Leishmania amazonensis. Chemotherapy 52:130–136. doi:10.1159/000092858
Monzote L, García M, Pastor J, Gil L, Scull R, Maes L, Cos P, Gille L (2014) Essential oil from Chenopodium ambrosioides and main components: activity against Leishmania, their mitochondria and other microorganisms. Exp Parasitol 136:20–26. doi:10.1016/j.exppara.2013.10.007
Moumaris M, Sestier C, Miltgen F, Halbreich A, Gentilini M, Sabolovic D (1995) Effect of fatty acid treatment in cerebral malaria-susceptible and nonsusceptible strains of mice. J Parasitol 81:997–999
Muñoz V, Sauvain M, Bourdy G, Callapa J, Bergeron S, Rojas I, Bravo JA, Balderrama L, Ortiz B, Gimenez A, Deharo E (2000) A search for natural bioactive compounds in Bolivia through a multidisciplinary approach. Part I. Evaluation of the antimalarial activity of plants used by the Chacobo Indians. J Ethnopharmacol 69:127–137. doi:10.1016/S0378-8741(99)00148-8
Nascimento FRF, Cruz GVB, Pereira PVS, Maciel MCG, Silva LA, Azevedo APS, Barroqueiro ESB, Guerra RNM (2006) Ascitic and solid Ehrlich tumor inhibition by Chenopodium ambrosioides L. treatment. Life Sci 78:2650–2653. doi:10.1016/j.lfs.2005.10.006
Neiva VS, Ribeiro MNS, Cartágenes MSS, Moraes-Coutinho DF, Nascimento FRF, Reis AS, Amaral FMM (2011) Estudos pré-clínicos de atividade giardicida de Chenopodium ambrosioides L. e a padronização dos extratos na pesquisa e desenvolvimento de fitoterápicos. Rev Ciências da Saúde 13:155–165
Neiva VA, Ribeiro MNS, Nascimento FRF, Cartágenes MSS, Coutinho-Moraes DF, Amaral FMM (2014) Plant species used in giardiasis treatment: ethnopharmacology and in vitro evaluation of anti-Giardia activity. Brazilian J Pharmacogn 24:215–224. doi:10.1016/j.bjp.2014.04.004
Oliveira AB, Dolabela MF, Braga FC, Jácome RL, Varotti FP, Póvoa MM (2009) Plant-derived antimalarial agents: new leads and efficient phythomedicines. Part I. alkaloids. An Acad Bras Cienc 81:715–740. doi:10.1590/S0001-37652009000400011
Osorio E, Arango GJ, Jiménez N, Alzate F, Ruiz G, Gutiérrez D, Paco MA, Giménez A, Robledo S (2007) Antiprotozoal and cytotoxic activities in vitro of Colombian Annonaceae. J Ethnopharmacol 111:630–635. doi:10.1016/j.jep.2007.01.015
Patrício FJ, Costa GC, Pereira PVS, Aragão-Filho WC, Sousa SM, Frazão JB, Pereira WS, Maciel MCG, Silva LA, Amaral FMM, Rebêlo JMM, Guerra RNM, Ribeiro MNS, Nascimento FRF (2008) Efficacy of the intralesional treatment with Chenopodium ambrosioides in the murine infection by Leishmania amazonensis. J Ethnopharmacol 115:313–319. doi:10.1016/j.jep.2007.10.009
Pollack Y, Segal R, Golenser J (1990) The effect of ascaridole on the in vitro development of Plasmodium falciparum. Parasitol Res 76:570–572
Reis AS, Rios CEP, Melo LP, Costa GC, Silva LA, Patrício FJB, Amaral FMM, Nascimento FRF (2012a) Atividade leishmanicida in vitro de frações do extrato hidroalcoólico das folhas de Chenopodium ambrosioides L. Rev Ciências da Saúde 14:119–126
Reis PA, Estato V, da Silva TI, d’Avila JC, Siqueira LD, Assis EF, Bozza PT, Bozza FA, Tibiriça EV, Zimmerman GA, Castro-Faria-Neto HC (2012b) Statins decrease neuroinflammation and prevent cognitive impairment after cerebral malaria. Plos Pathog. doi:10.1371/journal.ppat.1003099
Saxena S, Pant N, Jain DC, Bhakuni RS (2003) Antimalarial agents from plant sources. Curr Sci 85:1314–1329
Sousa LHA, Rios CEP, Assunção AKM, Fialho EMS, Costa GC, Nascimento FRF (2012) Avaliação da ação analgésica do extrato hidroalcoólico das folhas de Chenopodium ambrosioides L. em ensaios pré-clínicos. Rev Ciências da Saúde 14:73–82
Trager W, Jensen JB (1976) Human malaria parasites in continuous culture. Science 193:673–675
Trivellato-Grassi L, Malheiros A, Meyre-Silva C, Buss Zda S, Monguilhott ED, Fröde TS, da Silva KABS, de Souza MM (2013) From popular use to pharmacological validation: a study of the anti-inflammatory, anti-nociceptive and healing effects of Chenopodium ambrosioides extract. J Ethnopharmacol 145:127–138. doi:10.1016/j.jep.2012.10.040
Waako PJ, Gumede B, Smith P, Folb PI (2005) The in vitro and in vivo antimalarial activity of Cardiospermum halicacabum L. and Momordica foetida Schumch. Et Thonn. J Ethnopharmacol 99:137–143. doi:10.1016/j.jep.2005.02.017
World Health Organization (2013) Emergency response to artemisinin resistance in the Greater Mekong subregion: regional framework for action 2013-2015
World Health Organization (2014) Malaria world report
Wright CW (2005) Plant derived antimalarial agents: new leads and challenges. Phytochem Rev 4:55–61. doi:10.1007/s11101-005-3261-7
Acknowledgments
The authors are grateful to Valnice de Jesus Peres and Dr. Alejandro Miguel Katzin for technical support and to Programa de Desenvolvimento Tecnológico em Insumos para Saúde (PDTIS-FIOCRUZ) for allowing the use of their facilities. The authors also would like to thank the funding agencies Fundação de Amparo à Pesquisa e ao Desenvovilmento Científico e Tecnológico do Maranhão (FAPEMA), Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP), Conselho Nacional de Desenvolvimento em Pesquisa (CNPq), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES). CRFM is enrolled at the Programa Estratégico de Ciência, Tecnologia & Inovação nos Programas de Pós-Graduação do Estado do Amazonas (PECTI/AM-PG) from Fundação de Amparo à Pesquisa do Estado do Amazonas (FAPEAM, Brazil).
Authorship policy
Conceived and designed the experiments: DNC, TSF, CRFM, RN, and FRFN. Performed the experiments: DNC, TSF, ASR, and ASF. Analyzed the data: DNC, TSF, and BPR. Contributed reagents/materials/analysis tools: FMMA, CRFM, and RN. Wrote the paper: DNC, RNMG, RN and FRFN.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Cysne, D.N., Fortes, T.S., Reis, A.S. et al. Antimalarial potential of leaves of Chenopodium ambrosioides L.. Parasitol Res 115, 4327–4334 (2016). https://doi.org/10.1007/s00436-016-5216-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-016-5216-x