Abstract
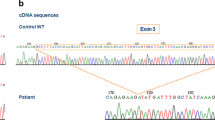
A membrane-bound cytochrome b 558 , a heterodimer consisting of gp91-phox and p22-phox, is a critical component of the superoxide (O2 -)-generating reduced nicotinamide adenine dinucleotide phosphate (NADPH) oxidase in phagocytes. Chronic granulomatous disease (CGD) is characterized by recurrent bacterial infection caused by a defect of the oxidase. Both subunits are absent from phagocytes in typical X-linked recessive CGD patients who are primarily defective in gp91-phox. We report here an atypical case of X-linked CGD in which neutrophils showed a complete absence of O2 –-forming NADPH oxidase activity, but a small amount (about 10% of control) of both subunits was detected by immunoblot analysis. Spectrophotometric studies of the neutrophils with a recently developed sensitive method gave no evidence for the heme spectrum in the cytochrome b 558 of this CGD. Reverse transcription/polymerase chain reaction and sequence analysis revealed a C to T transition replacing histidine at amino acid position 101 (His101) by tyrosine in gp91-phox. These results provide evidence that His101 of gp91-phox is the one of the heme-binding ligands of cytochrome b 558 .
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 18 June 1998 / Accepted: 11 July 1998
Rights and permissions
About this article
Cite this article
Tsuda, M., Kaneda, M., Sakiyama, T. et al. A novel mutation at a probable heme-binding ligand in neutrophil cytochrome b 558 in atypical X-linked chronic granulomatous disease. Hum Genet 103, 377–381 (1998). https://doi.org/10.1007/s004390050836
Issue Date:
DOI: https://doi.org/10.1007/s004390050836