Abstract
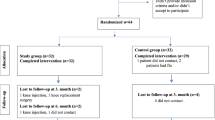
The purpose of the present investigation was to evaluate whether an anti-inflammatory effect together with an improvement of the regulation of the interaction between the inflammatory and stress responses underlies the clinical benefits of pelotherapy in osteoarthritis (OA) patients. This study evaluated the effects of a 10-day cycle of pelotherapy at the spa centre ‘El Raposo’ (Spain) in a group of 21 OA patients diagnosed with primary knee OA. Clinical assessments included pain intensity using a visual analog scale; pain, stiffness and physical function using the Western Ontario and McMaster Universities Arthritis Index; and health-related quality of life using the EuroQol-5D questionnaire. Serum inflammatory cytokine levels (IL-1β, TNF-α, IL-8, IL-6, IL-10 and TGF-β) were evaluated using the Bio-Plex® Luminex® system. Circulating neuroendocrine-stress biomarkers, such as cortisol and extracellular 72 kDa heat shock protein (eHsp72), were measured by ELISA. After the cycle of mud therapy, OA patients improved the knee flexion angle and OA-related pain, stiffness and physical function, and they reported a better health-related quality of life. Serum concentrations of IL-1β, TNF-α, IL-8, IL-6 and TGF-β, as well as eHsp72, were markedly decreased. Besides, systemic levels of cortisol increased significantly. These results confirm that the clinical benefits of mud therapy may well be mediated, at least in part, by its systemic anti-inflammatory effects and neuroendocrine-immune regulation in OA patients. Thus, mud therapy could be an effective alternative treatment in the management of OA.




Similar content being viewed by others
References
Altman R, Asch E, Bloch D, Bole G, Borenstein D, Brandt K, Christy W, Cooke TD, Greenwald R, Hochberg M et al (1986) Development of criteria for the classification and reporting of osteoarthritis. Classification of osteoarthritis of the knee. Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum 29(8):1039–1049
Asea A (2006) Initiation of the immune response by extracellular Hsp72: chaperokine activity of Hsp72. Curr Immunol Rev 2:209–215
Asea A, Kraeft SK, Kurt-Jones EA, Stevenson MA, Chen LB, Finberg RW, Koo GC, Calderwood SK (2000) Hsp70 stimulates cytokine production through a CD14-dependent pathway, demonstrating its dual role as a chaperone and cytokine. Nat Med 6:435–442
Bellamy N, Buchanan WW, Goldsmith CH, Campbell J, Stitt LW (1988) Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J Rheumatol 15(12):1833–1840
Bellometti S, Cecchettin M, Galzigna L (1997) Mud pack therapy in osteoarthrosis. Changes in serum levels of chondrocyte markers. Clin Chim Acta 268(1–2):101–106
Berenbaum F (2013) Osteoarthritis as an inflammatory disease (osteoarthritis is not osteoarthrosis!). Osteoarthr Cartil 21(1):16–21
Besedovsky HO, del Rey A (2000) The cytokine-HPA axis feed-back circuit. Z Rheumatol 59(2):26–30
Campisi J, Leem TM, Fleshner M (2003) Stress-induced extracellular Hsp72 is a functionally significant danger signal to the immune system. Cell Stress Chaperones 8:272–286
Carretero MI, Pozo M, Martín-Rubí JA, Pozo E, Maraver F (2010) Mobility of elements in interaction between artificial sweat and peloids used in Spanish spas. Appl Clay Sci 48(3):506–515
Cecchettin M, Bellometti S, Lalli A, Galzigna L (1995) Serum interleukin-1 changes in arthrosis patients after mud-pack treatment. Phys Rehab Kur Med 5:92–93
Daghestani HN, Kraus VB (2015) Inflammatory biomarkers in osteoarthritis. Osteoarthr Cartil 23(11):1890–1896
De la Fuente M, Cruces J, Hernandez O, Ortega E (2011) Strategies to improve the functions and redox state of the immune system in aged subjects. Curr Pharm Des 1 17(36):3966–3993
Elenkov IJ (2008) Neurohormonal-cytokine interactions: implications for inflammation, common human diseases and well-being. Neurochem Int 52(1–2):40–51
Espejo-Antúnez L, Cardero-Durán MA, Garrido-Ardila EM, Torres-Piles S, Caro-Puértolas B (2013a) Clinical effectiveness of mud pack therapy in knee osteoarthritis. Rheumatology (Oxford) 52(4):659–668
Espejo-Antúnez L, Caro-Puértolas B, Ibáñez-Burgos B, Porto-Payán JM, Torres-Piles ST (2013b) Effects of mud therapy on perceived pain and quality of life related to health in patients with knee osteoarthritis. Reumatol Clin 9(3):156–160
Fioravanti A, Bacaro G, Giannitti C, Tenti S, Cheleschi S, Gui Delli GM, Pascarelli NA, Galeazzi M (2015) One-year follow-up of mud-bath therapy in patients with bilateral knee osteoarthritis: a randomized, single-blind controlled trial. Int J Biometeorol 59(9):1333–1343
Fioravanti A, Cantarini L, Guidelli GM, Galeazzi M (2011) Mechanisms of action of spa therapies in rheumatic diseases: what scientific evidence is there? Rheumatol Int 31(1):1–8
Fioravanti A, Iacoponi F, Bellisai B, Cantarini L, Galeazzi M (2010) Short- and long-term effects of spa therapy in knee osteoarthritis. Am J Phys Med Rehabil 89(2):125–132
Fortunati NA, Fioravanti A, Seri G, Cinelli S, Tenti S (2016) May spa therapy be a valid opportunity to treat hand osteoarthritis? A review of clinical trials and mechanisms of action. Int J Biometeorol 60(1):1–8
Gálvez I, Torres-Piles S, Hinchado MD, Álvarez-Barrientos A, Torralbo-Jiménez P, Guerrero J, Martín-Cordero L, Ortega E (2017) Immune-neuroendocrine dysregulation in patients with osteoarthritis: a revision and a pilot study. Endocr Metab Immune Disord Drug Targets. doi:10.2174/1871530317666170320113613
Glyn-Jones S, Palmer AJR, Agricola R, Price AJ, Vincent TL, Weinans H, Carr AJ (2015) Osteoarthritis. Lancet 386(9991):376–387
Gomes C, Carretero MI, Pozo M, Maraver F, Cantista P, Armijo F, Legido JL, Teixeira F, Rautureau M, Delgado R (2013) Peloids and pelotherapy: historical evolution, classification and glossary. Appl Clay Sci 75-76:28–38
Harzy T, Ghani N, Akasbi N, Bono W, Nejjari C (2009) Short- and long-term therapeutic effects of thermal mineral waters in knee osteoarthritis: a systematic review of randomized controlled trials. Clin Rheumatol 28(5):501–507
Heinegård D, Saxne T (2011) The role of the cartilage matrix in osteoarthritis. Nat Rev Rheumatol 7(1):50–56
Hochberg MC, Altman RD, April KT, Benkhalti M, Guyatt G, McGowan J, Towheed T, Welch V, Wells G, Tugwell P (2012) American College of Rheumatology 2012 recommendations for the use of nonpharmacologic and pharmacologic therapies in osteoarthritis of the hand, hip, and knee. Arthritis Care Res (Hoboken) 64(4):465–474
Hügle T, Geurts J (2016) What drives osteoarthritis?—synovial versus subchondral bone pathology. Rheumatology (Oxford). doi:10.1093/rheumatology/kew389
Jin X, Beguerie JR, Zhang W, Blizzard L, Otahal P, Jones G, Ding C (2015) Circulating C reactive protein in osteoarthritis: a systematic review and meta-analysis. Ann Rheum Dis 74(4):703–710
Kuczera M, Kokot F (1996) Effect of spa therapy on the endocrine system. I Stress reaction hormones Pol Arch Med Wewn 95(1):11–20
McAlindon TE, Bannuru RR, Sullivan MC, Arden NK, Berenbaum F, Bierma-Zeinstra SM, Hawker GA, Henrotin Y, Hunter DJ, Kawaguchi H, Kwoh K, Lohmander S, Rannou F, Roos EM, Underwood M (2014) OARSI guidelines for the non-surgical management of knee osteoarthritis. Osteoarthr Cartil 22(3):363–388
Miller RE, Miller RJ, Malfait AM (2014) Osteoarthritis joint pain: the cytokine connection. Cytokine 70(2):185–193
Neogi T (2013) The epidemiology and impact of pain in osteoarthritis. Osteoarthr Cartil 21(9):1145–1153
Ortega E, Bote ME, Besedovsky HO, del Rey A (2012) Hsp72, inflammation, and aging: causes, consequences, and perspectives. Ann N Y Acad Sci 1261:64–71
Ortega E, García JJ, Bote ME, Martín-Cordero L, Escalante Y, Saavedra JM, Northoff H, Giraldo E (2009a) Exercise in fibromyalgia and related inflammatory disorders: known effects and unknown chances. Exerc Immunol Rev 15:42–65
Ortega E, Hinchado MD, Martín-Cordero L, Asea A (2009b) The effect of stress-inducible extracellular Hsp72 on human neutrophil chemotaxis: a role during acute intense exercise. Stress 12(3):240–249
Perruccio AV, Chandran V, Power JD, Kapoor M, Mahomed NN, Gandhi R (2017) Systemic inflammation and painful joint burden in osteoarthritis: a matter of sex? Osteoarthr Cartil 25(1):53–59
Pozo M, Carretero MI, Maraver F, Pozo E, Gómez I, Armijo F, Martín-Rubí JA (2013) Composition and physico-chemical properties of peloids used in Spanish spas: a comparative study. Appl Clay Sci 83–84:270–279
Sarsan A, Akkaya N, Ozgen M, Yildiz N, Atalay NS, Ardic F (2012) Comparing the efficacy of mature mud pack and hot pack treatments for knee osteoarthritis. J Back Musculoskelet Rehabil 25(3):193–199
Scanzello C, Goldring S (2012) The role of synovitis in osteoarthritis pathogenesis. Bone 51:249–257
Scanzello CR, Plaas A, Crow MK (2008) Innate immune system activation in osteoarthritis: is osteoarthritis a chronic wound? Curr Opin Rheumatol 20(5):565–572
Scott J, Huskisson EC (1976) Graphic representation of pain. Pain 2(2):175–184
Sohn D, Sokolove J, Sharpe O, Erhart J, Chandra P, Lahey L, Lindstrom TM, Hwang I, Boyer KA, Andriacchi TP, Robinson WH (2012) Plasma proteins present in osteoarthritic synovial fluid can stimulate cytokine production via toll-like receptor 4. Arthritis Res Ther 14(1):R7
Sokolove J, Lepus CM (2013) Role of inflammation in the pathogenesis of osteoarthritis: latest findings and interpretations. Ther Adv Musculoskelet Dis 5(2):77–94
Svetlova MS, Vezikova NN, Romashina VV, Sobanina TS, Pavlov IL, Chepliukova IA (2009) Content of C-reactive protein, interleukin-1, interleukin-6 and interleukin-1 receptor antagonist in the blood of patients with early osteoarthrosis of the knee joints. Ter Arkh 81(6):52–56
Tenti S, Cheleschi S, Galeazzi M, Fioravanti A (2015) Spa therapy: can be a valid option for treating knee osteoarthritis? Int J Biometeorol 59(8):1133–1343
The EuroQol Group (1990) EuroQol—a new facility for the measurement of health-related quality of life. Health Policy 16(3):199–208
Verhagen A, Bierma-Zeinstra S, Lambeck J, Cardoso JR, de Bie R, Boers M, de Vet HC (2008) Balneotherapy for osteoarthritis. A Cochrane review. J Rheumatol 35(6):1118–1123
Acknowledgements
This work was partially supported by the Gobierno de Extremadura-FEDER (GR 15041, GR EE-14-0082-4). Gálvez I is recipient of a ‘Formación del Profesorado Universitario (FPU)’ pre-doctoral contract (FPU15/02395) from the Ministerio de Educación, Cultura y Deporte, Spain. Funding sources had no role in the study design, collection, analysis and interpretation of the data or the decision to submit the manuscript for publication.
We are grateful to the Facility of Bioscience Applied Techniques (STAB, University of Extremadura, Spain) and the spa centre ‘El Raposo’ for technical and human support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Ortega, E., Gálvez, I., Hinchado, M.D. et al. Anti-inflammatory effect as a mechanism of effectiveness underlying the clinical benefits of pelotherapy in osteoarthritis patients: regulation of the altered inflammatory and stress feedback response. Int J Biometeorol 61, 1777–1785 (2017). https://doi.org/10.1007/s00484-017-1361-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00484-017-1361-x