Abstract
Certain guidelines for the prevention of ventilator-associated pneumonia (VAP) recommend oral care with chlorhexidine, but none refer to the use of a toothbrush for oral hygiene. The role of toothbrush use has received scant attention. Thus, the objective of this study was to compare the incidence of VAP in critical care patients receiving oral care with and without manual brushing of the teeth. This was a randomized clinical trial developed in a 24-bed medical-surgical intensive care unit (ICU). Patients undergoing invasive mechanical ventilation for than 24 h were included. Patients were randomly assigned to receive oral care with or without toothbrushing. All patients received oral care with 0.12 % chlorhexidine digluconate. Tracheal aspirate samples were obtained during endotracheal intubation, then twice a week, and, finally, on extubation. There were no significant differences between the two groups of patients in the baseline characteristics. We found no statistically significant differences between the groups regarding the incidence of VAP (21 of 217 [9.7 %] with toothbrushing vs. 24 of 219 [11.0 %] without toothbrushing; odds ratio [OR] = 0.87, 95 % confidence interval [CI] = 0.469–1.615; p = 0.75). Adding manual toothbrushing to chlorhexidine oral care does not help to prevent VAP in critical care patients on mechanical ventilation.
Similar content being viewed by others
Introduction
Ventilator-associated pneumonia (VAP) remains a major cause of morbidity, mortality, and increased cost of care in critically ill patients [1–5]. Different pharmacological and non-pharmacological measures have been proposed for the prevention of VAP [6, 7].
Oral care with chlorhexidine solution has been found to reduce the risk of VAP, according to some meta-analyses [8–13]; however, the role of toothbrushing has received scant attention [14–18]. Some cohort studies have indicated that oral care with an antiseptic agent and toothbrushing could reduce the incidence of VAP compared with no oral care [14–16]; however, a limitation of these cohort studies is that it is not possible to discriminate the influence of toothbrushing per se, since the intervention group received both the antiseptic agent and toothbrushing. In a randomized study published by Pobo et al. in 2009, including a total of 147 patients, toothbrush use did not reduce the incidence of VAP [17]. In another randomized study published by Munro et al. in 2009, including 471 patients mechanically ventilated for a period of more than 24 h assigned into four groups (119 patients received chlorhexidine oral care, 113 patients toothbrushing, 116 patients toothbrushing and chlorhexidine oral care, and 123 patients usual care only), toothbrush use did not reduce the incidence of VAP [18].
Certain guidelines on the prevention of VAP make no reference to the question of oral care with chlorhexidine [19], while others recommend its early use [20–22]. However, none of them refer to the use of a toothbrush for oral care. Some care bundles proposed for VAP prevention in Europa and the USA did not refer to the use of a toothbrush for oral care [23, 24]. Oral care was perceived to be of a high priority in mechanically ventilated patients in a questionnaire survey in 59 European intensive care units (ICUs) and it was found that oral cavity cleaning was considered to be difficult by 68 %, 63 % of cases have insufficient supplies, and about 37 % are under the impression that oral health worsens despite their efforts to keep the mouth healthy [25]. Given the potential benefit of toothbrushing and the absence of evidence, we designed this study.
We tested the hypothesis that adding toothbrush use to oral care with 0.12 % chlorhexidine could reduce the incidence of VAP, by reducing the presence of endogenous oropharyngeal flora. The study was designed to compare the incidence of VAP using oral care with and without brushing the teeth with a manual brush in patients undergoing invasive mechanical ventilation for more than 24 h. The new aspects of our randomized controlled trial in comparison with the previous randomized controlled trials are the larger sample size and the surveillance of throat flora to explore the effect of toothbrushing on endogenous VAP.
Methods
Study design
A randomized clinical trial was performed in a 24-bed medical-surgical ICU of the Hospital Universitario de Canarias (Tenerife, Spain), a 650-bed tertiary hospital, from 1 August 2010 to 25 August 2011. The study was approved by the Institutional Review Board and informed consent from the patients or from their legal guardians was obtained.
Patients
Consecutive patients undergoing invasive mechanical ventilation were randomly assigned to one of two oral care groups, using a list of random numbers generated with Excel software (Microsoft, Seattle, WA), to discriminate the benefit of toothbrushing in VAP incidence. In group A, the oral cleansing was performed with 0.12 % chlorhexidine-impregnated gauze and oral cavity injection only; in group B, the oral cleansing was performed with 0.12 % chlorhexidine-impregnated gauze and oral cavity injection, followed by manual brushing of the teeth with a brush impregnated with 0.12 % chlorhexidine.
Exclusion criteria were as follows: edentulous, age < 18 years, pregnancy, human immunodeficiency virus (HIV), white blood cell (WBC) count <1,000 cells/mm3, solid or hematological tumor, immunosuppressive therapy, and mechanical ventilation duration less than 24 h.
Oral care groups
In both groups, nurses performed oral cleansing every 8 h, as follows: first, the endotracheal cuff pressure was tested and oropharyngeal secretions were aspirated, then gauze impregnated with 20 mL of 0.12 % chlorhexidine digluconate was used to cleanse the teeth, tongue, and mucosal surfaces, followed by the injection of 10 mL of 0.12 % chlorhexidine digluconate into the oral cavity, and, finally, after 30 s, the oropharyngeal area was suctioned.
In the interventional group, after the application of oral chlorhexidine in the same way, the nurses used a manual toothbrush to brush the patient’s teeth (tooth by tooth, on the anterior and posterior surfaces), the gum line, and the tongue for a period of 90 s.
Measures to prevent VAP
Before the study was started, the following measures to prevent VAP in both groups of patients were established: no routine change of ventilator circuits, tracheal suction when necessary by an open system, respiratory secretion suctioning performed with barrier measures (hand washing, use of gloves and face masks), semi-recumbent body position to maintain a head elevation ≥30º, periodic verification every 8 h of intracuff pressure to maintain a pressure of 25 cm H2O, endotracheal tube with a separate dorsal lumen for subglottic secretion drainage (performed intermittently during 1-h periods with a 10-mL syringe), nasogastric tube and periodic verification of the residual gastric volume every 6 h (residual gastric volume lower than 250 cc was considered as acceptable), and no selective digestive decontamination. The sedation drugs were adjusted to achieve a level 3–4 Ramsay score [26]. The weaning of mechanical ventilation started by pressure support ventilation at a pressure support of 20 cm of water, after which, the pressure support is progressively reduced to zero and, finally, a T-tube circuit is used. The types of endotracheal tubes used were as follows: (1) TaperGuard Evac™ (Mallinckrodt, Athlone, Ireland), which incorporates a dorsal separate lumen ending in the subglottic area for subglottic secretion drainage and (2) Hi-Lo™ (Mallinckrodt, Athlone, Ireland), which does not incorporate the lumen for subglottic secretion drainage. Several months after the start of the study, we followed a national project for the control of the VAP, but this did not affect the measures that had been selected at the beginning of the study.
Microbiological vigilance
Tracheal aspirate samples were obtained during endotracheal intubation, then twice a week, and, finally, on extubation. A throat swab was taken at admission to the ICU, twice a week thereafter, and at discharge from the ICU, for the study of bacterial flora to classify pneumonia as being of endogenous or exogenous origin.
Definitions
The diagnosis of pneumonia was based on the fulfillment of all the following criteria: (a) new onset of bronchial purulent sputum, (b) body temperature >38 °C or <35.5 °C, (c) white blood cell count >10,000/mm3 or <4,000/mm3, (d) chest radiograph showing new or progressive infiltrates, (e) significant quantitative culture of respiratory secretions by tracheal aspirate (>106 cfu/mL).
Pneumonia was considered as VAP when it was diagnosed during mechanical ventilation and was not present at the time of initiating mechanical ventilation.
VAP was considered as being of early onset when it was diagnosed during the first 4 days of mechanical ventilation, and as late onset when diagnosed after 4 days of mechanical ventilation.
Cases of VAP were classified as endogenous or exogenous according to throat flora analysis [27]: VAP was considered as primary endogenous when caused by microorganisms already present in the patient’s oropharyngeal flora on admission to the ICU and as secondary endogenous when caused by microorganisms not found on admission but detected in the patient’s oropharyngeal flora during ICU stay. VAP was considered as exogenous when it was caused by microorganisms that were never identified in the patient’s oropharyngeal flora.
The diagnosis of VAP was made by an expert panel, blinded to group assignment. The information about the type of oral care (with or without toothbrushing) was removed before the experts read the patient charts. The medical experts in infection control who diagnosed VAP were María Lecuona and María José Ramos.
Variables recorded
The following variables were recorded for each patient: sex, age, diagnosis group, Acute Physiology and Chronic Health Evaluation (APACHE) II score [28], duration of mechanical ventilation, antibiotics prior to VAP onset, use of paralytic agents, tracheotomy, re-intubation, enteral nutrition, duration of ICU stay, and mortality. In addition, the following variables were recorded for each VAP: Predisposition, Insult, Response and Organ dysfunction (PIRO) score for VAP [29], Sepsis-related Organ Failure Assessment (SOFA) score [30], chest X-ray quadrants, and pressure of arterial oxygen/fraction inspired oxygen (PaO2/FiO2).
Statistical analysis
In the year before the study, of the patients undergoing mechanical ventilation for more than 24 h who received conventional oral cleansing without toothbrush use, 15 % developed VAP. For a power of 80 % and an alpha error of 5 %, we required 218 patients per group in order to detect a reduction in VAP incidence from 15 % (using conventional care without toothbrushing) to 7.5 % (using oral cleansing with manual toothbrushing).
Quantitative variables, described as mean ± standard deviation, were compared with Student’s t-test. Qualitative variables, described as percentages, were compared with the Chi-squared test or with Fisher’s exact test, as appropriate.
The proportion of VAP between groups was compared with the Kruskal–Wallis test for single-order classification. The incidence density of VAP (number of events/days of mechanical ventilation) between groups was compared using Poisson regression analysis. The probability of remaining VAP-free was represented using the Kaplan–Meier method and comparison between the two groups was performed with the log-rank test.
Differences with a p-value of less than 0.05 were considered to be statistically significant. For statistical analyses, we used SPSS 14.0.1 (SPSS Inc., Chicago, IL, USA) and StatXact 5.0.3 (Cyrus Mehta and Nitin Patel, Cambridge, MA, USA).
Results
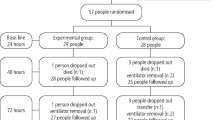
The present study included 436 ICU patients on mechanical ventilation divided into two groups, one with 217 patients receiving oral care with toothbrushing and the other with 219 patients receiving oral care without toothbrush use. There were no significant differences between the two groups of patients regarding age, sex, diagnosis groups, APACHE II score, use of antibiotics, paralytic agents, reintubation, tracheotomy, enteral nutrition, and duration of mechanical ventilation (Table 1).
We found no statistically significant differences in the incidence of VAP between the groups (21 of 217 [9.7 %] with toothbrush use vs. 24 of 219 [11.0 %] without; odds ratio [OR] = 0.87, 95 % confidence interval [CI] = 0.469–1.615; p = 0.75) (Table 2). We also found no statistically significant differences between the two groups in terms of the microorganisms responsible for VAP, early-onset or late-onset VAP, and endogenous or exogenous VAP (Table 2).
We have not found any significant differences in VAP incidence between patients with and without toothbrushing among surgical patients (OR = 0.85, 95 % CI = 0.300–2.440), medical patients (OR = 0.93, 95 % CI = 0.350–2.460), and trauma patients (OR = 0.72, 95 % CI = 0.053–7.430) (Table 3). We also found no significant differences in VAP incidence between patients with and without toothbrushing among patients with antibiotic exposure (OR = 0.88, 95 % CI = 0.440–1.740) and without antibiotic exposure (OR = 0.90, 95 % CI = 0.011–72.993) (Table 3). We have not found significant differences between survivor patients with (n = 155) and without (n = 150) toothbrushing in terms of antibiotic-free days (7.43 ± 14.84 vs. 8.39 ± 16.83; p = 0.59), mechanical ventilation-free days (4.03 ± 3.22 vs. 4.42 ± 3.93; p = 0.34), and days of ICU stay (14.58 ± 17.19 vs. 15.55 ± 18.87; p = 0.64).
We have not found significant differences in the PIRO score, SOFA score, chest X-ray quadrants, and PaO2/FIO2 between the VAP diagnosed in each group of patients (Table 4).
VAP was found in 21 patients during 1,993 days of mechanical ventilation in the toothbrush group and in 24 patients during 2,175 days of mechanical ventilation in the group without toothbrush use. Poisson regression analysis showed no difference in the VAP incidence density between patients with and without toothbrushing (10.54 vs. 11.03 VAPs/1,000 days of mechanical ventilation; OR = 0.95, 95 CI = 0.53–1.72; p = 0.88). We have not found significant differences in regard to the patients free of VAP according to the Kaplan–Meier curve (Fig. 1).
Table 5 provides a description of the microorganism responsible for VAP and of the microorganism classifying VAP according to throat flora (primary endogenous, secondary endogenous, and exogenous) and according to onset (early- or late-onset).
Discussion
No significant differences in the incidence of VAP were found according to the use or non-use of toothbrushing in oral care. To our knowledge, this is the largest randomized trial studying this issue for the prevention of VAP.
Poor oral hygiene is one of the main factors leading to the proliferation and accumulation of dental plaque and subsequent colonization by pathogens [31]. The number of positive dental plaque cultures in patients admitted to the ICU is high, being between 23 and 60 % [32–34]. The amount of dental plaque and its positive culture by aerobic pathogens increases during ICU stay [32]. Most VAP is caused by microorganisms that are present in the oropharynx [35–39], and aspiration of pathogenic bacteria from the oropharynx is, therefore, the main pathophysiologic mechanism involved. According to some studies, respiratory pathogens isolated from the lung are often genetically indistinguishable from strains of the same species isolated from dental plaque [33, 40] and the tongue [41]. A systematic review found an association between periodontal disease and nosocomial infections [42]. Therefore, it seems logical that improved oral care may reduce the risk of nosocomial respiratory infection. In this regard, some authors have investigated the utility of oral decontamination by the application of antibiotic or antiseptic agents [8–13, 43, 44]. However, the role of toothbrushing has scarcely been studied [14–18].
With respect to oral decontamination with chlorhexidine in critically ill patients, some studies have reported a reduction in positive dental plaque cultures [43, 44], and some meta-analyses have found a reduction in VAP [8–13]. A meta-analysis published by Chan et al. in 2007, including 11 randomized trials and 3,242 patients, evaluated the effect of oral decontamination on the incidence of VAP [11]. In the subanalysis of four studies with 1,098 patients, the oral application of antibiotics (gentamicin, colistin, vancomycin, polymyxin B, or iseganan) did not result in a significantly reduced incidence of VAP (relative risk [RR] = 0.69, 95 % CI = 0.41–1.18). In the subanalysis of seven studies with 2,144 patients, the oral application of antiseptics (chlorhexidine 0.12–2 % or 10 % povidone iodine) did significantly reduce the incidence of VAP (RR = 0.56, 95 % CI = 0.39–0.81). A later meta-analysis published by Carvajal et al. in 2010, including ten randomized trials and 2,978 patients, focused on the efficacy of oral chlorhexidine in reducing the rate of VAP. A lower risk of VAP was found in the intervention group compared to controls (OR = 0.56, 95 % CI = 0.44–0.73) [12]. However, no reduction was found in the mortality, duration of mechanical ventilation, or ICU stay with the oral application of chlorhexidine. In addition, there was a lack of uniformity in the concentration (0.12–2 %) and application interval (2–4 times/day) of chlorhexidine
With respect to toothbrushing, its use has been found to reduce the risk of caries and periodontal disease in the general population [45–47]. Controversy exists about the efficacy of toothbrushing to reduce dental plaque in mechanically ventilated patients. One randomized study with 46 patients found lower dental plaque in the toothbrushing group [48]; however, in another randomized study with 146 patients, there were no significant differences in the dental plaque associated with toothbrush use or not [49].
Some cohort studies [14–16] have found that oral care using an antiseptic agent and toothbrushing reduced the incidence of VAP compared with no oral care. The limitations of these studies are that a historical cohort was compared with a prospective cohort, and that it is not possible to discriminate the influence of toothbrushing alone, since oral care in the intervention group involved the simultaneous use of both an antiseptic agent and toothbrushing.
In the randomized study published by Pobo et al. in 2009, which included a total of 147 patients receiving oral care with 0.12 % chlorhexidine every 8 h, the patients were assigned to receive toothbrushing or not [17]. The authors found no significant differences between groups regarding the incidence of VAP (20.3 % vs. 24.7 %, p = 0.55), microbiologically documented VAP (RR = 0.84, 95 % CI = 0.41–1.73), mortality, days without antibiotics, duration of mechanical ventilation, or length of hospital or ICU stay. However, this study was stopped after enrolling only 37 % of the expected number of patients (the sample size calculated was 400 patients), because the estimated time to recruit the number of patients needed to achieve a statistically significant difference was too long for a single-center study.
In the randomized study published by Munro et al. in 2009, including 471 patients mechanically ventilated for more than 24 h assigned into four groups (119 patients received chlorhexidine oral care, 113 patients toothbrushing, 116 patients toothbrushing and chlorhexidine oral care, and 123 patients usual care only), toothbrush use did not reduce the incidence of VAP [18]. In this study, we analyzed VAP at days 3, 5, and 7, and the authors found that chlorhexidine, but not toothbrushing, reduced early VAP. However, in this study, data on VAP incidence beyond 7 days of mechanical ventilation were not reported.
The results of our study are consistent with those reported by Pobo et al. [17] and Munro et al. [18], since neither found significant differences in the incidence of VAP according to toothbrush use or not. Our study differed from that by Pobo et al. in that they used an electric toothbrush and we used a manual toothbrush; in the study by Munro et al., the toothbrush used was not described. In addition, our study involved a greater sample size (436 patients) to compare the efficacy of adding toothbrushing to chlorhexidine oral care in the VAP incidence reduction, compared with 147 patients in the study by Pobo et al. [17] and 235 patients in the study by Munro et al. [18].
The process of toothbrushing can give rise to certain complications, such as the appearance of oral bleeding in patients with severe coagulopathy due to the application of greater force than when applied by the patient. In addition, the action of toothbrushing could facilitate the accidental removal of the endotracheal tube, with the need for reintubation, and this fact has been associated with VAP in some studies [50–52].
These observations suggest that the oral cavity may be an important reservoir of pathogens that could cause VAP and that oral care could reduce the risk of VAP. For the moment, there is evidence that oral care with chlorhexidine reduces VAP; however, there is no evidence that toothbrushing provides an additional benefit.
Currently, there is insufficient evidence in the literature on oral hygiene to be able to recommend what measures should be performed in critically ill patients. Possible lines of research could include the chemical and mechanical aspects of oral care. With respect to the chemical agent, we could explore the type of agent used, the concentration, and the frequency of application. In relation to mechanical care, the use of a toothbrush and dental floss could be studied. And with respect to the brush, we could explore the effect of the brush type (manual or electric) and the frequency of application.
Our study has certain limitations. First, we did not perform an assessment of dental plaque, caries, and periodontal state. Another limitation is that we did not compare the incidence of complications, such as oral wounds and bleeding, and the rate of accidental removal of the endotracheal tube. Another point is that the study was performed within a single ICU, and the results may, therefore, not be applicable elsewhere. A further limitation is that the VAP diagnostic procedure was not invasive and we used only tracheal aspirate samples; however, a recent randomized clinical trial found no significant differences in clinical outcomes and the administration of antibiotics using a diagnostic strategy for VAP based on the quantitative culture of bronchoalveolar lavage fluid and the non-quantitative culture of endotracheal aspirate [53]. Another limitation was the blinding process; since toothbrushing or not is visually apparent, the study could not be blinded for the attending nurses and physicians; however, the kind of oral care was blinded for the expert panel who established the diagnosis of VAP. Another limitation is that we have not checked compliance with the VAP prevention measures and oral care; the problem of deficiency in the adherence to the proposed measures has been demonstrated in previous studies [54–56]. Finally, in our study, we found an absolute difference in the VAP rate between groups of 1.3 % and a 10 % reduction in VAP incidence, and the statistical power was 12 %. According to these previous results, we needed to include 13,576 patients in our study so as to reach an 80 % statistical power.
Conclusion
Adding manual toothbrushing to chlorhexidine oral care does not help to prevent ventilator-associated pneumonia (VAP) among critical care patients on mechanical ventilation.
References
Fagon JY, Chastre J, Hance AJ, Montravers P, Novara A, Gibert C (1993) Nosocomial pneumonia in ventilated patients: a cohort study evaluating attributable mortality and hospital stay. Am J Med 94:281–288
Heyland DK, Cook DJ, Griffith L, Keenan SP, Brun-Buisson C (1999) The attributable morbidity and mortality of ventilator-associated pneumonia in the critically ill patient. The Canadian Critical Trials Group. Am J Respir Crit Care Med 159:1249–1256
Bercault N, Boulain T (2001) Mortality rate attributable to ventilator-associated nosocomial pneumonia in an adult intensive care unit: a prospective case–control study. Crit Care Med 29:2303–2309
Warren DK, Shukla SJ, Olsen MA, Kollef MH, Hollenbeak CS, Cox MJ, Cohen MM, Fraser VJ (2003) Outcome and attributable cost of ventilator-associated pneumonia among intensive care unit patients in a suburban medical center. Crit Care Med 31:1312–1317
Safdar N, Dezfulian C, Collard HR, Saint S (2005) Clinical and economic consequences of ventilator-associated pneumonia: a systematic review. Crit Care Med 33:2184–2193
Lorente L, Blot S, Rello J (2007) Evidence on measures for the prevention of ventilator-associated pneumonia. Eur Respir J 30:1193–1207
Lorente L, Blot S, Rello J (2010) New issues and controversies in the prevention of ventilator-associated pneumonia. Am J Respir Crit Care Med 182:870–876
Pineda LA, Saliba RG, El Solh AA (2006) Effect of oral decontamination with chlorhexidine on the incidence of nosocomial pneumonia: a meta-analysis. Crit Care 10:R35
Kola A, Gastmeier P (2007) Efficacy of oral chlorhexidine in preventing lower respiratory tract infections. Meta-analysis of randomized controlled trials. J Hosp Infect 66:207–216
Chlebicki MP, Safdar N (2007) Topical chlorhexidine for prevention of ventilator-associated pneumonia: a meta-analysis. Crit Care Med 35:595–602
Chan EY, Ruest A, Meade MO, Cook DJ (2007) Oral decontamination for prevention of pneumonia in mechanically ventilated adults: systematic review and meta-analysis. BMJ 334:889
Carvajal C, Pobo A, Díaz E, Lisboa T, Llauradó M, Rello J (2010) Oral hygiene with chlorhexidine on the prevention of ventilator-associated pneumonia in intubated patients: a systematic review of randomized clinical trials. Med Clin (Barc) 135:491–497
Labeau SO, Van de Vyver K, Brusselaers N, Vogelaers D, Blot SI (2011) Prevention of ventilator-associated pneumonia with oral antiseptics: a systematic review and meta-analysis. Lancet Infect Dis 11:845–854
Mori H, Hirasawa H, Oda S, Shiga H, Matsuda K, Nakamura M (2006) Oral care reduces incidence of ventilator-associated pneumonia in ICU populations. Intensive Care Med 32:230–236
Hutchins K, Karras G, Erwin J, Sullivan KL (2009) Ventilator-associated pneumonia and oral care: a successful quality improvement project. Am J Infect Control 37:590–597
Sona CS, Zack JE, Schallom ME, McSweeney M, McMullen K, Thomas J, Coopersmith CM, Boyle WA, Buchman TG, Mazuski JE, Schuerer DJ (2009) The impact of a simple, low-cost oral care protocol on ventilator-associated pneumonia rates in a surgical intensive care unit. J Intensive Care Med 24:54–62
Pobo A, Lisboa T, Rodriguez A, Sole R, Magret M, Trefler S, Gómez F, Rello J; RASPALL Study Investigators (2009) A randomized trial of dental brushing for preventing ventilator-associated pneumonia. Chest 136:433–439
Munro CL, Grap MJ, Jones DJ, McClish DK, Sessler CN (2009) Chlorhexidine, toothbrushing, and preventing ventilator-associated pneumonia in critically ill adults. Am J Crit Care 18:428–437
Masterton RG, Galloway A, French G, Street M, Armstrong J, Brown E, Cleverley J, Dilworth P, Fry C, Gascoigne AD, Knox A, Nathwani D, Spencer R, Wilcox M (2008) Guidelines for the management of hospital-acquired pneumonia in the UK: report of the working party on hospital-acquired pneumonia of the British Society for Antimicrobial Chemotherapy. J Antimicrob Chemother 62:5–34
Muscedere J, Dodek P, Keenan S, Fowler R, Cook D, Heyland D; VAP Guidelines Committee and the Canadian Critical Care Trials Group (2008) Comprehensive evidence-based clinical practice guidelines for ventilator-associated pneumonia: prevention. J Crit Care 23:126–137
Coffin SE, Klompas M, Classen D, Arias KM, Podgorny K, Anderson DJ, Burstin H, Calfee DP, Dubberke ER, Fraser V, Gerding DN, Griffin FA, Gross P, Kaye KS, Lo E, Marschall J, Mermel LA, Nicolle L, Pegues DA, Perl TM, Saint S, Salgado CD, Weinstein RA, Wise R, Yokoe DS (2008) Practice recommendation of Society for Healthcare Epidemiology of America/Infectious Diseases Society of America (SHEA/IDSA). Strategies to prevent ventilator-associated pneumonia in acute care hospitals. Infect Control Hosp Epidemiol 29(Suppl 1):S31–S40
Torres A, Ewig S, Lode H, Carlet J; European HAP working group (2009) Defining, treating and preventing hospital acquired pneumonia: European perspective. Intensive Care Med 35:9–29
Rello J, Lode H, Cornaglia G, Masterton R; VAP Care Bundle Contributors (2010) A European care bundle for prevention of ventilator-associated pneumonia. Intensive Care Med 36:773–780
Berwick DM, Calkins DR, McCannon CJ, Hackbarth AD (2006) The 100,000 lives campaign: setting a goal and a deadline for improving health care quality. JAMA 295:324–327
Rello J, Koulenti D, Blot S, Sierra R, Diaz E, De Waele JJ, Macor A, Agbaht K, Rodriguez A (2007) Oral care practices in intensive care units: a survey of 59 European ICUs. Intensive Care Med 33:1066–1070
Ramsay MA, Savege TM, Simpson BR, Goodwin R (1974) Controlled sedation with alphaxalone–alphadolone. Br Med J 2:656–659
van Saene HKF, Damjanovic V, Murray AE, de la Cal MA (1996) How to classify infections in intensive care units—the carrier state, a criterion whose time has come? J Hosp Infect 33:1–12
Knaus WA, Draper EA, Wagner DP, Zimmerman JE (1985) APACHE II: a severity of disease classification system. Crit Care Med 13:818–829
Lisboa T, Diaz E, Sa-Borges M, Socias A, Sole-Violan J, Rodríguez A, Rello J (2008) The ventilator-associated pneumonia PIRO score: a tool for predicting ICU mortality and health-care resources use in ventilator-associated pneumonia. Chest 134:1208–1216
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonça A, Bruining H, Reinhart CK, Suter PM, Thijs LG (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 22:707–710
Gibbons RJ (1989) Bacterial adhesion to oral tissues: a model for infectious diseases. J Dent Res 68:750–760
Fourrier F, Duvivier B, Boutigny H, Roussel-Delvallez M, Chopin C (1998) Colonization of dental plaque: a source of nosocomial infections in intensive care unit patients. Crit Care Med 26:301–308
El-Solh AA, Pietrantoni C, Bhat A, Okada M, Zambon J, Aquilina A, Berbary E (2004) Colonization of dental plaques: a reservoir of respiratory pathogens for hospital-acquired pneumonia in institutionalized elders. Chest 126:1575–1582
Scannapieco FA, Stewart EM, Mylotte JM (1992) Colonization of dental plaque by respiratory pathogens in medical intensive care patients. Crit Care Med 20:740–745
Lorente L, Lecuona M, Málaga J, Revert C, Mora ML, Sierra A (2003) Bacterial filters in respiratory circuits: an unnecessary cost? Crit Care Med 31:2126–2130
Lorente L, Lecuona M, Galván R, Ramos MJ, Mora ML, Sierra A (2004) Periodically changing ventilator circuits is not necessary to prevent ventilator-associated pneumonia when a heat and moisture exchanger is used. Infect Control Hosp Epidemiol 25:1077–1082
Lorente L, Lecuona M, Martín MM, García C, Mora ML, Sierra A (2005) Ventilator-associated pneumonia using a closed versus an open tracheal suction system. Crit Care Med 33:115–119
Lorente L, Lecuona M, Jiménez A, Mora ML, Sierra A (2006) Tracheal suction by closed system without daily change versus open system. Intensive Care Med 32:538–544
Lorente L, Lecuona M, Jiménez A, Mora ML, Sierra A (2006) Ventilator-associated pneumonia using a heated humidifier or a heat and moisture exchanger: a randomized controlled trial [ISRCTN88724583]. Crit Care 10:R116
Heo SM, Haase EM, Lesse AJ, Gill SR, Scannapieco FA (2008) Genetic relationships between respiratory pathogens isolated from dental plaque and bronchoalveolar lavage fluid from patients in the intensive care unit undergoing mechanical ventilation. Clin Infect Dis 47:1562–1570
Bahrani-Mougeot FK, Paster BJ, Coleman S, Barbuto S, Brennan MT, Noll J, Kennedy T, Fox PC, Lockhart PB (2007) Molecular analysis of oral and respiratory bacterial species associated with ventilator-associated pneumonia. J Clin Microbiol 45:1588–1593
Scannapieco FA, Bush RB, Paju S (2003) Associations between periodontal disease and risk for nosocomial bacterial pneumonia and chronic obstructive pulmonary disease. A systematic review. Ann Periodontol 8:54–69
Fourrier F, Dubois D, Pronnier P, Herbecq P, Leroy O, Desmettre T, Pottier-Cau E, Boutigny H, Di Pompéo C, Durocher A, Roussel-Delvallez M; PIRAD Study Group (2005) Effect of gingival and dental plaque antiseptic decontamination on nosocomial infections acquired in the intensive care unit: a double-blind placebo-controlled multicenter study. Crit Care Med 33:1728–1735
Scannapieco FA, Yu J, Raghavendran K, Vacanti A, Owens SI, Wood K, Mylotte JM (2009) A randomized trial of chlorhexidine gluconate on oral bacterial pathogens in mechanically ventilated patients. Crit Care 13:R117
Attin T, Hornecker E (2005) Tooth brushing and oral health: how frequently and when should tooth brushing be performed? Oral Health Prev Dent 3:135–140
Löe H (2000) Oral hygiene in the prevention of caries and periodontal disease. Int Dent J 50:129–139
Bellini HT, Arneberg P, von der Fehr FR (1981) Oral hygiene and caries. A review. Acta Odontol Scand 39:257–265
Needleman IG, Hirsch NP, Leemans M, Moles DR, Wilson M, Ready DR, Ismail S, Ciric L, Shaw MJ, Smith M, Garner A, Wilson S (2011) Randomized controlled trial of toothbrushing to reduce ventilator-associated pneumonia pathogens and dental plaque in a critical care unit. J Clin Periodontol 38:246–252
Roca Biosca A, Anguera Saperas L, García Grau N, Rubio Rico L, Velasco Guillén MC (2011) Prevention of mechanical ventilator-associated pneumonia: a comparison of two different oral hygiene methods. Enferm Intensiva 22:104–111
Torres A, Gatell JM, Aznar E, el-Ebiary M, Puig de la Bellacasa J, González J, Ferrer M, Rodriguez-Roisin R (1995) Re-intubation increases the risk of nosocomial pneumonia in patients needing mechanical ventilation. Am J Respir Crit Care Med 152:137–141
Leal-Noval SR, Márquez-Vacaro JA, García-Curiel A, Camacho-Laraña P, Rincón-Ferrari MD, Ordoñez-Fernández A, Flores-Cordero JM, Loscertales-Abril J (2000) Nosocomial pneumonia in patients undergoing heart surgery. Crit Care Med 28:935–940
Kollef MH, von Harz B, Prentice D, Shapiro SD, Silver P, St John R, Trovillion E (1997) Patient transport from intensive care increases the risk of developing ventilator-associated pneumonia. Chest 112:765–773
Canadian Critical Care Trials Group (2006) A randomized trial of diagnostic techniques for ventilator-associated pneumonia. N Engl J Med 355:2619–2630
Rello J, Lorente C, Bodí M, Diaz E, Ricart M, Kollef MH (2002) Why do physicians not follow evidence-based guidelines for preventing ventilator-associated pneumonia?: a survey based on the opinions of an international panel of intensivists. Chest 122:656–661
McGuckin M, Waterman R, Govednik J (2009) Hand hygiene compliance rates in the United States—a one-year multicenter collaboration using product/volume usage measurement and feedback. Am J Med Qual 24:205–213
Rello J, Afonso E, Lisboa T, Ricart M, Balsera B, Rovira A, Valles J, Diaz E; FADO Project Investigators (2012) A care bundle approach for prevention of ventilator-associated pneumonia. Clin Microbiol Infect (accepted for publication). doi:10.1111/j.1469-0691.2012.03808.x
Acknowledgments
This study was supported, in part, by a grant from Instituto de Salud Carlos III (I3SNS-INT-11-063) (Madrid, Spain).
Competing interests
The authors do not have any financial interests to disclose.
Funding
Hospital funding.
Trial registration
Identifier
NCT01477099
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lorente, L., Lecuona, M., Jiménez, A. et al. Ventilator-associated pneumonia with or without toothbrushing: a randomized controlled trial. Eur J Clin Microbiol Infect Dis 31, 2621–2629 (2012). https://doi.org/10.1007/s10096-012-1605-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10096-012-1605-y