Abstract
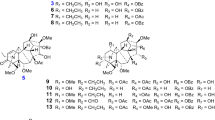
Two new oxaphenalenone dimers, talaromycesone A (1) and talaromycesone B (2), and a new isopentenyl xanthenone, talaroxanthenone (3), together with six known diphenyl ether derivatives, e.g., Δ1′,3′,-1′-dehydroxypenicillide (4), 1′,2′-dehydropenicillide (5), vermixocin A (6), vermixocin B (7), 3′-methoxy-1′2′-dehydropenicillide (8), and AS-186c (9), were isolated from the culture broth and mycelia of a marine fungus Talaromyces sp. strain LF458. Compound 2 represents the first example of 1-nor oxaphenalenone dimer carbon skeleton. All isolated compounds were subjected to bioactivity assays. Compounds 1, 2, and 9 exhibited potent antibacterial activities with IC50 3.70, 17.36, and 1.34 μM, respectively, against human pathogenic Staphylococcus strains. Compounds 1, 3, and 9 displayed potent acetylcholinesterase inhibitory activities with IC50 7.49, 1.61, and 2.60 μM, respectively. Interestingly, phosphodiesterase PDE-4B2 was inhibited by compounds 3 (IC50 7.25 μM) and 9 (IC50 2.63 μM).




Similar content being viewed by others
References
Dethoup T, Manoch L, Kijjoa A, Nascimento MSJ, Puaparoj P, Silva AMS, Eaton G, Herz W (2006) Bacillisporins D and E, new oxaphenalenone dimers from Talaromyces bacillisporus. Planta Med 72:957–960
Domsch K-H, Gams W, Anderson T-H (2007) Compendium of soil fungi. IHW-Verlag, Eching
Elsebai MF, Kehraus S, Lindequist U, Sasse F, Shaaban S, Cütscho M, Josten M, Sahl HG, König GM (2011) Antimicrobial phenalenone derivatives from the marine-derived fungus Coniothyrium cereal. Org Biomol Chem 9:802–808
Elsebai MF, Saleem M, Tejesvi MV, Kajula M, Mattila S, Mehiri M, Turpeinen A, Pirttilä AM (2014) Fungal phenalenones: chemistry, biology, biosynthesis and phylogeny. Nat Prod Rep 31:628–645
Guo Z, Shao C, She Z, Cai X, Liu F, Vrijimoed LLP, Lin Y (2007) 1H and 13C NMR assignments for two oxaphenalenones bacillosporin C and D from the mangrove endophytic fungus SBE-14. Magn Reson Chem 45:439–441
Haefliger W, Hauser D (1973) Isolierung und strukturaufklärung von 11-desacetoxy-wortmannin. Helv Chim Acta 56:2901–2904
Jansen N, Ohlendorf B, Erhard A, Imhoff JF (2013) Helicusin E, isochromophilone X and isochromophilone XI: new chloroazaphilones produced by the fungus Bartalinia robillardoides strain LF550. Mar Drugs 11:800–816
Kawamoto K, Yamazaki H, Ohte MR, Uchida R, Tomoda H (2011) Production of monapinones by fermentation of the dinapinone-producing fungus Penicillium pinophilum FKI-3864 in a seawater-containing medium. J Antibiot 64:503–508
Kawamura H, Kaneko T, Koshino H, Esum UJ, Sugawara F (2000) Penicillides from Penicillium sp. isolated from Taxus cuspidata. Nat Prod Lett 14:477–484
Kuroda K, Morishita Y, Saito Y, Ikuina Y, Ando K, Kawamoto I, Matsuda Y (1994) AS-186 compounds, new inhibitors of acyl-CoA: cholesterol acyltransferase from Penicillium asperosporum KY1635. J Antibiot 47:16–22
Lin Z, Zhu T, Fang Y, Gu Q, Zhu W (2008) Polyketides from Penicillium sp. JP-1, an endophytic fungus associated with the mangrove plant Aegiceras corniculatum. Phytochemistry 69:1273–1278
Murray AP, Faraoni MB, Castro MJ, Alza NP, Cavallaro V (2013) Natural AChE inhibitors from plants and their contribution to Alzheimer’s disease therapy. Curr Neuropharmacol 11:388–413
Nonaka K, Abe T, Iwatsuki M, Mori M, Yamamoto T, Shiomi K, Omura S, Masuma R (2011) Enhancement of metabolites productivity of Penicillium pinophilum FKI-5653, by co-culture with Trichoderma harzianum FKI-5655. J Antibiot 64:769–774
Ohlendorf B, Schulz D, Erhard A, Nagel K, Imhoff JF (2012) Geranylphenazinediol, an acetylcholinesterase inhibitor produced by a Streptomyces species. J Nat Prod 75:1400–1404
Page CP, Spina D (2012) Selective PDE inhibitors as novel treatments for respiratory diseases. Curr Opin Pharmacol 12:275–286
Prince M, Bryce R, Albanese E, Wimo A, Ribeiro W, Ferri CP (2013) The global prevalence of dementia: a systematic review and metaanalysis. Alzheimers and Dement 9:63–75
Proksa B, Uhrín D, Adamcová J, Fuska J (1992) Vermixocins A and B, two novel metabolites from Penicillium vermiculatum. J Antibiot 45:1268–1272
Ren J, Zhang F, Liu X, Li L, Liu G, Liu X, Che Y (2012) Neonectrolide A, a new oxaphenalenone spiroketal from the fungus Neonectria sp. Org Lett 14:6226–6229
Samson RA, Yilmaz N, Houbraken J, Spierenburg H, Seifert KA, Peterson SW, Varga J, Frisvad JC (2011) Phylogeny and nomenclature of the genus Talaromyces and taxa accommodated in Penicillium subgenus Biverticillium. Stud Mycol 70:159–183
Schulz D, Beese P, Ohlendorf B, Erhard A, Zinecker H, Dorador C, Imhoff JF (2011) Abenquines A–D: aminoquinone derivatives produced by streptomyces sp. strain DB634. J Antibiot 64:763–768
Seydametova E, Salihon J, Zainol N, Convey P (2012) Production of Lovastatin by Penicillium spp. soil microfungi. Int J Chem Eng Appl 3:337–339
Shope RE (1953) An antiviral substance from Penicillium funiculosum. I. Effect upon injection in mice with swine influenza virus and Columbia SK encephalomyelitis virus. J Exp Med 97:601–625
Silber J, Ohlendorf B, Labes A, Erhard A, Imhoff JF (2013) Calcarides A–E, antibacterial macrocyclic and linear polyesters from a Calcarisporium strain. Mar Drugs 11:3309–3323
Tomoda H, Tabata N, Masuma R, Si SY, Omura S (1998) Erabulenols, inhibitors of cholesteryl ester transfer protein produced by Penicillium sp. FO-5637. I. Production, isolation and biological properties. J Antibiot 51:618–623
van Reenen-Hoekstra ES, Frisvad JC, Samson RA, Stolk AC (1990) In: Samson RA, Pitt JI (eds) Modern concepts in Penicillium and Aspergillus classification. Plenum, New York
Wang M-H, Li X-M, Li C-S, Ji N-Y, Wang B-G (2013) Secondary metabolites from Penicillium pinophilum SD-272, a marine sediment-derived fungus. Mar Drugs 11:2230–2238
Wickerham LJ (1951) Taxonomy of yeasts. US Dept Tech Bull 29:1–56
Wiese J, Ohlendorf B, Blümel M, Schmaljohann R, Imhoff JF (2011) Phylogenetic identification of fungi isolated from the marine sponge Tethya aurantium and identification of their secondary metabolites. Mar Drugs 9:561–585
Witter DP, Chen Y, Rogel JK, Boldt GE, Wentworth P (2009) The natural products beauveriolide I and III: a new class of β-amyloid lowering compounds. ChemBioChem 10:1344–1347
Yamazaki M, Okuyama E (1980) Isolation and structures of oxaphenalenone dimers from Talaromyces bacillosporus. Chem Pharm Bull 28:3649–3655
Zhang Y, Li X-M, Shang Z, Li C-S, Ji N-Y, Wang B-G (2012) Meroterpenoid and diphenyl ether derivatives from Penicillium sp. MA-37, a fungus isolated from marine mangrove rhizospheric soil. J Nat Prod 75:1888–1895
Acknowledgments
The authors thank Dr. Stefan Steffens and Dr. Karsten Schaumann for providing strain LF458. We gratefully thank G. Kohlmeyer-Yilmaz, M. Höftmann, as well as Prof. F. Sönnichsen (Otto Diels Institute of Organic Chemistry, Christian-Albrechts-Universität zu Kiel, Germany) for running and processing NMR experiments. The authors are also grateful to A. Erhard, R. Koppe, and A. Schneider for excellent technical assistance. Thanks to NSFC (81273386) for supporting Bin Wu.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 2,266 kb)
Rights and permissions
About this article
Cite this article
Wu, B., Ohlendorf, B., Oesker, V. et al. Acetylcholinesterase Inhibitors from a Marine Fungus Talaromyces sp. Strain LF458. Mar Biotechnol 17, 110–119 (2015). https://doi.org/10.1007/s10126-014-9599-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10126-014-9599-3