Abstract
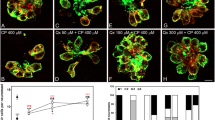
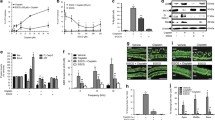
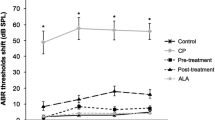
Cisplatin, a chemotherapeutic drug that is widely used to treat various cancers, promotes ototoxicity at higher doses. In this study, the effect of epicatechin (EC) on cisplatin-induced hair cell death was investigated in a cochlear organ of Corti-derived cell line, HEI-OC1, and in vivo in zebrafish. Cisplatin promoted apoptosis and altered mitochondrial membrane potential (MMP) in HEI-OC1 cells. EC inhibited cisplatin-induced apoptosis and intracellular reactive oxygen species (ROS) generation. Labeling of zebrafish lateral line hair cells by the fluorescent dye YO-PRO1 was lost upon exposure to cisplatin, and EC protected against this cisplatin-induced loss of labeling in a dose-dependent manner. Scanning and transmission electron micrographs showed that treatment with EC protected against cisplatin-induced loss of kinocilium and stereocilia in zebrafish neuromasts. These results suggest that EC prevents cisplatin-induced ototoxicity by blocking ROS generation and by preventing changes in MMP.








Similar content being viewed by others
References
Hartmann JT, Lipp HP (2003) Toxicity of platinum compounds. Expert Opin Pharmacother 4:889–901. doi:10.1517/14656566.4.6.889
Boulikas T, Vougiouka M (2004) Recent clinical trials using cisplatin, carboplatin and their combination chemotherapy drugs. Oncol Rep 11:559–595 (Review)
Estrem SA, Babin RW, Ryu JH, Moore KC (1981) Cis-diamminedichloroplatinum(II) ototoxicity in the guinea pig. Otolaryngol Head Neck Surg 89:638–645
Laurell G, Bagger-Sjoback D (1991) Degeneration of the organ of Corti following intravenous administration of cisplatin. Acta Otolaryngol 111:891–898. doi:10.3109/00016489109138427
Tange RA, Vuzevski VD (1984) Changes in the stria vascularis of the guinea pig due to cis-platinum. Arch Otorhinolaryngol 239:41–47. doi:10.1007/BF00454261
Tsukasaki N, Whitworth CA, Rybak LP (2000) Acute changes in cochlear potentials due to cisplatin. Hear Res 149:189–198. doi:10.1016/S0378-5955(00)00182-9
Gabaizadeh R, Staecker H, Liu W et al (1997) Protection of both auditory hair cells and auditory neurons from cisplatin induced damage. Acta Otolaryngol 117:232–238. doi:10.3109/00016489709117778
Campbell KC, Rybak LP, Meech RP, Hughes L (1996) d-methionine provides excellent protection from cisplatin ototoxicity in the rat. Hear Res 102:90–98. doi:10.1016/S0378-5955(96)00152-9
Rybak LP, Husain K, Evenson L, Morris C, Whitworth C, Somani SM (1997) Protection by 4-methylthiobenzoic acid against cisplatin-induced ototoxicity: antioxidant system. Pharmacol Toxicol 81:173–179
Cardinaal RM, de Groot JC, Huizing EH, Veldman JE, Smoorenburg GF (2000) Cisplatin-induced ototoxicity: morphological evidence of spontaneous outer hair cell recovery in albino guinea pigs? Hear Res 144:147–156. doi:10.1016/S0378-5955(00)00060-5
Li G, Sha SH, Zotova E, Arezzo J, Van de Water T, Schacht J (2002) Salicylate protects hearing and kidney function from cisplatin toxicity without compromising its oncolytic action. Lab Invest 82:585–596. doi:10.1038/labinvest.3780453
Rybak LP, Ravi R, Somani SM (1995) Mechanism of protection by diethyldithiocarbamate against cisplatin ototoxicity: antioxidant system. Fundam Appl Toxicol 26:293–300. doi:10.1006/faat.1995.1100
Schweitzer VG, Dolan DF, Abrams GE, Davidson T, Snyder R (1986) Amelioration of cisplatin-induced ototoxicity by fosfomycin. Laryngoscope 96:948–958
Li G, Frenz DA, Brahmblatt S et al (2001) Round window membrane delivery of l-methionine provides protection from cisplatin ototoxicity without compromising chemotherapeutic efficacy. Neurotoxicology 22:163–176. doi:10.1016/S0161-813X(00)00010-3
Rybak LP, Whitworth C, Somani S (1999) Application of antioxidants and other agents to prevent cisplatin ototoxicity. Laryngoscope 109:1740–1744. doi:10.1097/00005537-199911000-00003
Feghali JG, Liu W, Van de Water TR (2001) l-n-Acetyl-cysteine protection against cisplatin-induced auditory neuronal and hair cell toxicity. Laryngoscope 111:1147–1155. doi:10.1097/00005537-200107000-00005
Campbell KC, Larsen DL, Meech RP, Rybak LP, Hughes LF (2003) Glutathione ester but not glutathione protects against cisplatin-induced ototoxicity in a rat model. J Am Acad Audiol 14:124–133. doi:10.3766/jaaa.14.3.2
Erba D, Riso P, Bordoni A, Foti P, Biagi PL, Testolin G (2005) Effectiveness of moderate green tea consumption on antioxidative status and plasma lipid profile in humans. J Nutr Biochem 16:144–149. doi:10.1016/j.jnutbio.2004.11.006
Khan N, Mukhtar H (2007) Tea polyphenols for health promotion. Life Sci 81:519–533. doi:10.1016/j.lfs.2007.06.011
Kalinec GM, Webster P, Lim DJ, Kalinec F (2003) A cochlear cell line as an in vitro system for drug ototoxicity screening. Audiol Neurootol 8:177–189. doi:10.1159/000071059
Raible DW, Kruse GJ (2000) Organization of the lateral line system in embryonic zebrafish. J Comp Neurol 421:189–198. doi :10.1002/(SICI)1096-9861(20000529)421:2≤189::AID-CNE5≥3.0.CO;2-K
Kopke RD, Liu W, Gabaizadeh R et al (1997) Use of organotypic cultures of Corti’s organ to study the protective effects of antioxidant molecules on cisplatin-induced damage of auditory hair cells. Am J Otol 18:559–571
Alam SA, Ikeda K, Oshima T et al (2000) Cisplatin-induced apoptotic cell death in Mongolian gerbil cochlea. Hear Res 141:28–38. doi:10.1016/S0378-5955(99)00211-7
Watanabe KC, Jinnouchi K, Hess A, Michel O, Baba S, Yagi T (2002) Carboplatin induces less apoptosis in the cochlea of guinea pigs than cisplatin. Chemotherapy 48:82–87. doi:10.1159/000057667
Huang H, Zhu L, Reid BR, Drobny GP, Hopkins PB (1995) Solution structure of a cisplatin-induced DNA interstrand cross-link. Science 270:1842–1845. doi:10.1126/science.270.5243.1842
Jordan P, Carmo-Fonseca M (2000) Molecular mechanisms involved in cisplatin cytotoxicity. Cell Mol Life Sci 57:1229–1235. doi:10.1007/PL00000762
Trimmer EE, Essigmann JM (1999) Cisplatin. Essays Biochem 34:191–211
Clerici WJ, Hensley K, DiMartino DL, Butterfield DA (1996) Direct detection of ototoxicant-induced reactive oxygen species generation in cochlear explants. Hear Res 98:116–124. doi:10.1016/0378-5955(96)00075-5
Evans P, Halliwell B (1999) Free radicals and hearing. Cause, consequence, and criteria. Ann NY Acad Sci 884:19–40. doi:10.1111/j.1749-6632.1999.tb08633.x
Fisher DE (1994) Apoptosis in cancer therapy: crossing the threshold. Cell 78:539–542. doi:10.1016/0092-8674(94)90518-5
Henkels KM, Turchi JJ (1997) Induction of apoptosis in cisplatin-sensitive and -resistant human ovarian cancer cell lines. Cancer Res 57:4488–4492
Henkels KM, Turchi JJ (1999) Cisplatin-induced apoptosis proceeds by caspase-3-dependent and -independent pathways in cisplatin-resistant and -sensitive human ovarian cancer cell lines. Cancer Res 59:3077–3083
Dehne N, Lautermann J, Petrat F, Rauen U, de Groot H (2001) Cisplatin ototoxicity: involvement of iron and enhanced formation of superoxide anion radicals. Toxicol Appl Pharmacol 174:27–34. doi:10.1006/taap. 2001.9171
Ravi R, Somani SM, Rybak LP (1995) Mechanism of cisplatin ototoxicity: antioxidant system. Pharmacol Toxicol 76:386–394
Rybak LP, Husain K, Morris C, Whitworth C, Somani S (2000) Effect of protective agents against cisplatin ototoxicity. Am J Otol 21:513–520
Weijl NI, Hopman GD, Wipkink-Bakker A et al (1998) Cisplatin combination chemotherapy induces a fall in plasma antioxidants of cancer patients. Ann Oncol 9:1331–1337. doi:10.1023/A:1008407014084
Cheng AG, Cunningham LL, Rubel EW (2005) Mechanisms of hair cell death and protection. Curr Opin Otolaryngol Head Neck Surg 13:343–348. doi:10.1097/01.moo.0000186799.45377.63
Lazebnik YA, Kaufmann SH, Desnoyers S, Poirier GG, Earnshaw WC (1994) Cleavage of poly(ADP-ribose) polymerase by a proteinase with properties like ICE. Nature 371:346–347. doi:10.1038/371346a0
Cheng EH, Kirsch DG, Clem RJ et al (1997) Conversion of Bcl-2 to a Bax-like death effector by caspases. Science 278:1966–1968. doi:10.1126/science.278.5345.1966
Kothakota S, Azuma T, Reinhard C et al (1997) Caspase-3-generated fragment of gelsolin: effector of morphological change in apoptosis. Science 278:294–298. doi:10.1126/science.278.5336.294
Kirsch DG, Doseff A, Chau BN et al (1999) Caspase-3-dependent cleavage of Bcl-2 promotes release of cytochrome c. J Biol Chem 274:21155–21161. doi:10.1074/jbc.274.30.21155
Ton C, Parng C (2005) The use of zebrafish for assessing ototoxic and otoprotective agents. Hear Res 208:79–88. doi:10.1016/j.heares.2005.05.005
Ou HC, Raible DW, Rubel EW (2007) Cisplatin-induced hair cell loss in zebrafish (Danio rerio) lateral line. Hear Res 233:46–53. doi:10.1016/j.heares.2007.07.003
Masuda M, Suzui M, Weinstein IB (2001) Effects of epigallocatechin-3-gallate on growth, epidermal growth factor receptor signaling pathways, gene expression, and chemosensitivity in human head and neck squamous cell carcinomas cell lines. Clin Cancer Res 7:4220–4229
Chan MM, Soprano KJ, Weinstein K, Fong D (2006) Epigallocatechin-3-gallate delivers hydrogen peroxide to induce death of ovarian cancer cells and enhances their cisplatin susceptibility. J Cell Physiol 207:389–396. doi:10.1002/jcp. 20569
Acknowledgements
This research was supported by CCRB through the “GRRC” Project of the Gyeonggi Provincial Government, Republic of Korea, and the Korea Research Foundation Grant funded by the Korean government (MOEHRD; KRF-2006-331-E00260).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, CH., Kang, S.U., Pyun, J. et al. Epicatechin protects auditory cells against cisplatin-induced death. Apoptosis 13, 1184–1194 (2008). https://doi.org/10.1007/s10495-008-0242-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10495-008-0242-5