Abstract
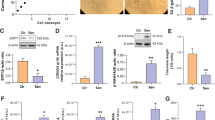
Cellular senescence is now recognized as an important mechanism of tumor suppression, and the accumulation of senescent cells may contribute to the aging of various human tissues. Alterations of the cellular energy metabolism are considered key events in tumorigenesis and are also known to play an important role for aging processes in lower eukaryotic model systems. In this study, we addressed senescence-associated changes in the energy metabolism of human endothelial cells, using the HUVEC model of in vitro senescence. We observed a drastic reduction in cellular ATP levels in senescent endothelial cells. Although consumption of glucose and production of lactate significantly increased in senescent cells, no correlation was found between both metabolite conversion rates, neither in young endothelial cells nor in the senescent cells, which indicates that glycolysis is not the main energy source in HUVEC. On the other hand, glutamine consumption was increased in senescent HUVEC and inhibition of glutaminolysis by DON, a specific inhibitor of glutaminase, led to a significant reduction in the proliferative capacity of both early passage and late passage cells. Moreover, inhibition of glutaminase activity induced a senescent-like phenotype in young HUVEC within two passages. Together, the data indicate that glutaminolysis is an important energy source in endothelial cells and that alterations in this pathway play a role in endothelial cell senescence.






Similar content being viewed by others
Abbreviations
- ATP:
-
Adenosine triphosphate
- DON:
-
6-diazo-5-oxo-l-norleucine
- GAPDH:
-
Glyceraldehyde 3-phosphate dehydrogenase
- HDF:
-
Human diploid fibroblasts
- HUVEC:
-
Human umbilical vein endothelial cell
- LDH:
-
Lactate dehydrogenase
- PDL:
-
Population doubling
- PK:
-
Pyruvate kinase
References
Aledo JC (2004) Glutamine breakdown in rapidly dividing cells: waste or investment? Bioessays 26:778–785
Anderson RM, Weindruch R (2006) Calorie restriction: progress during mid-2005-mid-2006. Exp Gerontol 41:1247–1249
Bensaad K, Tsuruta A, Selak MA, Vidal MN, Nakano K, Bartrons R, Gottlieb E, Vousden KH (2006) TIGAR, a p53-inducible regulator of glycolysis and apoptosis. Cell 126:107–120
Bergmeyer HU (ed) (1974) Methoden der enzymatischen Analyse. Verlag Chemie, Weinheim
Braig M, Lee S, Loddenkemper C, Rudolph C, Peters AH, Schlegelberger B, Stein H, Dorken B, Jenuwein T, Schmitt CA (2005) Oncogene-induced senescence as an initial barrier in lymphoma development. Nature 436:660–665
Campisi J (1998) The role of cellular senescence in skin aging. J Investig Dermatol Symp Proc 3:1–5
Collado M, Gil J, Efeyan A, Guerra C, Schuhmacher AJ, Barradas M, Benguria A, Zaballos A, Flores JM, Barbacid M, Beach D, Serrano M (2005) Tumour biology: senescence in premalignant tumours. Nature 436:642
Dierick JF, Eliaers F, Remacle J, Raes M, Fey SJ, Larsen PM, Toussaint O (2002) Stress-induced premature senescence and replicative senescence are different phenotypes, proteomic evidence. Biochem Pharmacol 64:1011–1017
Dimri GP, Lee X, Basile G, Acosta M, Scott G, Roskelley C, Medrano EE, Linskens M, Rubelj I, Pereira-Smith O, Smith J (1995) A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc Natl Acad Sci USA 92:9363–9367
Harley CB (1997) Human ageing and telomeres. Ciba Found Symp 211:129–139
Hutter E, Renner K, Pfister G, Stockl P, Jansen-Durr P, Gnaiger E (2004) Senescence-associated changes in respiration and oxidative phosphorylation in primary human fibroblasts. Biochem J 380:919–928
Hutter E, Unterluggauer H, Garedew A, Jansen-Durr P, Gnaiger E (2006) High-resolution respirometry – a modern tool in aging research. Exp Gerontol 41:103–109
Jacobs HT (2003) The mitochondrial theory of aging: dead or alive? Aging Cell 2:11–17
Jazwinski SM (2000) Metabolic control and ageing. Trends Genet 16:506–511
Jeyapalan JC, Ferreira M, Sedivy JM, Herbig U (2007) Accumulation of senescent cells in mitotic tissue of aging primates. Mech Ageing Dev 128:36–44
Kim JW, Dang CV (2006) Cancer’s molecular sweet tooth and the Warburg effect. Cancer Res 66:8927–8930
Krishnamurthy J, Torrice C, Ramsey MR, Kovalev GI, Al-Regaiey K, Su L, Sharpless NE (2004) Ink4 a/Arf expression is a biomarker of aging. J Clin Invest 114:1299–1307
Kujoth GC, Hiona A, Pugh TD, Someya S, Panzer K, Wohlgemuth SE, Hofer T, Seo AY, Sullivan R, Jobling WA, Morrow JD, Van Remmen H, Sedivy JM, Yamasoba T, Tanokura M, Weindruch R, Leeuwenburgh C, Prolla TA (2005) Mitochondrial DNA mutations, oxidative stress, and apoptosis in mammalian aging. Science 309:481–484
Lane MA, Black A, Handy A, Tilmont EM, Ingram DK, Roth GS (2001) Caloric restriction in primates. Ann NY Acad Sci 928: 287–295
Lin SJ, Defossez PA, Guarente L (2000) Requirement of NAD and SIR2 for life-span extension by calorie restriction in Saccharomyces cerevisiae. Science 289:2126–2128
Lobo C, Ruiz-Bellido MA, Aledo JC, Marquez J, Nunez De Castro I, Alonso FJ (2000) Inhibition of glutaminase expression by antisense mRNA decreases growth and tumourigenicity of tumour cells. Biochem J (348 Pt 2): 257–261
Lowe SW, Cepero E, Evan G (2004) Intrinsic tumour suppression. Nature 432:307–315
Matoba S, Kang JG, Patino WD, Wragg A, Boehm M, Gavrilova O, Hurley PJ, Bunz F, Hwang PM (2006) p53 regulates mitochondrial respiration. Science 312:1650–1653
Matsuno T, Goto I (1992) Glutaminase and glutamine synthetase activities in human cirrhotic liver and hepatocellular carcinoma. Cancer Res 52:1192–1194
Mazurek S, Michel A, Eigenbrodt E (1997) Effect of extracellular AMP on cell proliferation and metabolism of breast cancer cell lines with high and low glycolytic rates. J Biol Chem 272:4941–4952
Mazurek S, Boschek CB, Hugo F, Eigenbrodt E (2005) Pyruvate kinase type M2 and its role in tumor growth and spreading. Semin Cancer Biol 15:300–308
Melk A, Schmidt BM, Takeuchi O, Sawitzki B, Rayner DC, Halloran PF (2004) Expression of p16 INK4a and other cell cycle regulator and senescence associated genes in aging human kidney. Kidney Int 65:510–520
Michaloglou C, Vredeveld LC, Soengas MS, Denoyelle C, Kuilman T, van der Horst CM, Majoor DM, Shay JW, Mooi WJ, Peeper DS (2005) BRAFE600-associated senescence-like cell cycle arrest of human naevi. Nature 436:720–724
Minamino T, Miyauchi H, Yoshida T, Ishida Y, Yoshida H, Komuro I (2002) Endothelial cell senescence in human atherosclerosis: role of telomere in endothelial dysfunction. Circulation 105:1541–1544
Miquel J (1991) An integrated theory of aging as the result of mitochondrial-DNA mutation in differentiated cells. Arch Gerontol Geriatr 12:99–117
Muck C, Micutkova L, Zwerschke W, Jansen-Durr P (2008) Role of Insulin-like growth factor binding protein-3 in human umbilical vein endothelial cell senescence. Rejuvenation Res 11(1):1–6
Ovejera AA, Houchens DP, Catane R, Sheridan MA, Muggia FM (1979) Efficacy of 6-diazo-5-oxo-L-norleucine and N-[N-gamma-glutamyl-6-diazo-5-oxo-norleucinyl]-6-diazo-5-oxo-norleucine against experimental tumors in conventional and nude mice. Cancer Res 39:3220–3224
Passos JF, Saretzki G, Ahmed S, Nelson G, Richter T, Peters H, Wappler I, Birket MJ, Harold G, Schaeuble K, Birch-Machin MA, Kirkwood TB, von Zglinicki T (2007) Mitochondrial dysfunction accounts for the stochastic heterogeneity in telomere-dependent senescence. PLoS Biol 5:e110
Perez-Gomez C, Campos-Sandoval JA, Alonso FJ, Segura JA, Manzanares E, Ruiz-Sanchez P, Gonzalez ME, Marquez J, Mates JM (2005) Co-expression of glutaminase K and L isoenzymes in human tumour cells. Biochem J 386:535–542
Phanvijhitsiri K, Musch MW, Ropeleski MJ, Chang EB (2006) Heat induction of heat shock protein 25 requires cellular glutamine in intestinal epithelial cells. Am J Physiol Cell Physiol 291: C290–9
Reitzer LJ, Wice BM, Kennell D (1979) Evidence that glutamine, not sugar, is the major energy source for cultured HeLa cells. J Biol Chem 254:2669–2676
Ressler S, Bartkova J, Niederegger H, Bartek J, Scharffetter-Kochanek K, Jansen-Durr P, Wlaschek M (2006) p16 INK4A is a robust in vivo biomarker of cellular aging in human skin. Aging Cell 5:379–389
Rossignol R, Gilkerson R, Aggeler R, Yamagata K, Remington SJ, Capaldi RA (2004) Energy substrate modulates mitochondrial structure and oxidative capacity in cancer cells. Cancer Res 64:985–993
Serrano M, Blasco MA (2001) Putting the stress on senescence. Curr Opin Cell Biol 13:748–753
Shapiro RA, Clark VM, Curthoys NP (1979) Inactivation of rat renal phosphate-dependent glutaminase with 6-diazo-5-oxo-L-norleucine. Evidence for interaction at the glutamine binding site. J Biol Chem 254:2835–2838
Shaw RJ (2006) Glucose metabolism and cancer. Curr Opin Cell Biol 18:598–608
Spittler A, Oehler R, Goetzinger P, Holzer S, Reissner CM, Leutmezer F, Rath V, Wrba F, Fuegger R, Boltz-Nitulescu G, Roth E (1997) Low glutamine concentrations induce phenotypical and functional differentiation of U937 myelomonocytic cells. J Nutr 127:2151–2157
Stockl P, Hutter E, Zwerschke W, Jansen-Durr P (2006) Sustained inhibition of oxidative phosphorylation impairs cell proliferation and induces premature senescence in human fibroblasts. Exp Gerontol 41:674–682
Trifunovic A, Wredenberg A, Falkenberg M, Spelbrink JN, Rovio AT, Bruder CE, Bohlooly YM, Gidlof S, Oldfors A, Wibom R, Tornell J, Jacobs HT, Larsson NG (2004) Premature ageing in mice expressing defective mitochondrial DNA polymerase. Nature 429:417–423
Trifunovic A, Hansson A, Wredenberg A, Rovio AT, Dufour E, Khvorostov I, Spelbrink JN, Wibom R, Jacobs HT, Larsson NG (2005) Somatic mtDNA mutations cause aging phenotypes without affecting reactive oxygen species production. Proc Natl Acad Sci USA 102:17993–17998
Turowski GA, Rashid Z, Hong F, Madri JA, Basson MD (1994) Glutamine modulates phenotype and stimulates proliferation in human colon cancer cell lines. Cancer Res 54:5974–5980
Vasile E, Tomita Y, Brown LF, Kocher O, Dvorak HF (2001) Differential expression of thymosin beta-10 by early passage and senescent vascular endothelium is modulated by VPF/VEGF: evidence for senescent endothelial cells in vivo at sites of atherosclerosis. Faseb J 15:458–466
Wagner M, Hampel B, Bernhard D, Hala M, Zwerschke W, Jansen-Durr P (2001) Replicative senescence of human endothelial cells in vitro involves G1 arrest, polyploidization and senescence-associated apoptosis. Exp Gerontol 36:1327–1347
Wang W, Yang X, Lopez De Silanes I, Carling D, Gorospe M (2003) Increased AMP: ATP ratio and AMP-activated kinase activity during cellular senescence linked to reduced HuR function. J Biol Chem 278:27016–27023
Zwerschke W, Mazurek S, Stockl P, Hutter E, Eigenbrodt E, Jansen-Durr P (2003) Metabolic analysis of senescent human fibroblasts reveals a role for adenosine monophosphate in cellular senescence. Biochem J 376:403–411
Acknowledgements
We thank Bianca Kulik for excellent technical assistance. We thank Lutfan Lazuardi for help with statistical calculations. This work was supported by the Austrian Science Funds (FWF NRN S93), the European Union (CELLAGE project QLK6-CT-2001-00616) and the Austrian Ministry of Science and Traffic.
Author information
Authors and Affiliations
Corresponding author
Additional information
Prof. Dr. Erich Eigenbrodt deceased in 2004.
Rights and permissions
About this article
Cite this article
Unterluggauer, H., Mazurek, S., Lener, B. et al. Premature senescence of human endothelial cells induced by inhibition of glutaminase. Biogerontology 9, 247–259 (2008). https://doi.org/10.1007/s10522-008-9134-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10522-008-9134-x