Abstract
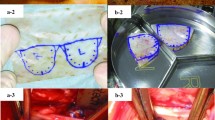
Glutaraldehyde preservation is the gold standard for cardiovascular biological prosthesis. However, secondary calcifications and the absence of tissue growth remain major limitations. Our study assessed in vitro and in vivo the biocompatibility of human (fascia lata, pericardium) and porcine tissues (pericardium, peritoneum) treated with a physicochemical procedure for decellularization and non-conventional pathogens inactivation. Biopsies were performed before and after treatment to assess decellularization (HE/Dapi staining/DNA quantification/MHC I/alpha gal immunostaining) and mechanical integrity. Forty-five rats received an abdominal aortic patch of native cryopreserved tissues (n = 20), treated tissues (n = 20) or glutaraldehyde-preserved bovine pericardium (GBP, control, n = 5). Grafts were explanted at 4 weeks and processed for HE/von Kossa staining and immunohistochemistries for lymphocytes (CD3)/macrophages (CD68) histomorphometry. 95% of decellularization was obtained for all tissues except for fascia lata (75%). Mechanical properties were slightly altered. In the in vivo model, a significant increase of CD3 and CD68 infiltrations was found in native and control implants in comparison with decellularized tissues (p < 0.05). Calcifications were found in 3 controls. Decellularized tissues were recolonized. GBP showed the most inflammatory response. This physicochemical treatment improves the biocompatibility of selected xeno/allogeneic tissues in comparison with their respective native cryopreserved tissues and with GBP. Incomplete decellularization is associated with a significantly higher inflammatory response. Our treatment is a promising tool in the field of tissue decellularization and tissue banking.







Similar content being viewed by others
References
Badylak SF (2014) Decellularized allogeneic and xenogeneic tissue as a bioscaffold for regenerative medicine: factors that influence the host response. Ann Biomed Eng 42:1517–1527. doi:10.1007/s10439-013-0963-7
Badylak SF, Freytes DO, Gilbert TW (2009) Extracellular matrix as a biological scaffold material: structure and function. Acta Biomater 5:1–13. doi:10.1016/j.actbio.2008.09.013
Bonvini S, Albiero M, Ferretto L et al (2012) The peritoneum as a natural scaffold for vascular regeneration. PLoS ONE 7:e33557. doi:10.1371/journal.pone.0033557
Burkhart HM, Moody SA, Ensing GJ, Brown JW (1996) Ventricular septal aneurysm after atrioventricular septal repair with pericardium. Ann Thorac Surg 61:1838–1839. doi:10.1016/0003-4975(96)80207-4
Butera G, Aggoun Y, Bonnet D, Bonhoeffer P (2001) Aneurysmal dilation of a pericardial patch prepared with glutharaldehyde and used for closure of a ventricular septal defect. Ital Heart J Off J Ital Fed Cardiol 2:317–318
Cigliano A, Gandaglia A, Lepedda AJ et al (2012) Fine structure of glycosaminoglycans from fresh and decellularized porcine cardiac valves and pericardium. Biochem Res Int 2012:979351. doi:10.1155/2012/979351
Cornu O, Schubert T, Libouton X et al (2009) Particle size influence in an impaction bone grafting model. Comparison of fresh-frozen and freeze-dried allografts. J Biomech 42:2238–2242. doi:10.1016/j.jbiomech.2009.06.045
Dufrane D, Cornu O, Delloye C, Schneider YJ (2002) Physical and chemical processing for a human dura mater substitute. Biomaterials 23:2979–2988
Dufrane D, Marchal C, Cornu O et al (2003) Clinical application of a physically and chemically processed human substitute for dura mater. J Neurosurg 98:1198–1202. doi:10.3171/jns.2003.98.6.1198
Dufrane D, Mourad M, van Steenberghe M et al (2008) Regeneration of abdominal wall musculofascial defects by a human acellular collagen matrix. Biomaterials 29:2237–2248. doi:10.1016/j.biomaterials.2008.01.027
Fawzi-Grancher S, Goebbels RM, Bigare E et al (2009) Human tissue allograft processing: impact on in vitro and in vivo biocompatibility. J Mater Sci Mater Med 20:1709–1720. doi:10.1007/s10856-009-3726-0
Gilbert TW, Sellaro TL, Badylak SF (2006) Decellularization of tissues and organs. Biomaterials 27:3675–3683. doi:10.1016/j.biomaterials.2006.02.014
Hopkins RA, Lofland GK, Marshall J et al (2014) Pulmonary arterioplasty with decellularized allogeneic patches. Ann Thorac Surg 97:1407–1412. doi:10.1016/j.athoracsur.2013.12.005
Iablonskii P, Cebotari S, Tudorache I et al (2015) Tissue-engineered mitral valve: morphology and biomechanics. Interact CardioVasc Thorac Surg 20:712–719. doi:10.1093/icvts/ivv039 (discussion 719)
Kneib C, von Glehn CQC, Costa FDA et al (2012) Evaluation of humoral immune response to donor HLA after implantation of cellularized versus decellularized human heart valve allografts. Tissue Antigens 80:165–174. doi:10.1111/j.1399-0039.2012.01885.x
Lafosse A, Desmet C, Aouassar N et al (2015) Autologous adipose stromal cells seeded onto a human collagen matrix for dermal regeneration in chronic wounds: clinical proof of concept. Plast Reconstr Surg 136:279–295. doi:10.1097/PRS.0000000000001437
López Marco A, Nawaytou O, Von Oppell UO (2013) Late aortic aneurysm after supravalvar aortic stenosis repaired with autologous pericardial patch. Ann Thorac Surg 95:346–348. doi:10.1016/j.athoracsur.2012.05.090
Mirsadraee S, Wilcox HE, Watterson KG et al (2007) Biocompatibility of acellular human pericardium. J Surg Res 143:407–414. doi:10.1016/j.jss.2007.01.026
Neethling WML, Strange G, Firth L, Smit FE (2013) Evaluation of a tissue-engineered bovine pericardial patch in paediatric patients with congenital cardiac anomalies: initial experience with the ADAPT-treated CardioCel(R) patch. Interact CardioVasc Thorac Surg 17:698–702. doi:10.1093/icvts/ivt268
Neumann A, Sarikouch S, Breymann T et al (2014) Early systemic cellular immune response in children and young adults receiving decellularized fresh allografts for pulmonary valve replacement. Tissue Eng Part A 20:1003–1011. doi:10.1089/ten.TEA.2013.0316
Papadimitriou J, Tsiftsis D, Papadimitriou K et al (1976) Long-term results from use of fascia lata as arterial graft. Am J Surg 132:15–18
Saleeb SF, Newburger JW, Geva T et al (2014) Accelerated degeneration of a bovine pericardial bioprosthetic aortic valve in children and young adults. Circulation 130:51–60. doi:10.1161/CIRCULATIONAHA.114.009835
Tudorache I, Calistru A, Baraki H et al (2013) Orthotopic replacement of aortic heart valves with tissue-engineered grafts. Tissue Eng Part A 19:1686–1694. doi:10.1089/ten.TEA.2012.0074
Tudorache I, Horke A, Cebotari S et al (2016) Decellularized aortic homografts for aortic valve and aorta ascendens replacement†. Eur J Cardio-Thorac Surg Off J Eur Assoc Cardio-Thorac Surg. doi:10.1093/ejcts/ezw013
Umashankar PR, Mohanan PV, Kumari TV (2012) Glutaraldehyde treatment elicits toxic response compared to decellularization in bovine pericardium. Toxicol Int 19:51–58. doi:10.4103/0971-6580.94513
Vinci MC, Tessitore G, Castiglioni L et al (2013) Mechanical compliance and immunological compatibility of fixative-free decellularized/cryopreserved human pericardium. PLoS ONE 8:e64769. doi:10.1371/journal.pone.0064769
Wong ML, Griffiths LG (2014) Immunogenicity in xenogeneic scaffold generation: antigen removal vs. decellularization. Acta Biomater 10:1806–1816. doi:10.1016/j.actbio.2014.01.028
Yamashita K, Kazui T, Suzuki K et al (2007) Successful repair of ascending aortic pseudoaneurysm using autograft patch from fascia lata and saphenous vein. Gen Thorac Cardiovasc Surg 55:502–504. doi:10.1007/s11748-007-0174-y
Zaidi AH, Nathan M, Emani S et al (2014) Preliminary experience with porcine intestinal submucosa (CorMatrix) for valve reconstruction in congenital heart disease: histologic evaluation of explanted valves. J Thorac Cardiovasc Surg 148:2216–2214, 2225.e1. doi:10.1016/j.jtcvs.2014.02.081
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
van Steenberghe, M., Schubert, T., Guiot, Y. et al. Enhanced vascular biocompatibility of decellularized xeno-/allogeneic matrices in a rodent model. Cell Tissue Bank 18, 249–262 (2017). https://doi.org/10.1007/s10561-017-9610-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-017-9610-0