Abstract
1. The aim of this study was to validate the role of postconditioning, used 2 days after lethal ischemia, for protection of selectively vulnerable brain neurons against delayed neuronal death.
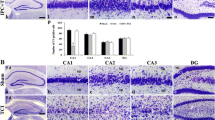
2. Eight, 10, or 15 min of transient forebrain ischemia in rat (four-vessel occlusion model) was used as initial lethal ischemia. Fluoro Jade B, the marker of neurodegeneration, and NeuN, a specific neuronal marker were used for visualization of changes 7 or 28 days after ischemia without and with delayed postconditioning.
3. Our results confirm that postconditioning if used at right time and with optimal intensity can prevent process of delayed neuronal death. At least three techniques, known as preconditioners, can be used as postconditioning: short ischemia, 3-nitropropionic acid and norepinephrine. A cardinal role for the prevention of death in selectively vulnerable neurons comprises synthesis of proteins during the first 5 h after postconditioning. Ten minutes of ischemia alone is lethal for 70% of pyramidal CA1 neurons in hippocampus. Injection of inhibitor of protein synthesis (Cycloheximide), if administered simultaneously with postconditioning, suppressed beneficial effect of postconditioning and resulted in 50% of CA1 neurons succumbing to neurodegeneration. Although, when Cycloheximide was injected 5 h after postconditioning, this treatment resulted in survival of 90% of CA1 neurons.
4. Though postconditioning significantly protects hippocampal CA1 neurons up to 10 min of ischemia, its efficacy at 15 min ischemia is exhausted. However, protective impact of postconditioning in less-sensitive neuronal populations (cortex and striatum) is very good after such a damaging insult like 15 min ischemia. This statement also means that up to 15 min of ischemia, postconditioning does not induce cumulation of injuries produced by the first and the second stress.



Similar content being viewed by others
REFERENCES
Araki, T., Kato, H., Inoue, T., and Kogure, K. (1990). Regional impairment of protein synthesis following brief cerebral ischemia in the gerbil. Acta Neuropathol. (Berl.) 79:501–505.
Barone, F. C., White, R. F., Spera, P. A., Ellison, J., Currie, R. W., Wang, X., and Feuerstein, G. Z. (1998). Ischemic preconditioning and brain tolerance: Temporal histological and functional outcomes, protein synthesis requirement, and interleukin-1 receptor antagonist and early gene expression. Stroke 29:1937–1950.
Blondeau, N., Widmann, C., Lazdunski, M., and Heurteaux, C. (2001). Activation of the nuclear factor-kappaB is a key event in brain tolerance. J. Neurosci. 21:4668–4677.
Blondeau, N., Widmann, C., Lazdunski, M., and Heurteaux, C. (2002). Polyunsaturated fatty acids induce ischemic and epileptic tolerance. Neuroscience 109:231–241.
Bodsch, W., Barbier, A., Oehmichen, M., Grosse Ophoff, B., and Hossmann, K. A. (1986). Recovery of monkey brain after prolonged ischemia. II. Protein synthesis and morphological alterations. J. Cereb. Blood Flow Metab. 6:22–33.
Bordet, R., Deplanque, D., Maboudou, P., Puisieux, F., Pu, Q., Robin, E., Martin, A., Bastide, M., Leys, D., Lhermitte, M., and Dupuis, B. (2000). Increase in endogenous brain superoxide dismutase as a potential mechanism of lipopolysaccharide-induced brain ischemic tolerance. J. Cereb. Blood Flow Metab. 20:1190–1196.
Brambrink, T. M., Schneider, T., Noga, H., Astheimer, A., Gotz, T., Korner, T., Heimann, A., Welschof, T., and Kempski, O. (2000). Tolerance-inducing dose of 3-nitropropionic acid modulates bcl-2 and bax balance in the rat brain: A potential mechanism of chemical preconditioning. J. Cereb. Blood Flow Metab. 20:1425–1436.
Burda, J., Gottlieb, M., Vanicky, I., Chavko, M., and Marsala, J. (1995). Short-term postischemic hypoperfusion improves recovery of protein synthesis in the rat brain cortex. Mol. Chem. Neuropathol. 25:189–198.
Burda, J., Hrehorovská, M., Bonilla, L. G., Danielisova, V., Cizkova, D., Burda, R., Nemethova, M., Fando, J. L., and Salinas, M. (2003). Role of protein synthesis in the ischemic tolerance acquisition induced by transient forebrain ischemia in the rat. Neurochem. Res. 28:1213–1219.
Burda, J., Hrehorovská, M., Gottlieb, M., Danielisová, V., Nemethova, M., Garcia, L., Salinas, M., and Burda, R. (2005a). Evidence for a role of second pathophysiological stress in prevention of delayed neuronal death in the hippocampal CA1 region. Neurochem. Res. 30:1397–1405.
Burda, J., Hrehorovská, M., Gottlieb, M., Danielisová, V., Nemethova, M., Garcia, L., Salinas, M., and Burda, R. (2005b). Role of second patophysiologic stress in the prevention of the delayed neuronal death. Psychatrie 9:28–29.
Burda, J., Marsala, M., Radonak, J., and Marsala, J. (1991). Graded postischemic reoxygenation ameliorates inhibition of cerebral cortical protein synthesis in dogs. J. Cereb. Blood Flow Metab. 11:1001–1006.
Chen, Y., Ginis, I., and Hallenbeck, J. M. (2001). The protective effect of ceramide in immature rat brain hypoxia-ischemia involves up-regulation of bcl-2 and reduction of TUNEL-positive cells. J. Cereb. Blood Flow Metab. 21:34–40.
Chopp, M., Chen, H., and Ho, K.-L. (1989). Transient hyperthermia protects against subsequent forebrain ischemic damage in the rat. Neurology 39:1396–1398.
Danielisova, V., Nemethova, M., Gottlieb, M., and Burda, J. (2005). Changes of endogenous antioxidant enzymes during ischemic tolerance acquisition. Neurochem. Res. 30:559–565.
Dienel, G. A., Pulsinelli, W. A., and Duffy, T. E. (1980). Regional protein synthesis in rat brain following acute hemispheric ischemia. J. Neurochem. 35:1216–1226.
Fujiki, M., Kobayashi, H., Abe, T., and Kamida, T. (2003). Repetitive transcranial magnetic stimulation for protection against delayed neuronal death induced by transient ischemia. J. Neurosurg. 99:1063–1069.
Furuta, S., Ohta, S., Hatakeyama, T., Nakamura, K., and Sakaki, S. (1993). Recovery of protein synthesis in tolerance-induced hippocampal CA1 neurons after transient forebrain ischemia. Acta Neuropathol. (Berl.) 86:329–336.
Gabai, V. L., and Kabakov, A. E. (1993). Rise in heat-shock protein level confers tolerance to energy deprivation. FEBS Lett. 327:247–250.
Gage, A. T., and Stanton, P. K. (1996). Hypoxia triggers neuroprotective alterations in hippocampal gene expression via a heme-containing sensor. Brain Res. 719:172–178.
Glazier, S. S., O’Rourke, D. M., Graham, D. I., and Welsh, F. A. (1994). Induction of ischemic tolerance following brief focal ischemia in rat brain. J. Cereb. Blood Flow Metab. 14:545–553.
Heurteaux, C., Lauritzen, I., Widmann, C., and Lazdunski, M. (1995). Essential role of adenosine, adenosine A1 receptors, and ATP-sensitive K+ channels in cerebral ischemic preconditioning. Proc. Natl. Acad. Sci. U.S.A. 92:4666–4670.
Kakimoto, M., Kawaguchi, M., Sakamoto, T., Inoue, S., Furuya, H., Nakamura, M., and Konishi, N. (2003). Evaluation of rapid ischemic preconditioning in a rabbit model of spinal cord ischemia. Anesthesiology 99:1112–1117.
Kato, H., Kogure, K., Nakata, N., Araki, T., and Itoyama, Y. (1995). Facilitated recovery from postischemic suppression of protein synthesis in the gerbil brain with ischemic tolerance. Brain Res. Bull. 36:205–208.
Kato, H., Liu, Y., Araki, T., and Kogure, K. (1991). Temporal profile of the effects of pretreatment with brief cerebral ischemia on the neuronal damage following secondary ischemic insult in the gerbil: Cumulative damage and protective effects. Brain Res. 553:238–242.
Kawahara, N., Ruetzler, C. A., Mies, G., and Klatzo, I. (1999). Cortical spreading depression increases protein synthesis and upregulates basic fibroblast growth factor. Exp. Neurol. 158:27–36.
Kin, H., Zhao, Z. Q., Sun, H. Y., Wang, N. P., Corvera, J. S., Halkos, M. E., Kerendi, F., Guyton, R. A., and Vinten-Johansen, J. (2004). Postconditioning attenuates myocardial ischemia-reperfusion injury by inhibiting events in the early minutes of reperfusion. Cardiovasc. Res. 62:74–85.
Kirino, T., Tsujita, Y., and Tamura, A. (1991). Induced tolerance to ischemia in gerbil hippocampal neurons. J. Cereb. Blood Flow Metab. 11:299–307.
Kitagawa, K., Matsumoto, M., Kuwabara, K., Tagaya, M., Ohtsuki, T., Hata, R., Ueda, H., Handa, N., Kimura, K., and Kamada, T. (1991). ‘Ischemic tolerance’ phenomenon detected in various brain regions. Brain Res. 561:203–211.
Kitagawa, K., Matsumoto, M., Tagaya, M., Hata, R., Ueda, H., Niinobe, M., Handa, N., Fukunaga, R., Kimura, K., Mikoshiba, K., et al. (1990). ‘Ischemic tolerance’ phenomenon found in the brain. Brain Res. 528:21–24.
Liu, Y., Kato, H., Nakata, N., and Kogure, K. (1992). Protection of rat hippocampus against ischemic neuronal damage by pretreatment with sublethal ischemia. Brain Res. 586:121–124.
Liu, Y., Kato, H., Nakata, N., and Kogure, K. (1993). Temporal profile of heat shock protein 70 synthesis in ischemic tolerance induced by preconditioning ischemia in rat hippocampus. Neuroscience 56:921–927.
Matsushima, K., Hogan, M. J., and Hakim, A. M. (1996). Cortical spreading depression protects against subsequent focal cerebral ischemia in rats. J. Cereb. Blood Flow Metab. 16:221–226.
Meng, X., Cleveland, J. C., Jr., Rowland, R. T., Mitchell, M. B., Brown, J. M., Banerjee, A., and Harken, A. H. (1996). Norepinephrine-induced sustained myocardial adaptation to ischemia is dependent on alpha 1-adrenoceptors and protein synthesis. J. Mol. Cell. Cardiol. 28:2017–2025.
Minatoguchi, S., Uno, Y., Kariya, T., Arai, M., Wang, N., Hashimoto, K., Nishida, Y., Maruyama, R., Takemura, G., Fujiwara, T., and Fujiwara, H. (2003). Cross-talk among noradrenaline, adenosine and protein kinase C in the mechanisms of ischemic preconditioning in rabbits. J. Cardiovasc. Pharmacol. 2003:S39–S47.
Mizuno, S., Ishii, A., Murakami, Y., and Akagawa, H. (1997). Stress dose-dependent suppression of heat shock protein gene expression by inhibiting protein synthesis during heat shock treatment. Cell Struct. Funct. 22:7–13.
Murry, C. E., Jennings, R. B., and Reimer, K. A. (1986). Preconditioning with ischemia: A delay of lethal cell injury in ischemic myocardium. Circulation 74:1124–1136.
Nishi, S., Taki, W., Uemura, Y., Higashi, T., Kikuchi, H., Kudoh, H., Satoh, M., and Nagata, K. (1993). Ischemic tolerance due to the induction of HSP70 in a rat ischemic recirculation model. Brain Res. 615:281–288.
Nishio, S., Chen, Z. F., Yunoki, M., Toyoda, T., Anzivino, M., and Lee, K. S. (1999). Hypothermia-induced ischemic tolerance. Ann. N. Y. Acad. Sci. 890:26–41.
Nishio, S., Yunoki, M., Chen, Z. F., Anzivino, M. J., and Lee, K. S. (2000). Ischemic tolerance in the rat neocortex following hypothermic preconditioning. J. Neurosurg. 93:845–851.
Ohtsuki, T., Matsumoto, M., Kuwabara, K., Kitagawa, K., Suzuki, K., Taniguchi, N., and Kamada, T. (1992). Influence of oxidative stress on induced tolerance to ischemia in gerbil hippocampal neurons. Brain Res. 599:246–252.
Orendacova, J., Racekova, E., Kucharova, K., Pousova, B., Ondrejcak, T., Martoncikova, M., Daxnerova, Z., and Marsala, J. (2004). Ependyma as a possible morphological basis of ischemic preconditioning tolerance in rat spinal cord ischemia model: Nestin and Fluoro-Jade B observations. Cell. Mol. Neurobiol. 24:477–489.
Perez-Pinzon, M. A., and Born, J. G. (1999). Rapid preconditioning neuroprotection following anoxia in hippocampal slices: Role of the K+ ATP channel and protein kinase C. Neuroscience 89:453–459.
Puisieux, F., Deplanque, D., Pu, Q., Souil, E., Bastide, M., and Bordet, R. (2000). Differential role of nitric oxide pathway and heat shock protein in preconditioning and lipopolysaccharide-induced brain ischemic tolerance. Eur. J. Pharmacol. 389:71–78.
Pulsinelli, W. A., and Brierley, J. B. (1979). A new model of bilateral hemispheric ischemia in the unanesthetized rat. Stroke 10:267–272.
Ravingerova, T., Pancza, D., Ziegelhoffer, A., and Styk, J. (2002). Preconditioning modulates susceptibility to ischemia-induced arrhythmias in the rat heart: The role of alpha-adrenergic stimulation and K(ATP) channels. Physiol. Res. 51:109–119.
Ravingerova, T., Pyne, N. J., and Parratt, J. R. (1995). Ischaemic preconditioning in the rat heart: The role of G-proteins and adrenergic stimulation. Mol. Cell. Biochem. 147:123–128.
Riepe, M. W., Esclaire, F., Kasischke, K., Schreiber, S., Nakase, H., Kempski, O., Ludolph, A. C., Dirnagl, U., and Hugon, J. (1997). Increased hypoxic tolerance by chemical inhibition of oxidative phosphorylation: “Chemical preconditioning”. J. Cereb. Blood Flow Metab. 17:257–264.
Riksen, N. P., Smits, P., and Rongen, G. A. (2004). Ischaemic preconditioning: From molecular characterisation to clinical application. Part I. Neth. J. Med. 62:353–363.
Serviddio, G., Di Venosa, N., Federici, A., D’Agostino, D., Rollo, T., Prigigallo, F., Altomare, E., Fiore, T., and Vendemiale, G. (2005). Brief hypoxia before normoxic reperfusion (postconditioning) protects the heart against ischemia-reperfusion injury by preventing mitochondria peroxyde production and glutathione depletion. FASEB J. 19:354–361.
Shimazaki, K., Ishida, A., and Kawai, N. (1994). Increase in bcl-2 oncoprotein and the tolerance to ischemia-induced neuronal death in the gerbil hippocampus. Neurosci. Res. 20:95–99.
Schmidt-Kastner, R., Paschen, W., Ophoff, B. G., and Hossmann, K. A. (1989). A modified four-vessel occlusion model for inducing incomplete forebrain ischemia in rats. Stroke 20:938–946.
Sugino, T., Nozaki, K., Takagi, Y., and Hashimoto, N. (1999). 3-Nitropropionic acid induces ischemic tolerance in gerbil hippocampus in vivo. Neurosci. Lett. 259:9–12.
Tanaka, H., Yokota, H., Jover, T., Cappuccio, I., Calderone, A., Simionescu, M., Bennett, M. V., and Zukin, R. S. (2004). Ischemic preconditioning: Neuronal survival in the face of caspase-3 activation. J. Neurosci. 24:2750–2759.
Thilmann, R., Xie, Y., Kleihues, P., and Kiessling, M. (1986). Persistent inhibition of protein synthesis precedes delayed neuronal death in postischemic gerbil hippocampus. Acta Neuropathol. (Berl.) 71:88–93.
Trendelenburg, G., and Dirnagl, U. (2005). Neuroprotective role of astrocytes in cerebral ischemia: Focus on ischemic preconditioning. Glia 50:307–20.
Tsang, A., Hausenloy, D. J., Mocanu, M. M., and Yellon, D. M. (2004). Postconditioning: A form of “modified reperfusion” protects the myocardium by activating the phosphatidylinositol 3-kinase-Akt pathway. Circ. Res. 95:230–232.
Wada, K., Ito, M., Miyazawa, T., Katoh, H., Nawashiro, H., Shima, K., and Chigasaki, H. (1996). Repeated hyperbaric oxygen induces ischemic tolerance in gerbil hippocampus. Brain Res. 740:15–20.
Widmann, R., Miyazawa, T., and Hossmann, K. A. (1993). Protective effect of hypothermia on hippocampal injury after 30 min of forebrain ischemia in rats is mediated by postischemic recovery of protein synthesis. J. Neurochem. 61:200–209.
Wiegand, F., Liao, W., Busch, C., Castell, S., Knapp, F., Lindauer, U., Megow, D., Meisel, A., Redetzky, A., Ruscher, K., Trendelenburg, G., Victorov, I., Riepe, M., Diener, H. C., and Dirnagl, U. (1999). Respiratory chain inhibition induces tolerance to focal cerebral ischemia. J. Cereb. Blood Flow Metab. 19:1229–1237.
Zhao, Z. Q., Corvera, J. S., Halkos, M. E., Kerendi, F., Wang, N. P., Guyton, R. A., and Vinten-Johansen, J. (2003). Inhibition of myocardial injury by ischemic postconditioning during reperfusion: Comparison with ischemic preconditioning. Am. J. Physiol. Heart Circ. Physiol. 285:H579–H588.
Zvara, D. A., Colonna, D. M., Deal, D. D., Vernon, J. C., Gowda, M., and Lundell, J. C. (1999). Ischemic preconditioning reduces neurologic injury in a rat model of spinal cord ischemia. Ann. Thorac. Surg. 68:874–880.
ACKNOWLEDGMENTS
This work was supported by grants SK-VEGA 2/3219/23 and APVT 51-0219-04. We gratefully acknowledge the expert technical assistance of Viera Ujháziová, Dana Jurušová, and Zuzana Burdová.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Burda, J., Danielisová, V., Némethová, M. et al. Delayed Postconditionig Initiates Additive Mechanism Necessary for Survival of Selectively Vulnerable Neurons After Transient Ischemia in Rat Brain. Cell Mol Neurobiol 26, 1139–1149 (2006). https://doi.org/10.1007/s10571-006-9036-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-006-9036-x