Abstract
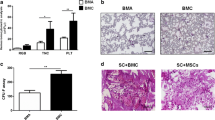
Bone marrow stimulation (BMS) has been regarded as a first-line procedure for the repair of articular cartilage. However, cartilage repair using BMS alone has so far not been ideal because cell homing to the required area has not been sufficient. The aim of this study was to investigate the feasibility of autologous bone marrow concentrate transplantation for the repair of large, full-thickness cartilage defects. Thirty rabbits were divided into five groups: untreated (control); BMS only (BMS); BMS followed by PGA implantation (PGA); BMS followed by a combination of PGA and autologous bone marrow concentrate (BMC); and BMS together with a composite of PGA and cultured bone marrow stem cells (BME). The animals were sacrificed at week 8 after operation, and HE staining, toluidine blue staining and immunohistochemistry were used to assess the repair of defects. The results showed that improved repair, including more newly formed cartilage tissue and hyaline cartilage-specific extracellular matrix, was observed in BMC group relative to the first three groups, in addition similar results were found between BMC and BME groups, however it took longer time for in vitro cell expansion in the BME group. This study demonstrates that the transplantation of autologous bone marrow concentrate is an easy, safe and potentially viable method to contribute to articular cartilage repair.





Similar content being viewed by others
References
Cao L, Yang F, Liu G, Yu D, Li H, Fan Q, et al. The promotion of cartilage defect repair using adenovirus mediated Sox9 gene transfer of rabbit bone marrow mesenchymal stem cells. Biomaterials. 2011;32(16):3910–20.
Hunziker EB. Articular cartilage repair: basic science and clinical progress. A review of the current status and prospects. Osteoarthritis Cartilage. 2002;10(6):432–63.
Jin LH, Choi BH, Kim YJ, Park SR, Jin CZ, Min BH. Implantation of bone marrow-derived buffy coat can supplement bone marrow stimulation for articular cartilage repair. Osteoarthritis Cartilage. 2011;19:1440–8.
Logeart-Avramoglou D, Anagnostou F, Bizios R, Petite H. Engineering bone: challenges and obstacles. J Cell Mol Med. 2005;9(1):72–84.
Bobacz K, Erlacher L, Smolen J, Soleiman A, Graninger WB. Chondrocyte number and proteoglycan synthesis in the aging and osteoarthritic human articular cartilage. Ann Rheum Dis. 2004;63(12):1618–22.
Zhou G, Liu W, Cui L, Wang X, Liu T, Cao Y. Repair of porcine articular osteochondral defects in non-weight bearing areas with autologous bone marrow stromal cells. Tissue Eng. 2006;12(11):3209–21.
D’Amour K, Gage FH. New tools for human developmental biology. Nat Biotechnol. 2000;18(4):381–2.
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284(5411):143–7.
Hernigou P, Poignard A, Manicom O, Mathieu G, Rouard H. The use of percutaneous autologous bone marrow transplantation in nonunion and avascular necrosis of bone. J Bone Joint Surg Br. 2005;87(7):896–902.
Karatoprak O, Korkmaz MF, Kara AN, Gogus A, Isiklar ZU. Early results of autologous mononuclear bone marrow cell implantation in nontraumatic avascular necrosis of the femoral head. Acta Orthop Traumatol Turc. 2008;42(3):178–83.
Makela J, Ylitalo K, Lehtonen S, Dahlbacka S, Niemela E, Kiviluoma K, et al. Bone marrow-derived mononuclear cell transplantation improves myocardial recovery by enhancing cellular recruitment and differentiation at the infarction site. J Thorac Cardiovasc Surg. 2007;134(3):565–73.
Gan Y, Dai K, Zhang P, Tang T, Zhu Z, Lu J. The clinical use of enriched bone marrow stem cells combined with porous beta-tricalcium phosphate in posterior spinal fusion. Biomaterials. 2008;29(29):3973–82.
Im GI, Kim DY, Shin JH, Hyun CW, Cho WH. Repair of cartilage defect in the rabbit with cultured mesenchymal stem cells from bone marrow. J Bone Joint Surg Br. 2001;83(2):289–94.
Yoshimura H, Muneta T, Nimura A, Yokoyama A, Koga H, Sekiya I. Comparison of rat mesenchymal stem cells derived from bone marrow, synovium, periosteum, adipose tissue, and muscle. Cell Tissue Res. 2007;327(3):449–62.
Qian Y, Lin Z, Chen J, Fan Y, Davey T, Cake M, et al. Natural bone collagen scaffold combined with autologous enriched bone marrow cells for induction of osteogenesis in an ovine spinal fusion model. Tissue Eng Part A. 2009;15(11):3547–58.
Jiang Y, Chen LK, Zhu DC, Zhang GR, Guo C, Qi YY, et al. The inductive effect of bone morphogenetic protein-4 on chondral-lineage differentiation and in situ cartilage repair. Tissue Eng Part A. 2010;16(5):1621–32.
Wu SC, Chang JK, Wang CK, Wang GJ, Ho ML. Enhancement of chondrogenesis of human adipose derived stem cells in a hyaluronan-enriched microenvironment. Biomaterials. 2010;31(4):631–40.
Ochi M, Adachi N, Nobuto H, Yanada S, Ito Y, Agung M. Articular cartilage repair using tissue engineering technique–novel approach with minimally invasive procedure. Artif Organs. 2004;28(1):28–32.
Chen H, Hoemann CD, Sun J, Chevrier A, McKee MD, Shive MS, et al. Depth of subchondral perforation influences the outcome of bone marrow stimulation cartilage repair. J Orthop Res. 2011;29(8):1178–84.
Hernigou P, Beaujean F. Treatment of osteonecrosis with autologous bone marrow grafting. Clin Orthop Relat Res. 2002;405:14–23.
Fortier LA, Potter HG, Rickey EJ, Schnabel LV, Foo LF, Chong LR, et al. Concentrated bone marrow aspirate improves full-thickness cartilage repair compared with microfracture in the equine model. J Bone Joint Surg Am. 2010;92(10):1927–37.
Suzuki Y, Kim KJ, Kotake S, Itoh T. Stromal cell activity in bone marrow from the tibia and iliac crest of patients with rheumatoid arthritis. J Bone Miner Metab. 2001;19(1):56–60.
Vilquin JT, Rosset P. Mesenchymal stem cells in bone and cartilage repair: current status. Regen Med. 2006;1(4):589–604.
Krampera M, Pizzolo G, Aprili G, Franchini M. Mesenchymal stem cells for bone, cartilage, tendon and skeletal muscle repair. Bone. 2006;39(4):678–83.
Muschler GF, Matsukura Y, Nitto H, Boehm CA, Valdevit AD, Kambic HE, et al. Selective retention of bone marrow-derived cells to enhance spinal fusion. Clin Orthop Relat Res. 2005;432:242–51.
Muschler GF, Nitto H, Matsukura Y, Boehm C, Valdevit A, Kambic H, et al. Spine fusion using cell matrix composites enriched in bone marrow-derived cells. Clin Orthop Relat Res. 2003;407:102–18.
Rosland GV, Svendsen A, Torsvik A, Sobala E, McCormack E, Immervoll H, et al. Long-term cultures of bone marrow-derived human mesenchymal stem cells frequently undergo spontaneous malignant transformation. Cancer Res. 2009;69(13):5331–9.
Matsumoto T, Okabe T, Ikawa T, Iida T, Yasuda H, Nakamura H, et al. Articular cartilage repair with autologous bone marrow mesenchymal cells. J Cell Physiol. 2010;225(2):291–5.
Tateishi-Yuyama E, Matsubara H, Murohara T, Ikeda U, Shintani S, Masaki H, et al. Therapeutic angiogenesis for patients with limb ischaemia by autologous transplantation of bone-marrow cells: a pilot study and a randomised controlled trial. Lancet. 2002;360(9331):427–35.
Ishimoto Y, Hattori K, Ohgushi H. Articular cartilage regeneration using scaffold. Clin Calcium. 2008;18(12):1775–80.
Sinha VR, Bansal K, Kaushik R, Kumria R, Trehan A. Poly-epsilon-caprolactone microspheres and nanospheres: an overview. Int J Pharm. 2004;278(1):1–23.
Rezwan K, Chen QZ, Blaker JJ, Boccaccini AR. Biodegradable and bioactive porous polymer/inorganic composite scaffolds for bone tissue engineering. Biomaterials. 2006;27(18):3413–31.
Acknowledgments
We acknowledge the support of the Specialized Research Fund for the Doctoral Program of Higher Education of China (Grant No. 20100073120090) and the Research funds for combination of engineering (science) with medicine of Shanghai Jiao Tong University (Grant No. YG2011MS28).
Author information
Authors and Affiliations
Corresponding author
Additional information
Shouguo Wang—Co-first author.
Rights and permissions
About this article
Cite this article
Zhao, Q., Wang, S., Tian, J. et al. Combination of bone marrow concentrate and PGA scaffolds enhance bone marrow stimulation in rabbit articular cartilage repair. J Mater Sci: Mater Med 24, 793–801 (2013). https://doi.org/10.1007/s10856-012-4841-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10856-012-4841-x