Abstract
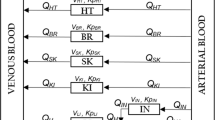
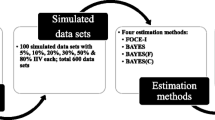
The objectives of the simulation study were to evaluate the impact of BQL data on pharmacokinetic (PK) parameter estimates when the incidence of BQL data is low (e.g. ≤10%), and to compare the performance of commonly used modeling methods for handling BQL data such as data exclusion (M1) and likelihood-based method (M3). Simulations were performed by adapting the method of a recent publication by Ahn et al. (J Phamacokinet Pharmacodyn 35(4):401–421, 2008). The BQL data in the terminal elimination phase were created at frequencies of 1, 2.5, 5, 7.5, and 10% based on a one- and a two-compartment model. The impact of BQL data on model parameter estimates was evaluated based on bias and imprecision. The simulations demonstrated that for the one-compartment model, the impact of ignoring the low percentages of BQL data (≤10%) in the elimination phase was minimal. For the two-compartment model, when the BQL incidence was less than 5%, omission of the BQL data generally did not inflate the bias in the fixed-effect parameters, whereas more pronounced bias in the estimates of inter-individual variability (IIV) was observed. The BQL data in the elimination phase had the greatest impact on the volume of distribution estimate of the peripheral compartment of the two-compartment model. The M3 method generally provided better parameter estimates for both PK models than the M1 method. However, the advantages of the M3 over the M1 method varied depending on different BQL censoring levels, PK models and parameters. As the BQL percentages decreased, the relative gain of the M3 method based on more complex likelihood approaches diminished when compared to the M1 method. Therefore, it is important to balance the trade-off between model complexity and relative gain in model improvement when the incidence of BQL data is low. Understanding the model structure and the distribution of BQL data (percentage and location of BQL data) allows selection of an appropriate and effective modeling approach for handling low percentages of BQL data.




Similar content being viewed by others
References
Ahn JE et al (2008) Likelihood based approaches to handling data below the quantification limit using NONMEM VI. J Pharmacokinet Pharmacodyn 35(4):401–421
Beal SL (2001) Ways to fit a PK model with some data below the quantification limit. J Pharmacokinet Pharmacodyn 28(5):481–504
Bergstrand M, Karlsson MO (2009) Handling data below the limit of quantification in mixed effect models. AAPS J 11(2):371–380
Byon W, Fletcher CV, Brundage RC (2008) Impact of censoring data below an arbitrary quantification limit on structural model misspecification. J Pharmacokinet Pharmacodyn 35(1):101–116
Duval V, Karlsson MO (2002) Impact of omission or replacement of data below the limit of quantification on parameter estimates in a two-compartment model. Pharm Res 19(12):1835–1840
Hing JP et al (2001) Analysis of toxicokinetic data using NONMEM: impact of quantification limit and replacement strategies for censored data. J Pharmacokinet Pharmacodyn 28(5):465–479
Acknowledgments
All authors are employees of Johnson & Johnson Pharmaceutical Research & Development. Steven Xu is an adjunct assistant professor in the School of Public Health at the University of Medicine and Dentistry of New Jersey.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10928_2011_9201_MOESM1_ESM.ppt
Supplementary Figure 1. Relative bias (%) for parameter estimates of the one-compartment model (additive residual error) at different BQL levels with M1 (°) and M3 (×) methods (N=1,000; successful minimizations only). The vertical lines represent the relative bias for parameter estimates based on the complete datasets without BQL censoring. (PPT 106 kb)
10928_2011_9201_MOESM2_ESM.ppt
Supplementary Figure 2. Root relative mean square error (%) for parameter estimates of the one-compartment model (additive residual error) at different BQL levels with M1 (°) and M3 (×) methods (N=1,000; successful minimizations only). The vertical lines represent the Root relative mean square errors for parameter estimates based on the complete datasets without BQL censoring. (PPT 106 kb)
10928_2011_9201_MOESM3_ESM.ppt
Supplementary Figure 3. Relative bias (%) for parameter estimates of the two-compartment model (additive residual error) at different BQL levels with M1 (°) and M3 (×) methods (N=1,000; successful minimizations only). The vertical lines represent the relative bias for parameter estimates based on the complete datasets without BQL censoring. (PPT 174 kb)
10928_2011_9201_MOESM4_ESM.ppt
Supplementary Figure 4. Root relative mean square error (%) for parameter estimates of the two compartment model (additive residual error) at different BQL levels with M1 (°) and M3 (×) methods (N=1,000; successful minimizations only). The vertical lines represent the Root relative mean square errors for parameter estimates based on the complete datasets without BQL censoring. (PPT 117 kb)
Rights and permissions
About this article
Cite this article
Xu, X.S., Dunne, A., Kimko, H. et al. Impact of low percentage of data below the quantification limit on parameter estimates of pharmacokinetic models. J Pharmacokinet Pharmacodyn 38, 423–432 (2011). https://doi.org/10.1007/s10928-011-9201-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-011-9201-9