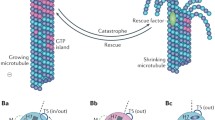
Abstract
Traditionally, kinesins have been identified as proteins that use the energy of ATP to translocate along microtubules. However, in the last decade some kinesin-like proteins were found to destabilize microtubule ends. The kinesins that destabilize microtubules are known as “catastrophe kinesins”. Analyses of a Xenopus member of the catastrophe kinesins called MCAK/XKCM1 have shown that, in fact, catastrophe kinesins are essential for controlling the distribution of microtubules by inducing their depolymerization. Therefore, unraveling the mechanisms of how microtubule destabilization promoted by these catastrophe kinesins is controlled is essential for understanding how microtubules in a cell are distributed. Here we give an overview of the studies that have focused on the global and local control of microtubule destabilization promoted by MCAK/XKCM1.
Similar content being viewed by others
References
Andrews PD, Ovechkina Y, Morrice N, Wagenbach M, Duncan K, Wordeman L, Swedlow JR (2004) Aurora B regulates MCAK at the mitotic centromere. Dev Cell 6:253–268
Barros TP, Kinoshita K, Hyman AA and Raff JW (2005) Aurora A activates D-TACC–Msps complexes exclusively at centrosomes to stabilize centrosomal microtubules. J Cell Biol 170:1039–1046
Belmont LD, Hyman AA, Sawin KE and Mitchison TJ (1990) Real-time visualization of cell cycle-dependent changes in microtubule dynamics in cytoplasmic extracts. Cell 62:579–589
Belmont LD and Mitchison TJ (1996) Identification of a protein that interacts with tubulin dimers and increases the catastrophe rate of microtubules. Cell 84:623–631
Blower MD, Nachury M, Heald R and Weis K (2005) A Rae1-containing ribonucleoprotein complex is required for mitotic spindle assembly. Cell 121:223–234
Brittle AL and Ohkura H (2005) Mini spindles, the XMAP215 homologue, suppresses pausing of interphase microtubules in Drosophila. EMBO J 24:1387–1396
Cassimeris L, Pryer NK and Salmon ED (1988) Real-time observations of microtubule dynamic instability in living cells. J Cell Biol 107:2223–2231
Cassimeris L and Morabito J (2004) TOGp, the human homolog of XMAP215/Dis1, is required for centrosome integrity, spindle pole organization, and bipolar spindle assembly. Mol Biol Cell 15:1580–1590
Cullen CF and Ohkura H (2001) Msps protein is localized to acentrosomal poles to ensure bipolarity of Drosophila meiotic spindles. Nat Cell Biol 3:637–642
Desai A and Mitchison TJ (1997) Microtubule polymerization dynamics. Annu Rev Cell Dev Biol 13:83–117
Desai A, Verma S, Mitchison TJ and Walczak CE (1999) Kin I kinesins are microtubule-destabilizing enzymes. Cell 96:69–78
Drechsel DN, Hyman AA, Cobb MH and Kirschner MW (1992) Modulation of the dynamic instability of tubulin assembly by the microtubule-associated protein tau. Mol Biol Cell 3:1141–1154
Gaetz J and Kapoor TM (2004) Dynein/dynactin regulate metaphase spindle length by targeting depolymerizing activities to spindle poles. J Cell Biol 166: 465–471
Ganem NJ and Compton DA (2004) The KinI kinesin Kif2a is required for bipolar spindle assembly through a functional relationship with MCAK. J Cell Biol 166:473–478
Gard DL and Kirschner MW (1987) A microtubule-associated protein from Xenopus eggs that specifically promotes assembly at the plus-end. J Cell Biol 105:2203–2215
Hayden JH, Bowser SS and Rieder CL (1990) Kinetochores capture astral microtubules during chromosome attachment to the mitotic spindle: direct visualization in live newt lung cells. J Cell Biol 111:1039–1045
Hertzer KM, Ems-McClung SC and Walczak CE (2003) Kin I kinesins: insights into the mechanism of depolymerization. Crit Rev Biochem Mol Biol 38:453–469
Holmfeldt P, Brattsand G and Gullberg M (2002) MAP4 counteracts microtubule catastrophe promotion but not tubulin-sequestering activity in intact cells. Curr Biol 12: 1034–1039
Holmfeldt P, Stenmark S and Gullberg M (2004) Differential functional interplay of TOGp/XMAP215 and the KinI kinesin MCAK during interphase and mitosis. EMBO J 23:627–637
Holmfeldt P, Zhang X, Stenmark S, Walczak CE and Gullberg M (2005) CaMKIIgamma-mediated inactivation of the Kin I kinesin MCAK is essential for bipolar spindle formation. EMBO J 24:1256–1266
Horio T and Hotani H (1986) Visualization of the dynamic instability of individual microtubules by dark-field microscopy. Nature 321:605–607
Howard J and Hyman AA (2003) Dynamics and mechanics of the microtubule plus end. Nature 422:753–758
Job D, Valiron O and Oakley B (2003) Microtubule nucleation. Curr Opin Cell Biol 15:111–117
Kinoshita K, Arnal I, Desai A, Drechsel DN and Hyman AA (2001) Reconstitution of physiological microtubule dynamics using purified components. Science 294:1340–1343
Kinoshita K, Habermann B and Hyman AA (2002) XMAP215: a key component of the dynamic microtubule cytoskeleton. Trends Cell Biol 12: 267–273
Kinoshita K, Noetzel TL, Pelletier L, Mechtler K, Drechsel DN, Schwager A, Lee M, Raff JW and Hyman AA (2005) Aurora A phosphorylation of TACC3/Maskin is required for centrosome-dependent microtubule assembly in mitosis. J Cell Biol 170:1047–1055
Kline-Smith SL and Walczak CE (2002) The microtubule-destabilizing kinesin XKCM1 regulates microtubule dynamic instability in cells. Mol Biol Cell 13:2718–2731
Lan W, Zhang X, Kline-Smith SL, Rosasco SE, Barrett-Wilt GA, Shabanowitz J, Hunt DF, Walczak CE and Stukenberg PT (2004) Aurora B phosphorylates centromeric MCAK and regulates its localization and microtubule depolymerization activity. Curr Biol 14: 273–286
Lee MJ, Gergely F, Jeffers K, Peak-Chew SY and Raff JW (2001) Msps/XMAP215 interacts with the centrosomal protein D-TACC to regulate microtubule behaviour. Nat Cell Biol 3:643–649
Lewis SA, Wang DH and Cowan NJ (1988) Microtubule-associated protein MAP2 shares a microtubule binding motif with tau protein. Science 242:936–939
Mennella V, Rogers GC, Rogers SL, Buster DW, Vale RD, and Sharp DJ (2005) Functionally distinct kinesin-13 family members cooperate to regulate microtubule dynamics during interphase. Nat Cell Biol 7:235–245
McNally FJ (1996) Modulation of microtubule dynamics during the cell cycle. Curr Opin Cell Biol 8:23–29
Mitchison T and Kirschner M (1984) Dynamic instability of microtubule growth. Nature 312:237–242
Moore A and Wordeman L (2004) The mechanism, function and regulation of depolymerizing kinesins during mitosis. Trends Cell Biol 14:537–546
Moore AT, Rankin KE, von Dassow G, Peris L, Wagenbach M, Ovechkina Y, Andrieux A, Job D and Wordeman L (2005) MCAK associates with the tips of polymerizing microtubules. J Cell Biol 169:391–397
Morfini G, Quiroga S, Rosa A, Kosik K and Cáceres A (1997) Suppression of KIF2 in PC12 cells alters the distribution of a growth cone nonsynaptic membrane receptor and inhibits neurite extension. J Cell Biol 138:657–669
Noda Y, Sato-Yoshitake R, Kondo S, Nangaku M and Hirokawa N (1995) KIF2 is a new microtubule-based anterograde motor that transports membranous organelles distinct from those carried by kinesin heavy chain or KIF3A/B. J Cell Biol 129:157–167
Noetzel TL, Drechsel DN, Hyman AA and Kinoshita K (2005) A comparison of the ability of XMAP215 and tau to inhibit the microtubule destabilizing activity of XKCM1. Phil Trans R Soc Lond B Biol Sci 360:591–594
O’Brien LL, Albee AJ, Liu L, Tao W, Dobrzyn P, Lizarraga SB and Wiese C (2005) The Xenopus TACC homologue, maskin, functions in mitotic spindle assembly. Mol Biol Cell 16:2836–2847
Ohi R, Coughlin ML, Lane WS and Mitchison TJ (2003) An inner centromere protein that stimulates the microtubule depolymerizing activity of a KinI kinesin. Dev Cell 5: 309–321
Ohi R, Sapra T, Howard J and Mitchison TJ (2004) Differentiation of cytoplasmic and meiotic spindle assembly MCAK functions by Aurora B-dependent phosphorylation. Mol Biol Cell 15:2895–2906
Ovechkina Y and Wordeman L (2003) Unconventional motoring: an overview of the Kin C and Kin I kinesins. Traffic 4:367–375
Peset I, Seiler J, Sardon T, Bejarano LA, Rybina S and Vernos I (2005) Function and regulation of Maskin, a TACC family protein, in microtubule growth during mitosis. J Cell Biol 170:1057–1066
Raff JW (2002) Centrosomes and cancer: lessons from a TACC. Trends Cell Biol 12:222–225
Rusan NM, Fagerstrom CJ, Yvon AM and Wadsworth P (2001) Cell cycle-dependent changes in microtubule dynamics in living cells expressing green fluorescent protein-alpha tubulin. Mol Biol Cell 12:971–980
Sampath SC, Ohi R, Leismann O, Salic A, Pozniakovski A and Funabiki H (2004) The chromosomal passenger complex is required for chromatin-induced microtubule stabilization and spindle assembly. Cell 118:187–202
Sharp DJ, Rogers GC and Scholey JM (2000) Roles of motor proteins in building microtubule-based structures: a basic principle of cellular design. Biochim Biophys Acta 1496:128–141
Shirasu-Hiza M, Coughlin P and Mitchison T (2003) Identification of XMAP215 as a microtubule-destabilizing factor in Xenopus egg extract by biochemical purification. J Cell Biol 161:349–358
Spittle C, Charrasse S, Larroque C and Cassimeris L (2000) The interaction of TOGp with microtubules and tubulin. J Biol Chem 275:20748–20753
Srayko M, Kaya A, Stamford J and Hyman AA (2005) Identification and characterization of factors required for microtubule growth and nucleation in the early C. elegans embryo. Dev Cell 9:223–236
Stebbins-Boaz B, Cao Q, de Moor CH, Mendez R and Richter JD (1999) Maskin is a CPEB-associated factor that transiently interacts with elF-4E. Mol Cell 4:1017–1027
Still IH, Hamilton M, Vince P, Wolfman A and Cowell JK (1999a) Cloning of TACC1, an embryonically expressed, potentially transforming coiled coil containing gene, from the 8p11 breast cancer amplicon. Oncogene 18:4032–4038
Still IH, Vince P and Cowell JK (1999b) The third member of the transforming acidic coiled coil-containing gene family, TACC3, maps in 4p16, close to translocation breakpoints in multiple myeloma, and is upregulated in various cancer cell lines. Genomics 58:165–170
Still IH, Vettaikkorumakankauv AK, DiMatteo A and Liang P (2004) Structure-function evolution of the transforming acidic coiled coil genes revealed by analysis of phylogenetically diverse organisms. BMC Evol Biol 4:16
Tanaka TU, Rachidi N, Janke C, Pereira G, Galova M, Schiebel E, Stark MJ and Nasmyth K (2002) Evidence that the Ipl1-Sli15 (Aurora kinase-INCENP) complex promotes chromosome bi-orientation by altering kinetochore-spindle pole connections. Cell 108:317–329
Tournebize R, Popov A, Kinoshita K, Ashford AJ, Rybina S, Pozniakovsky A, Mayer TU, Walczak CE, Karsenti E and Hyman AA (2000) Control of microtubule dynamics by the antagonistic activities of XMAP215 and XKCM1 in Xenopus egg extracts. Nat Cell Biol 2:13–19
Vasquez RJ, Gard DL and Cassimeris L (1994) XMAP from Xenopus eggs promotes rapid plus end assembly of microtubules and rapid microtubule polymer turnover. J Cell Biol 127:985–993
Walczak CE (2000) Microtubule dynamics and tubulin interacting proteins. Curr Opin Cell Biol 12:52–56
Walczak CE, Mitchison TJ and Desai A (1996) XKCM1: a Xenopus kinesin-related protein that regulates microtubule dynamics during mitotic spindle assembly. Cell 84:37–47
Walker RA, O’Brien ET, Pryer NK, Soboeiro MF, Voter WA, Erickson HP and Salmon ED (1988) Dynamic instability of individual microtubules analyzed by video light microscopy: rate constants and transition frequencies. J Cell Biol 107:1437–1448
Wordeman L and Mitchison TJ (1995) Identification and partial characterization of mitotic centromere-associated kinesin, a kinesin-related protein that associates with centromeres during mitosis. J Cell Biol 128:95–104
Wordeman L (2005) Microtubule-depolymerizing kinesins. Curr Opin Cell Biol 17:82–88
Acknowledgements
We thank Martin Srayko (MPI-CBG) for helpful comments on the manuscript. Our studies described here were supported by a grant from DFG-priority program ‘Molecular Motors’.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kinoshita, K., Noetzel, T.L., Arnal, I. et al. Global and local control of microtubule destabilization promoted by a catastrophe kinesin MCAK/XKCM1. J Muscle Res Cell Motil 27, 107–114 (2006). https://doi.org/10.1007/s10974-005-9045-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-005-9045-2