Abstract
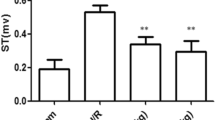
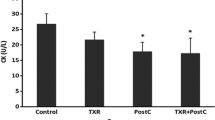
Toll-like receptor 4 (TLR4) activation has been implicated in the pathogenesis of myocardial ischemia/reperfusion (I/R) injury. The activated TLR4 is capable of activating a variety of proinflammatory mediators, such as tumor necrosis factor-a (TNF-a) and interleukin-6 (IL-6). Valsartan as a kind of Angiotensin II type 1 receptor blockers is gradually used for the treatment of ischemic heart disease depending on its anti-inflammation function. Therefore, we hypothesized that valsartan protects against myocardial I/R injury by suppressing TLR4 activation. We constructed the rat model of myocardial I/R injury. The rats were pretreated with valsartan for 2 weeks, and then subjected to 30 min ischemia and 2 h reperfusion. TLR4 and Nuclear factor kappa-B (NF-κB) levels were detected by quantitative real-time PCR and western blot. In order to evaluate myocardial damage, the myocardial infarct size, histopathologic changes, and the release of myocardial enzymes, proinflammation cytokines and Angiotensin II were analyzed by triphenyl tetrazolium chloride (TTC) staining, light microscopy, and enzyme-linked immunosorbent assay (ELISA), respectively. Valsartan preconditioning inhibited TLR4 and NF-κB expressions concomitant with an improvement in myocardial injury, such as smaller infarct size, fewer release of myocardial enzymes, and proinflammation mediators. These findings suggest that valsartan plays a pivotal role in the protective effects on myocardial I/R injury. This protection mechanism is possibly due to its anti-inflammation function via TLR4/NF-κB signaling pathway.






Similar content being viewed by others
References
Gruppo Italiano per lo Studio della Streptochinasi nell’Infarto Miocardico (GISSI) (1986) Effectiveness of intravenous thrombolytic treatment in acute myocardial infarction. Lancet 1:397–402
ISIS-2 (Second International Study of Infarct Survival) Collaborative Group (1988) Randomised trial of intravenous streptokinase, oral aspirin, both, or neither among 17, 187 cases of suspected acute myocardial infarction: ISIS-2. Lancet 2:349–360
Cannon CP, Gibson CM, Lambrew CT, Shoultz DA, Levy D, French WJ et al (2000) Relationship of symptom-onset-to-balloon time and door-to-balloon time with mortality in patients undergoing angioplasty for acute myocardial infarction. JAMA 283:2941–2947. doi:10.1001/jama.283.22.2941
Ribichini F, Wijns W (2002) Acute myocardial infarction: reperfusion treatment. Heart 88:298–305. doi:10.1136/heart.88.3.298
Hansen PR (1995) Myocardial reperfusion injury: experimental evidence and clinical relevance. Eur Heart J 16:734–740
Vakeva AP, Agah A, Rollins SA, Matis LA, Li L, Stahl GL (1998) Myocardial infarction and apoptosis after myocardial ischemia and reperfusion: role of the terminal complement components and inhibition by anti-C5 therapy. Circulation 97:2259–2267
Frangogiannis NG, Smith CW, Entman ML (2002) The inflammatory response in myocardial infarction. Cardiovasc Res 53:31–47. doi:10.1016/S0008-6363(01)00434-5
Serhan CN, Jain A, Marleau S, Clish C, Kantarci A, Behbehani B et al (2003) Reduced inflammation and tissue damage in transgenic rabbits overexpressing 15-lipoxygenase and endogenous anti-inflammatory lipid mediators. J Immunol 171:6856–6865
Serhan CN (2005) Novel omega-3-derived local mediators in anti-inflammation and resolution. Pharmacol Ther 105:7–21. doi:10.1016/j.pharmthera.2004.09.002
Medzhitov R, Preston-Hurlburt P, Janeway CA Jr (1997) A human homologue of the Drosophila toll protein signals activation of adaptive immunity. Nature 388:394–397. doi:10.1038/41131
Miyake K (2007) Innate immune sensing of pathogens and danger signals by cell surface toll-like receptors. Semin Immunol 19:3–10. doi:10.1016/j.smim.2006.12.002
Akira S, Takeda K, Kaisho T (2001) Toll-like receptors: critical proteins linking innate and acquired immunity. Nat Immunol 2:675–680. doi:10.1038/90609
Baumgarten G, Knuefermann P, Nozaki N, Sivasubramanian N, Mann DL, Vallejo JG (2001) In vivo expression of proinflammatory mediators in the adult heart after endotoxin administration: the role of toll-like receptor-4. J Infect Dis 183:1617–1624. doi:10.1086/320712
Oyama J, Blais C Jr, Liu X, Pu M, Kobzik L, Kelly RA et al (2004) Reduced myocardial ischemia-reperfusion injury in toll-like receptor 4-deficient mice. Circulation 109:784–789. doi:10.1161/01.CIR.0000112575.66565.84
Chong AJ, Shimamoto A, Hampton CR, Takayama H, Spring DJ, Rothnie CL et al (2004) Toll-like receptor 4 mediates ischemia/reperfusion injury of the heart. J Thorac Cardiovasc Surg 128:170–179. doi:10.1016/j.jtcvs.2003.11.036
Yang J, Yang J, Ding JW, Chen LH, Wang YL, Li S et al (2008) Sequential expression of TLR4 and its effects on the myocardium of rats with myocardial ischemia-reperfusion injury. Inflammation 31:304–312. doi:10.1007/s10753-008-9079-x
Dahlöf B, Devereux RB, Kjeldsen SE, Julius S, Beevers G, de Faire U et al (2002) Cardiovascular morbidity and mortality in the Losartan Intervention For Endpoint reduction in hypertension study (LIFE): a randomised trial against atenolol. Lancet 359:995–1003. doi:10.1016/S0140-6736(02)08089-3
Brunner HR, Gavras H (2002) Angiotensin blockade for hypertension: a promise fulfilled. Lancet 359:990–992. doi:10.1016/S0140-6736(02)08062-5
Zannad F, Fay R (2007) Blood pressure-lowering efficacy of olmesartan relative to other angiotensin II receptor antagonists: an overview of randomized controlled studies. Fundam Clin Pharmacol 21:181–190. doi:10.1111/j.1472-8206.2007.00464.x
Navalkar S, Parthasarathy S, Santanam N, Khan BV (2001) Irbesartan, an angiotensin type 1 receptor inhibitor, regulates markers of inflammation in patients with premature atherosclerosis. J Am Coll Cardiol 37:440–444. doi:10.1016/S0735-1097(00)01138-4
Cianchetti S, Del Fiorentino A, Colognato R, Di Stefano R, Franzoni F, Pedrinelli R (2008) Anti-inflammatory and anti-oxidant properties of telmisartan in cultured human umbilical vein endothelial cells. Atherosclerosis 198:22–28. doi:10.1016/j.atherosclerosis.2007.09.013
Candido R, Allen TJ, Lassila M, Cao Z, Thallas V, Cooper ME et al (2004) Irbesartan but not amlodipine suppresses diabetes-associated atherosclerosis. Circulation 109:1536–1542. doi:10.1161/01.CIR.0000124061.78478.94
Sironi L, Gelosa P, Guerrini U, Banfi C, Crippa V, Brioschi M et al (2004) Anti-inflammatory effects of AT1 receptor blockade provide end-organ protection in stroke-prone rats independently from blood pressure fall. J Pharmacol Exp Ther 311:989–995. doi:10.1124/jpet.104.072066
Varagic J, Frohlich ED, Susic D, Ahn J, Matavelli L, López B et al (2008) AT1 receptor antagonism attenuates target organ effects of salt excess in SHRs without affecting pressure. Am J Physiol Heart Circ Physiol 294:H853–H858. doi:10.1152/ajpheart.00737.2007
Chan YC, Leung PS (2007) Angiotensin II type 1 receptor-dependent nuclear factor-kappaB activation-mediated proinflammatory actions in a rat model of obstructive acute pancreatitis. J Pharmacol Exp Ther 323:10–18. doi:10.1124/jpet.107.124891
Maulik N, Engelman RM, Rousou JA, Flack JEIII, Deaton D, Das DK (1999) Ischemic preconditioning reduces apoptosis by upregulating anti-death gene Bcl-2. Circulation 100:II369–II375
Marino JH, Cook P, Miller KS (2003) Accurate and statistically verified quantification of relative mRNA abundances using SYBR Green I and real-time RT-PCR. J Immunol Methods 283:291–306. doi:10.1016/S0022-1759(03)00103-0
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A et al (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:RESEARCH0034
Altavilla D, Saitta A, Guarini S, Galeano M, Squadrito G, Cucinotta D et al (2001) Oxidative stress causes nuclear factor-kappaB activation in acute hypovolemic hemorrhagic shock. Free Radic Biol Med 30:1055–1066. doi:10.1016/S0891-5849(01)00492-0
Zheng YQ, Wei W (2005) Total glucosides of paeony suppresses adjuvant arthritis in rats and intervenes cytokine-signaling between different types of synoviocytes. Int Immunopharmacol 5:1560–1573. doi:10.1016/j.intimp.2005.03.010
Zeuke S, Ulmer AJ, Kusumoto S, Katus HA, Heine H (2002) TLR4-mediated inflammatory activation of human coronary artery endothelial cells by LPS. Cardiovasc Res 56:126–134. doi:10.1016/S0008-6363(02)00512-6
Nemoto S, Vallejo JG, Knuefermann P, Misra A, Defreitas G, Carabello BA et al (2002) Escherichia coli LPS-induced LV dysfunction: role of toll-like receptor-4 in the adult heart. Am J Physiol Heart Circ Physiol 282:H2316–H2323
Beg AA (2002) Endogenous ligands of toll-like receptors: implications for regulating inflammatory and immune responses. Trends Immunol 23:509–512. doi:10.1016/S1471-4906(02)02317-7
Dybdahl B, Wahba A, Lien E, Flo TH, Waage A, Qureshi N et al (2002) Inflammatory response after open heart surgery: release of heat-shock protein 70 and signaling through toll-like receptor-4. Circulation 105:685–690. doi:10.1161/hc0602.103617
Altavilla D, Deodato B, Campo GM, Arlotta M, Miano M, Squadrito G et al (2000) IRFI 042, a novel dual vitamin E-like antioxidant, inhibits activation of nuclear factor-kappaB and reduces the inflammatory response in myocardial ischemia-reperfusion injury. Cardiovasc Res 47:515–528. doi:10.1016/S0008-6363(00)00124-3
Shimamoto A, Chong AJ, Yada M, Shomura S, Takayama H, Fleisig AJ et al (2006) Inhibition of toll-like receptor 4 with eritoran attenuates myocardial ischemia-reperfusion injury. Circulation 114:I270–I274. doi:10.1161/CIRCULATIONAHA.105.000901
Ridker PM, Danielson E, Rifai N, Glynn RJ, Val-MARC Investigators (2006) Valsartan, blood pressure reduction, and C-reactive protein: primary report of the Val-MARC trial. Hypertension 48:73–79. doi:10.1161/01.HYP.0000226046.58883.32
Mueller EA, Griffin WS, Wildenthal K (1977) Isoproterenol-induced cardiomyopathy: changes in cardiac enzymes and protection by methylprednisolone. J Mol Cell Cardiol 9:565–578. doi:10.1016/S0022-2828(77)80371-4
van Kats JP, Duncker DJ, Haitsma DB, Schuijt MP, Niebuur R, Stubenitsky R et al (2000) Angiotensin-converting enzyme inhibition and angiotensin II type 1 receptor blockade prevent cardiac remodeling in pigs after myocardial infarction: role of tissue angiotensin II. Circulation 102:1556–1563
Silvestre JS, Heymes C, Oubénaïssa A, Robert V, Aupetit-Faisant B, Carayon A et al (1999) Activation of cardiac aldosterone production in rat myocardial infarction: effect of angiotensin II receptor blockade and role in cardiac fibrosis. Circulation 99:2694–2701
Dai Q, Xu M, Yao M, Sun B (2007) Angiotensin AT1 receptor antagonists exert anti-inflammatory effects in spontaneously hypertensive rats. Br J Pharmacol 152:1042–1048. doi:10.1038/sj.bjp.0707454
Seeger H, Mueck AO, Lippert TH (2000) Effects of valsartan and 17 beta-estradiol on the oxidation of low-density lipoprotein in vitro. Coron Artery Dis 11:347–349. doi:10.1097/00019501-200006000-00008
Acknowledgment
We thank Jia-jun Wang from the Department of Immunology, Medical Science College of China Three Gorges University for his technical support and helpful suggestions on this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, J., Jiang, H., Yang, J. et al. Valsartan preconditioning protects against myocardial ischemia–reperfusion injury through TLR4/NF-κB signaling pathway. Mol Cell Biochem 330, 39–46 (2009). https://doi.org/10.1007/s11010-009-0098-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-009-0098-1