Abstract
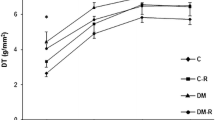
It has been previously shown that regulators of physiological growth such as thyroid hormone (TH) can favorably remodel the post ischaemic myocardium. Here, we further explored whether this effect can be preserved in the presence of co-morbidities such as diabetes which accelerates cardiac remodeling and increases mortality after myocardial infarction. Acute myocardial infarction (AMI) was induced by left coronary ligation in rats with type I diabetes (DM) induced by streptozotocin administration (STZ; 35 mg/kg; i.p.) while sham-operated animals served as controls (SHAM). AMI resulted in distinct changes in cardiac function and geometry; EF% was significantly decreased in DM-AMI [37.9 ± 2.0 vs. 74.5 ± 2.1 in DM-SHAM]. Systolic and diastolic chamber dimensions were increased without concomitant increase in wall thickness and thus, wall tension index [WTI, the ratio of (Left Ventricular Internal Diameter at diastole)/2*(Posterior Wall thickness)], an index of wall stress, was found to be significantly increased in DM-AMI; 2.27 ± 0.08 versus 1.70 ± 0.05. 2D-Strain echocardiographic analysis showed reduced systolic radial strain in all segments, indicating increased loss of cardiac myocytes in the infarct related area and less compensatory hypertrophy in the viable segments. This response was accompanied by a marked decrease in the expression of TRα1 and TRβ1 receptors in the diabetic myocardium without changes in circulating T3 and T4. Accordingly, the expression of TH target genes related to cardiac contractility was altered; β-MHC and PKCα were significantly increased. TH (L-T4 and L-T3) administration prevented these changes and resulted in increased EF%, normal wall stress and increased systolic radial strain in all myocardial segments. Acute myocardial infarction in diabetic rats results in TH receptor down-regulation with important physiological consequences. TH treatment prevents this response and improves cardiac hemodynamics.




Similar content being viewed by others
References
Swynghedauw B (1999) Molecular mechanisms of myocardial remodeling. Physiol Rev 79:215–262
Pantos C, Mourouzis I, Xinaris C, Kokkinos AD, Markakis K, Dimopoulos A, Panagiotou M, Saranteas T, Kostopanagiotou G, Cokkinos DV (2007) Time-dependent changes in the expression of thyroid hormone receptor {alpha}1 in the myocardium after acute myocardial infarction: possible implications in cardiac remodelling. Eur J Endocrinol 156:415–424
Chen YF, Kobayashi S, Chen J, Redetzke RA, Said S, Liang Q, Gerdes AM (2008) Short term triiodo-L-thyronine treatment inhibits cardiac myocyte apoptosis in border area after myocardial infarction in rats. J Mol Cell Cardiol 44:180–187
Pantos C, Mourouzis I, Markakis K, Dimopoulos A, Xinaris C, Kokkinos AD, Panagiotou M, Cokkinos DV (2007) Thyroid hormone attenuates cardiac remodeling and improves hemodynamics early after acute myocardial infarction in rats. Eur J Cardiothorac Surg 32:333–339
Pantos C, Mourouzis I, Saranteas T, Clave G, Ligeret H, Noack-Fraissignes P, Renard PY, Massonneau M, Perimenis P, Spanou D, Kostopanagiotou G, Cokkinos DV (2009) Thyroid hormone improves postischaemic recovery of function while limiting apoptosis: a new therapeutic approach to support hemodynamics in the setting of ischaemia-reperfusion? Basic Res Cardiol 104:69–77
Hassouna A, Loubani M, Matata BM, Fowler A, Standen NB, Galinanes M (2006) Mitochondrial dysfunction as the cause of the failure to precondition the diabetic human myocardium. Cardiovasc Res 69:450–458
Pantos C, Mourouzis I, Cokkinos DV (2007) Protection of the abnormal heart. Heart Fail Rev 12:319–330
Haffner SM, Lehto S, Ronnemaa T, Pyorala K, Laakso M (1998) Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 339:229–234
Kalofoutis C, Piperi C, Kalofoutis A, Harris F, Phoenix D, Singh J (2007) Type II diabetes mellitus and cardiovascular risk factors: current therapeutic approaches. Exp Clin Cardiol 12:17–28
Chandler MP, Morgan EE, McElfresh TA, Kung TA, Rennison JH, Hoit BD, Young ME (2007) Heart failure progression is accelerated following myocardial infarction in type 2 diabetic rats. Am J Physiol Heart Circ Physiol 293:H1609–H1616
Shiomi T, Tsutsui H, Ikeuchi M, Matsusaka H, Hayashidani S, Suematsu N, Wen J, Kubota T, Takeshita A (2003) Streptozotocin-induced hyperglycemia exacerbates left ventricular remodeling and failure after experimental myocardial infarction. J Am Coll Cardiol 42:165–172
Landau D, Chayat C, Zucker N, Golomb E, Yagil C, Yagil Y, Segev Y (2008) Early blood pressure-independent cardiac changes in diabetic rats. J Endocrinol 197:75–83
Soliman H, Craig GP, Nagareddy P, Yuen VG, Lin G, Kumar U, McNeill JH, Macleod KM (2008) Role of inducible nitric oxide synthase in induction of RhoA expression in hearts from diabetic rats. Cardiovasc Res 79:322–330
Van Linthout S, Seeland U, Riad A, Eckhardt O, Hohl M, Dhayat N, Richter U, Fischer JW, Bohm M, Pauschinger M, Schultheiss HP, Tschope C (2008) Reduced MMP-2 activity contributes to cardiac fibrosis in experimental diabetic cardiomyopathy. Basic Res Cardiol 103:319–327
Pantos C, Mourouzis I, Markakis K, Tsagoulis N, Panagiotou M, Cokkinos DV (2008) Long-term thyroid hormone administration re-shapes left ventricular chamber and improves cardiac function after myocardial infarction in rats. Basic Res Cardiol 103:308–318
Migrino RQ, Zhu X, Pajewski N, Brahmbhatt T, Hoffmann R, Zhao M (2007) Assessment of segmental myocardial viability using regional 2-dimensional strain echocardiography. J Am Soc Echocardiogr 20:342–351
Notomi Y, Lysyansky P, Setser RM, Shiota T, Popovic ZB, Martin-Miklovic MG, Weaver JA, Oryszak SJ, Greenberg NL, White RD, Thomas JD (2005) Measurement of ventricular torsion by two-dimensional ultrasound speckle tracking imaging. J Am Coll Cardiol 45:2034–2041
Pantos C, Mourouzis I, Saranteas T, Paizis I, Xinaris C, Malliopoulou V, Cokkinos DV (2005) Thyroid hormone receptors alpha1 and beta1 are downregulated in the post-infarcted rat heart: consequences on the response to ischaemia-reperfusion. Basic Res Cardiol 100:422–432
Pantos C, Mourouzis I, Galanopoulos G, Gavra M, Perimenis P, Spanou D and Cokkinos DV (2010) Thyroid hormone receptor α1 down-regulation in postischemic heart failure progression: the potential role of tissue hypothyroidism. Hormone and Metabolic Research. doi: 10.1055/s-0030-1255035
Pantos C, Xinaris C, Mourouzis I, Malliopoulou V, Kardami E, Cokkinos DV (2007) Thyroid hormone changes cardiomyocyte shape and geometry via ERK signaling pathway: potential therapeutic implications in reversing cardiac remodeling? Mol Cell Biochem 297:65–72
Pantos C, Xinaris C, Mourouzis I, Perimenis P, Politi E, Spanou D, Cokkinos DV (2008) Thyroid hormone receptor alpha 1: a switch to cardiac cell “metamorphosis”? J Physiol Pharmacol 59:253–269
Satoh M, Minami Y, Takahashi Y, Nakamura M (2008) Immune modulation: role of the inflammatory cytokine cascade in the failing human heart. Curr Heart Fail Rep 5:69–74
Kinugawa K, Jeong MY, Bristow MR, Long CS (2005) Thyroid hormone induces cardiac myocyte hypertrophy in a thyroid hormone receptor alpha1-specific manner that requires TAK1 and p38 mitogen-activated protein kinase. Mol Endocrinol 19:1618–1628
Kinugawa K, Yonekura K, Ribeiro RC, Eto Y, Aoyagi T, Baxter JD, Camacho SA, Bristow MR, Long CS, Simpson PC (2001) Regulation of thyroid hormone receptor isoforms in physiological and pathological cardiac hypertrophy. Circ Res 89:591–598
Belakavadi M, Saunders J, Weisleder N, Raghava PS, Fondell JD (2010) Repression of cardiac phospholamban gene expression is mediated by thyroid hormone receptor-{alpha}1 and involves targeted covalent histone modifications. Endocrinology 151:2946–2956
Rybin V, Steinberg SF (1996) Thyroid hormone represses protein kinase C isoform expression and activity in rat cardiac myocytes. Circ Res 79:388–398
Hambleton M, Hahn H, Pleger ST, Kuhn MC, Klevitsky R, Carr AN, Kimball TF, Hewett TE, Dorn GW II, Koch WJ, Molkentin JD (2006) Pharmacological- and gene therapy-based inhibition of protein kinase C alpha/beta enhances cardiac contractility and attenuates heart failure. Circulation 114:574–582
Scruggs SB, Walker LA, Lyu T, Geenen DL, Solaro RJ, Buttrick PM, Goldspink PH (2006) Partial replacement of cardiac troponin I with a non-phosphorylatable mutant at serines 43/45 attenuates the contractile dysfunction associated with PKCepsilon phosphorylation. J Mol Cell Cardiol 40:465–473
Tang YD, Kuzman JA, Said S, Anderson BE, Wang X, Gerdes AM (2005) Low thyroid function leads to cardiac atrophy with chamber dilatation, impaired myocardial blood flow, loss of arterioles, and severe systolic dysfunction. Circulation 112:3122–3130
Pantos C, Mourouzis I, Tsagoulis N, Markakis K, Galanopoulos G, Roukounakis N, Perimenis P, Liappas A, Cokkinos DV (2009) Thyroid hormone at supra-physiological dose optimizes cardiac geometry and improves cardiac function in rats with old myocardial infarction. J Physiol Pharmacol 60:49–56
Migrino RQ, Aggarwal D, Konorev E, Brahmbhatt T, Bright M, Kalyanaraman B (2008) Early detection of doxorubicin cardiomyopathy using two-dimensional strain echocardiography. Ultrasound Med Biol 34:208–214
Suzuki S, Miyamoto T, Opsahl A, Sakurai A, DeGroot LJ (1994) Two thyroid hormone response elements are present in the promoter of human thyroid hormone receptor beta 1. Mol Endocrinol 8:305–314
Maruvada P, Baumann CT, Hager GL, Yen PM (2003) Dynamic shuttling and intranuclear mobility of nuclear hormone receptors. J Biol Chem 278:12425–12432
Bunn CF, Neidig JA, Freidinger KE, Stankiewicz TA, Weaver BS, McGrew J, Allison LA (2001) Nucleocytoplasmic shuttling of the thyroid hormone receptor alpha. Mol Endocrinol 15:512–533
Ojamaa K (2010) Signaling mechanisms in thyroid hormone-induced cardiac hypertrophy. Vascul Pharmacol 52:113–119
Pantos C, Mourouzis I, Cokkinos DV (2010) Rebuilding the post-infarcted myocardium by activating ‘physiologic’ hypertrophic signaling pathways: the thyroid hormone paradigm. Heart Fail Rev 15:143–154
Pantos C, Mourouzis I, Cokkinos DV (2010) Thyroid hormone as a therapeutic option for treating ischaemic heart disease: from early reperfusion to late remodelling. Vascul Pharmacol 52:157–165
Acknowledgments
S. Niarchos foundation and the Hellenic Cardiology Society for supporting this piece of work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kalofoutis, C., Mourouzis, I., Galanopoulos, G. et al. Thyroid hormone can favorably remodel the diabetic myocardium after acute myocardial infarction. Mol Cell Biochem 345, 161–169 (2010). https://doi.org/10.1007/s11010-010-0569-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0569-4