Abstract
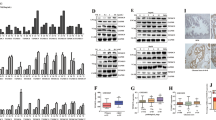
Reactive oxygen species (ROS) play a central role in oxidative stress, which leads to the onset of diseases, such as cancer. Furthermore, ROS contributes to the delicate balance between tumor cell survival and death. However, the mechanisms by which tumor cells decide to elicit survival or death signals during oxidative stress are not completely understood. We have previously reported that ROS enhanced tumorigenic functions in prostate cancer cells, such as transendothelial migration and invasion, which depended on CXCR4 and AKT signaling. Here, we report a novel mechanism by which ROS facilitated cell death through activation of AKT. We initially observed that ROS enhanced the expression of phosphorylated AKT (p-AKT) in 22Rv1 human prostate cancer cells. The tumor suppressor PTEN, a negative regulator of AKT signaling, was rendered catalytically inactive through oxidation by ROS, although the expression levels remained consistent. Despite these events, cells still underwent apoptosis. Further investigation into apoptosis revealed that expression of the tumor suppressor pVHL increased, and contains a target site for p-AKT phosphorylation. pVHL and p-AKT associated in vitro, and knockdown of pVHL rescued HIF1α expression and the cells from apoptosis. Collectively, our study suggests that in the context of oxidative stress, p-AKT facilitated apoptosis by inducing pVHL function.






Similar content being viewed by others
References
Gunn RM, Hailes HC (2008) Insights into the PI3-K-PKB-mTOR signalling pathway from small molecules. J Chem Biol 1:49–62
Davies MA, Stemke-Hale K, Tellez C, Calderone TL, Deng W, Prieto VG, Lazar AJ, Gershenwald JE, Mills GB (2008) A novel AKT3 mutation in melanoma tumours and cell lines. Br J Cancer 99:1265–1268
Osaki M, Oshimura M, Ito H (2004) PI3K-Akt pathway: its functions and alterations in human cancer. Apoptosis 9:667–676
Nogueira V, Park Y, Chen CC, Xu PZ, Chen ML, Tonic I, Unterman T, Hay N (2008) Akt determines replicative senescence and oxidative or oncogenic premature senescence and sensitizes cells to oxidative apoptosis. Cancer Cell 14:458–470
Turrens JF (2003) Mitochondrial formation of reactive oxygen species. J Physiol 552:335–344
Balaban RS, Nemoto S, Finkel T (2005) Mitochondria, oxidants, and aging. Cell 120:483–495
Chetram MA, Don-Salu-Hewage AS, Hinton CV (2011) ROS enhances CXCR4-mediated functions through inactivation of PTEN in prostate cancer cells. Biochem Biophys Res Commun 410(2):195–200
Wang GL, Jiang BH, Rue EA, Semenza GL (1995) Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci USA 92:5510–5514
Ohh M, Park CW, Ivan M, Hoffman MA, Kim TY, Huang LE, Pavletich N, Chau V, Kaelin WG (2000) Ubiquitination of hypoxia-inducible factor requires direct binding to the beta-domain of the von Hippel–Lindau protein. Nat Cell Biol 2:423–427
Shen C, Kaelin WG, Jr (2012) The VHL/HIF axis in clear cell renal carcinoma. Seminars in Cancer Biology. PMID:22705278, 13 Jun 2012
Parrinello S, Samper E, Krtolica A, Goldstein J, Melov S, Campisi J (2003) Oxygen sensitivity severely limits the replicative lifespan of murine fibroblasts. Nat Cell Biol 5:741–747
Huang HL, Fang LW, Lu SP, Chou CK, Luh TY, Lai MZ (2003) DNA-damaging reagents induce apoptosis through reactive oxygen species-dependent Fas aggregation. Oncogene 22:8168–8177
Massaoka MH, Matsuo AL, Figueiredo CR, Farias CF, Girola N, Arruda DC, Scutti JA, Romoff P, Favero OA, Ferreira MJ et al (2012) Jacaranone induces apoptosis in melanoma cells via ROS-mediated downregulation of Akt and p38 MAPK activation and displays antitumor activity in vivo. PLoS One 7:e38698
Yu CC, Wu PJ, Hsu JL, Ho YF, Hsu LC, Chang YJ, Chang HS, Chen IS, Guh JH (2012) Ardisianone, a natural benzoquinone, efficiently induces apoptosis in human hormone-refractory prostate cancers through mitochondrial damage stress and survivin downregulation. Prostate. doi:10.1002/pros.22548
Chetram MA, Odero-Marah V, Hinton CV (2011) Loss of PTEN permits CXCR4-mediated tumorigenesis through ERK1/2 in prostate cancer cells. Mol Cancer Res 9:90–102
Lee S, Yang K, Kwon J, Lee C, Jeong W, Rhee S (2002) Reversible inactivation of the tumor suppressor PTEN by H2O2. J Biol Chem 277:20336–20342
Maehama T, Dixon JE (1998) The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-trisphosphate. J Biol Chem 273:13375–13378
Wang X, Jiang X (2008) PTEN: a default gate-keeping tumor suppressor with a versatile tail. Cell Res 18:807–816
Cantley LC, Neel BG (1999) New insights into tumor suppression: PTEN suppresses tumor formation by restraining the phosphoinositide 3-kinase/AKT pathway. Proc Natl Acad Sci USA 96:4240–4245
Bortner CD, Cidlowski JA (2002) Apoptotic volume decrease and the incredible shrinking cell. Cell Death Differ 9:1307–1310
Janicke RU, Ng P, Sprengart ML, Porter AG (1998) Caspase-3 is required for alpha-fodrin cleavage but dispensable for cleavage of other death substrates in apoptosis. J Biol chem 273:15540–15545
Li M, Kim WY (2011) Two sides to every story: the HIF-dependent and HIF-independent functions of pVHL. J Cell Mol Med 15:187–195
Nyhan MJ, O’Sullivan GC, McKenna SL (2008) Role of the VHL (von Hippel–Lindau) gene in renal cancer: a multifunctional tumour suppressor. Biochem Soc Trans 36:472–478
Roe JS, Kim HR, Hwang IY, Ha NC, Kim ST, Cho EJ, Youn HD (2011) Phosphorylation of von Hippel–Lindau protein by checkpoint kinase 2 regulates p53 transactivation. Cell Cycle 10:3920–3928
Kumar B, Koul S, Khandrika L, Meacham R, Koul H (2008) Oxidative stress is inherent in prostate cancer cells and is required for aggressive phenotype. Cancer Res 68:1777–1785
Ha HL, Yu DY (2010) HBx-induced reactive oxygen species activates hepatocellular carcinogenesis via dysregulation of PTEN/Akt pathway. World J Gastroenterol 16:4932–4937
Park SE, Park JW, Cho YS, Ryu JH, Paick JS, Chun YS (2007) HIF-1alpha promotes survival of prostate cells at a high zinc environment. Prostate 67:1514–1523
Hergovich A, Lisztwan J, Thoma CR, Wirbelauer C, Barry RE, Krek W (2006) Priming-dependent phosphorylation and regulation of the tumor suppressor pVHL by glycogen synthase kinase 3. Mol Cell Biol 26:5784–5796
Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P, Hemmings BA (1996) Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J 15:6541–6551
Datta SR, Dudek H, Tao X, Masters S, Fu H, Gotoh Y, Greenberg ME (1997) Akt phosphorylation of BAD couples survival signals to the cell-intrinsic death machinery. Cell 91:231–241
Ashcroft M, Ludwig RL, Woods DB, Copeland TD, Weber HO, MacRae EJ, Vousden KH (2002) Phosphorylation of HDM2 by Akt. Oncogene 21:1955–1962
Rena G, Guo S, Cichy SC, Unterman TG, Cohen P (1999) Phosphorylation of the transcription factor forkhead family member FKHR by protein kinase B. J Biol Chem 274:17179–17183
Hinton CV, Fitzgerald LD, Thompson ME (2007) Phosphatidylinositol 3-kinase/Akt signaling enhances nuclear localization and transcriptional activity of BRCA1. Exp Cell Res 313:1735–1744
Maddika S, Panigrahi S, Wiechec E, Wesselborg S, Fischer U, Schulze-Osthoff K, Los M (2009) Unscheduled Akt-triggered activation of cyclin-dependent kinase 2 as a key effector mechanism of apoptin’s anticancer toxicity. Mol Cell Biol 29:1235–1248
Los M, Maddika S, Erb B, Schulze-Osthoff K (2009) Switching Akt: from survival signaling to deadly response. Bioessays 31:492–495
Acknowledgments
Research in this laboratory is supported, in part, by the National Institutes of Health Grants F31CA153908 (MAC), G12RR003062-22 (CVH) and P20MD002285 (CVH and VOM), and the American Association for the Advancement of Science (AAAS) Women’s International Research Collaborations (WIRC) for Minority Serving Institutions (MSIs), a National Science Foundation Grant.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chetram, M.A., Bethea, D.A., Odero-Marah, V.A. et al. ROS-mediated activation of AKT induces apoptosis via pVHL in prostate cancer cells. Mol Cell Biochem 376, 63–71 (2013). https://doi.org/10.1007/s11010-012-1549-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-012-1549-7