Abstract
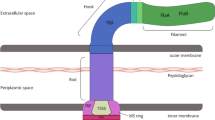
The Cag Type IV secretion apparatus proteins in Helicobacter pylori can mediate the injection of effector CagA protein into eukaryotic target cells. Although this apparatus forms an important pathway for bacterium–host interaction, its assembly process in vivo is poorly understood, and the proteins which contribute to break the bacterial cell walls in Cag-PAI have not yet been identified. The cagγ gene in Cag-PAI is a unique member that contains a conserved SLT catalysis domain, which makes it an attracting question whether cagy gene has the capacity to digest the bacterial cell wall. In the current study, therefore, the cagγ gene was cloned from the H. pylori NCTC 11637 and expressed in Escherichia coli, and its lytic effect on cell walls in vitro was observed. Results indicated that Cagγ protein has a lytic activity against bacterial cell walls. An allelic-exchange mutant (Δcagγ) was further constructed to investigate the relationship between Cagγ and effector CagA translocation. These results suggested that Cagγ contributed to the assembly of Cag Type IV secretion apparatus by digesting the peptidoglycan meshwork of bacterial cell walls.




Similar content being viewed by others
References
Dundon WG, de Bernard M, Montecucco C (2001) Virulence factors of Helicobacter pylori. Int J Med Microbiol 290:647–658
Xiao B, Li W, Guo G et al (2009) Screening and identification of natural antisense transcripts in Helicobacter pylori by a novel approach based on RNase I protection assay. Mol Biol Rep 36:1853–1858
Zhang X, Shen W, Lu Y et al (2010) Expression of UreB and HspA of Helicobacter pylori in silkworm pupae and identification of its immunogenicity. Mol Biol Rep [Epub ahead of print]
Backert S, Ziska E, Brinkmann V et al (2000) Translocation of the Helicobacter pylori CagA protein in gastric epithelial cells by a type IV secretion apparatus. Cell Microbiol 2:155–164
Bourzac KM, Guillemin K (2005) Helicobacter pylori–host cell interactions mediated by type IV secretion. Cell Microbiol 7:911–919
Handa O, Naito Y, Yoshikawa T (2007) CagA protein of Helicobacter pylori: a hijacker of gastric epithelial cell signaling. Biochem Pharmacol 73:1697–1702
Zhang Q, Li Y, Li X et al (2009) PARP-1 Val762Ala polymorphism, CagA + H. pylori infection and risk for gastric cancer in Han Chinese population. Mol Biol Rep 36:1461–1467
Backert S, Meyer TF (2006) Type IV secretion systems and their effectors in bacterial pathogenesis. Curr Opin Microbiol 9:207–217
Juhas M, Crook DW, Hood DW (2008) Type IV secretion systems: tools of bacterial horizontal gene transfer and virulence. Cell Microbiol 10:2377–2386
Demchick P, Koch AL (1996) The permeability of the wall fabric of Escherichia coli and Bacillus subtilis. J Bacteriol 178:768–773
Hoppner C, Liu Z, Domke N, Binns AN, Baron C (2004) VirB1 orthologs from Brucella suis and pKM101 complement defects of the lytic transglycosylase required for efficient type IV secretion from Agrobacterium tumefaciens. J Bacteriol 186:1415–1422
Bayer M, Eferl R, Zellnig G et al (1995) Gene 19 of plasmid R1 is required for both efficient conjugative DNA transfer and bacteriophage R17 infection. J Bacteriol 177:4279–4288
Fullner KJ (1998) Role of Agrobacterium virB genes in transfer of T complexes and RSF1010. J Bacteriol 180:430–434
Hong PC, Tsolis RM, Ficht TA (2000) Identification of genes required for chronic persistence of Brucella abortus in mice. Infect Immun 68:4102–4107
Loessner MJ (2005) Bacteriophage endolysins—current state of research and applications. Curr Opin Microbiol 8:480–487
Kim KM, Lee SG, Park MG et al (2007) Gamma-glutamyltranspeptidase of Helicobacter pylori induces mitochondria-mediated apoptosis in AGS cells. Biochem Biophys Res Commun 355:562–567
Blackburn NT, Clarke AJ (2000) Assay for lytic transglycosylases: a family of peptidoglycan lyases. Anal Biochem 284:388–393
Bendtsen JD, Nielsen H, von Heijne G, Brunak S (2004) Improved prediction of signal peptides: SignalP 3.0. J Mol Biol 340:783–795
Wang J, Xu C (2000) Biochemistry, vol 1, 3rd edn. Higher Education, Beijing, pp 390–392 (in Chinese)
Thunnissen AM, Rozeboom HJ, Kalk KH, Dijkstra BW (1995) Structure of the 70-kDa soluble lytic transglycosylase complexed with bulgecin A. Implications for the enzymatic mechanism. Biochemistry 34:12729–12737
van Asselt EJ, Thunnissen AM, Dijkstra BW (1999) High resolution crystal structures of the Escherichia coli lytic transglycosylase Slt70 and its complex with a peptidoglycan fragment. J Mol Biol 291:877–898
Baron C, Coombes B (2007) Targeting bacterial secretion systems: benefits of disarmament in the microcosm. Infect Disord Drug Targets 7:19–27
Ziedaite G, Daugelavicius R, Bamford JK, Bamford DH (2005) The holin protein of bacteriophage PRD1 forms a pore for small-molecule and endolysin translocation. J Bacteriol 187:5397–5405
Hoppner C, Carle A, Sivanesan D, Hoeppner S, Baron C (2005) The putative lytic transglycosylase VirB1 from Brucella suis interacts with the type IV secretion system core components VirB8, VirB9 and VirB11. Microbiology 151:3469–3482
Zupan J, Hackworth CA, Aguilar J, Ward D, Zambryski P (2007) VirB1* promotes T-pilus formation in the vir-Type IV secretion system of Agrobacterium tumefaciens. J Bacteriol 189:6551–6563
Holtje JV (1996) Lytic transglycosylases. Exs 75:425–429
Reid CW, Blackburn NT, Legaree BA, Auzanneau FI, Clarke AJ (2004) Inhibition of membrane-bound lytic transglycosylase B by NAG-thiazoline. FEBS Lett 574:73–79
Inohara N, Ogura Y, Nunez G (2002) Nods: a family of cytosolic proteins that regulate the host response to pathogens. Curr Opin Microbiol 5:76–80
Viala J, Chaput C, Boneca IG et al (2004) Nod1 responds to peptidoglycan delivered by the Helicobacter pylori cag pathogenicity island. Nat Immunol 5:1166–1174
Liu X, Liang J, Li G (2009) Lipopolysaccharide promotes adhesion and invasion of hepatoma cell lines HepG2 and HepG2.2.15. Mol Biol Rep [Epub ahead of print]
Acknowledgments
We thank Prof. Seung-chul Baik (Gyeongsang National University College of Medicine, Republic of Korea) for kindly providing us the suicide plasmid pBlue-KM40 and technical assistant for mutant construct of H. pylori. This work was supported by grants from the National Natural Science Foundation of China (no. 30870096) and the Natural Science Foundation for college in Jiangsu Province (08KJB310001).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhong, Q., Shao, S., Mu, R. et al. Characterization of peptidoglycan hydrolase in Cag pathogenicity island of Helicobacter pylori . Mol Biol Rep 38, 503–509 (2011). https://doi.org/10.1007/s11033-010-0134-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-010-0134-y