Abstract
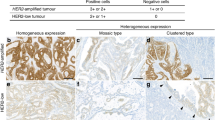
To evaluate the KRAS, BRAF, EGFR, and HER2 gene status in colorectal cancer by novel techniques and evaluate whether anti-HER2 therapies could be offered in the treatment of these patients. There are conflicting data on the prevalence of BRAF mutations and EGFR and HER2 gene amplification in colorectal KRAS wild type patients. In our study we tried to evaluate these expressions and their relationship to future treatment assays. Clinical–pathological data and paraffin-embedded specimens were collected from 186 patients who underwent colorectal resections at General Yagüe Hospital in Burgos, Spain. KRAS and BRAF status was analyzed by real-time PCR in all patients. EGFR and HER2/NEU gene amplification was detected using fluorescent in situ hybridisation technique (FISH) in 38 KRAS and BRAF wild type patients. KRAS mutations were present in 48% of the colorectal cancer patients. BRAF mutations were present in 6.25% of the KRAS wild type patients. EGFR and HER2 gene amplification was observed in 5.3% and 26.3%, respectively, of KRAS and BRAF wild type colorectal cancer patients. HER2, but not EGFR gene amplification, was frequently observed in KRAS and BRAF wild type colorectal cancer patients. These data indicate that HER2 amplification could be one of the genes to be considered in the therapeutic management of colorectal cancer.
Similar content being viewed by others
Abbreviations
- BRAF:
-
BRAF murine sarcoma viral oncogene homolog B1
- KRAS:
-
Human homolog of the Kirsten rat sarcoma-2 virus oncogene
- EGFR:
-
Epidermal growth factor receptor
- FISH:
-
Fluorescence In situ Hybridization
- HER2:
-
Human epidermal growth factor receptor-2
- mCRC:
-
Metastatic colorectal cancer
References
Jemal A et al (2003) Cancer statistics. CA Cancer J Clin 53(1):5–26
Venook AP (2005) Epidermal growth factor receptor-targeted treatment for advanced colorectal carcinoma. Cancer 103(12):2435–2446
Moore MJ et al (2007) Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 25(15):1960–1966
Van Cutsem E et al (2007) Open-label phase III trial of panitumumab plus best supportive care compared with best supportive care alone in patients with chemotherapy-refractory metastatic colorectal cancer. J Clin Oncol 25(13):1658–1664
Hynes NE, Lane HA (2005) ERBB receptors and cancer: the complexity of targeted inhibitors. Nat Rev Cancer 5(5):341–354
Mendelsohn J, Baselga J (2006) Epidermal growth factor receptor targeting in cancer. Semin Oncol 33(4):369–385
Downward J (2003) Targeting RAS signalling pathways in cancer therapy. Nat Rev Cancer 3(1):11–22
Malumbres M, Barbacid M (2003) RAS oncogenes: the first 30 years. Nat Rev Cancer 3(6):459–465
Schubbert S, Shannon K, Bollag G (2007) Hyperactive Ras in developmental disorders and cancer. Nat Rev Cancer 7(4):295–308
Andreyev HJ et al (2001) Kirsten ras mutations in patients with colorectal cancer: the ‘RASCAL II’ study. Br J Cancer 85(5):692–696
Bazan V et al (2002) Specific codon 13 K-ras mutations are predictive of clinical outcome in colorectal cancer patients, whereas codon 12 K-ras mutations are associated with mucinous histotype. Ann Oncol 13(9):1438–1446
Benvenuti S et al (2007) Oncogenic activation of the RAS/RAF signaling pathway impairs the response of metastatic colorectal cancers to anti-epidermal growth factor receptor antibody therapies. Cancer Res 67(6):2643–2648
De Roock W et al (2008) KRAS wild-type state predicts survival and is associated to early radiological response in metastatic colorectal cancer treated with cetuximab. Ann Oncol 19(3):508–515
Di Fiore F et al (2007) Clinical relevance of KRAS mutation detection in metastatic colorectal cancer treated by Cetuximab plus chemotherapy. Br J Cancer 96(8):1166–1169
Lievre A et al (2006) KRAS mutation status is predictive of response to cetuximab therapy in colorectal cancer. Cancer Res 66(8):3992–3995
Adams R, Maughan T (2007) Predicting response to epidermal growth factor receptor-targeted therapy in colorectal cancer. Expert Rev Anticancer Ther 7(4):503–518
Nahta R et al (2006) Mechanisms of disease: understanding resistance to HER2-targeted therapy in human breast cancer. Nat Clin Pract Oncol 3(5):269–280
Lu Y et al (2007) Epidermal growth factor receptor (EGFR) ubiquitination as a mechanism of acquired resistance escaping treatment by the anti-EGFR monoclonal antibody cetuximab. Cancer Res 67(17):8240–8247
Cunningham D et al (2004) Cetuximab monotherapy and cetuximab plus irinotecan in irinotecan-refractory metastatic colorectal cancer. N Engl J Med 351(4):337–345
Chung KY et al (2005) Cetuximab shows activity in colorectal cancer patients with tumors that do not express the epidermal growth factor receptor by immunohistochemistry. J Clin Oncol 23(9):1803–1810
Moroni M et al (2005) Gene copy number for epidermal growth factor receptor (EGFR) and clinical response to antiEGFR treatment in colorectal cancer: a cohort study. Lancet Oncol 6(5):279–286
Dowsett M et al (2009) Disease-free survival according to degree of HER2 amplification for patients treated with adjuvant chemotherapy with or without 1 year of trastuzumab: the HERA Trial. J Clin Oncol 27(18):2962–2969
Paez JG et al (2004) EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 304(5676):1497–1500
Newton CR et al (1989) Analysis of any point mutation in DNA. The amplification refractory mutation system (ARMS). Nucleic Acids Res 17(7):2503–2516
Thelwell N et al (2000) Mode of action and application of Scorpion primers to mutation detection. Nucleic Acids Res 28(19):3752–3761
Whitcombe D et al (1999) Detection of PCR products using self-probing amplicons and fluorescence. Nat Biotechnol 17(8):804–807
Benlloch S et al (2006) Detection of BRAF V600E mutation in colorectal cancer: comparison of automatic sequencing and real-time chemistry methodology. J Mol Diagn 8(5):540–543
Amado RG et al (2008) Wild-type KRAS is required for panitumumab efficacy in patients with metastatic colorectal cancer. J Clin Oncol 26(10):1626–1634
Di Nicolantonio F et al (2008) Wild-type BRAF is required for response to panitumumab or cetuximab in metastatic colorectal cancer. J Clin Oncol 26(35):5705–5712
Kavanagh DO et al (2009) Is overexpression of HER-2 a predictor of prognosis in colorectal cancer? BMC Cancer 9:1
Ooi A et al (2004) Protein overexpression and gene amplification of HER-2 and EGFR in colorectal cancers: an immunohistochemical and fluorescent in situ hybridization study. Mod Pathol 17(8):895–904
Schuell B et al (2006) HER 2/neu protein expression in colorectal cancer. BMC Cancer 6:123
Moroni M et al (2008) EGFR FISH in colorectal cancer: what is the current reality? Lancet Oncol 9(5):402–403
Acknowledgements
We would like to thank Celia Barrrio for her technical help with the FISH analysis and Susana Santamaría for her technical help. MHV is especially thankful to CVP, IHH and AHV, for their support. This work was supported by a grant FIS CA08/00070 and Caja de Burgos.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Herreros-Villanueva, M., Rodrigo, M., Claver, M. et al. KRAS, BRAF, EGFR and HER2 gene status in a Spanish population of colorectal cancer. Mol Biol Rep 38, 1315–1320 (2011). https://doi.org/10.1007/s11033-010-0232-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-010-0232-x