Abstract
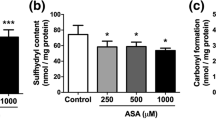
Guanidinoacetate methyltransferase (GAMT) deficiency is an inherited neurometabolic disorder biochemically characterized by tissue accumulation of guanidinoacetate (GAA) and depletion of creatine. Affected patients present epilepsy and mental retardation whose pathogeny is unclear. In the present study we investigated the in vitro and in vivo (intrastriatal administration) effects of GAA on some oxidative stress parameters in rat striatum. Sixty-day-old rats were used for intrastriatal infusion of GAA. For the in vitro studies, 60-day-old Wistar rats were killed by decapitation and the striatum was pre-incubated for 1 h at 37°C in the presence of GAA at final concentrations ranging from 10 to 100 μM. Parameters of oxidative stress such as total radical-trapping antioxidant potential (TRAP), antioxidant enzymes (SOD, GPx, and CAT), protein carbonyl and thiol contents were measured. DNA damage was also evaluated. Results showed that GAA administration (in vivo studies) or the addition of 100 μM GAA to assays (in vitro studies) significantly decreased TRAP, SOD activity, and total thiol levels in rat striatum. In contrast, this guanidino compound did not alter protein carbonyl content and the activities of CAT and GPx. DNA damage was not found after intrastriatal administration of GAA. The data indicate that the metabolite accumulating in GAMT deficiency decreases antioxidant capacity and total thiol content in the striatum. It is therefore presumed that this pathomechanism may contribute at least in part to the pathophysiology of the brain injury observed in patients affected by GAMT deficiency.




Similar content being viewed by others
References
Von Figura K, Hanefeld F, Isbrandt D, Stöckler-Ipsiroglu S (2001) Guanidinoacetate methyltransferase deficiency. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds), The metabolic and molecular bases of inherited disease, 8th edn. McGraw-Hill, New York, pp 1897–1908
Leuzzi V, Bianchi MC, Tosetti M, Carducci C, Cerquiglini CA, Cioni G, Antonozzi I (2000) Brain creatine depletion: guanidinoacetate methyltransferase deficiency (improving with creatine supplementation). Neurology 55:1407–1409
Neu A, Neuhoff H, Trube G, Fehr S, Ullrich K, Roeper J, Isbrandt D (2002) Activation of GABAA receptors by guanidinoacetate: a novel pathophysiological mechanism. Neurobiol Dis 11:298–307
Hiramatsu M, Ohba S, Edamatsu R, Kadowari D, Mori A (1992) Effect of guanidino compounds on membrane fluidity of rats synaptosomes. In: De Deyn PP, Marescau IA, Quereshi SD, Mori A (eds) Guanidino compounds in biology and medicine, vol 1. John Libbey & Company Lta, Guildford, UK, pp 387–393
Schulze A, Ebinger F, Rating D, Mayaetepek E (2001) Improving treatment of guanidinoacetate methyltransferase deficiency: reduction of guanidinoacetic acid in body fluids by arginine restriction and ornithine supplementation. Mol Genet Metab 74:413–419
Schulze A (2003) Creatine deficiency syndromes. Mol Cell Biochem 244:143–150
Sykut-Cegielska J, Gradowska W, Mercimek-Mahmutoglu S, Stockler-Ipsiroglu S (2004) Biochemical and clinical characteristics of creatine deficiency syndromes. Acta Biochim Pol 51:875–882
Halliwell B (2006) Oxidative stress and neurodegeneration: where are we now? J Neurochem 97:1634–1658
Reznick AZ, Packer L (1993) Free radicals and antioxidants in muscular neurological diseases and disorders. In: Poli G, Albano E, Dianzani MU (eds) Free radicals: from basic science to medicine. Birkhäuser, Basel, pp 425–437
Halliwell B (1996) Free radicals, proteins and DNA: oxidative damage versus redox regulation. Biochem Soc Trans 24:1023–1027
Zugno AI, Scherer EBS, Schuck PF, Oliveira DL, Wofchuk S, Wannmacher CMD, Wajner M, Wyse ATS (2006) Intrastriatal administration of guanidinoacetate inhibits Na+,K+-ATPase and creatine kinase activities in rat striatum. Met Brain Dis 21:41–50
Mori A, Kohno M, Masumizu T, Nosa Y, Packer I (1996) Guanidino compounds generate reactive oxygen species. Biochem Mol Biol Int 40:135–143
Zugno AI, Franzon R, Chiarani F, Bavaresco CS, Wannmacher CMD, Wajner M, Wyse ATS (2004) Evaluation of the mechanism underlying the inhibitory effect of guanidinoacetate on brain Na+,K+-ATPase activity. Int J Dev Neurosci 22:191–196
Lissi E, Pascual C, Del Castillo MD (1992) Luminol luminescence induced by 2,2’-azo-bis-(2-amidinopropane) thermolysis. Free Rad Res Commun 17:299–311
Marklund SL (1985) Pyrogallol autoxidation. In: Greenwald RA (ed) Handbook for oxygen radical research. CRC Press, Boca Raton, pp 243–247
Aebi H (1984) Catalase, in vitro. Methods Enzymol 105:121–126
Wendel A (1981) Glutathione peroxidase. Methods Enzymol 77:325–332
Aksenov MY, Markesbery WR (2001) Change in thiol content and expression of glutathione redox system gene in the hippocampus and cerebellum in Alzheimer’s disease. Neurosci Lett 302:141–145
Stadtman EA (1990) Metal ion-catalyzed oxidation of proteins: biochemical mechanism and biological consequences. Free Radic Biol Med 9:315–325
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–267
Tice RR, Agurell E, Anderson D, Burlinson B, Hartmann A, Kobayashi H, Miyamae Y, Rojas E, Ryu JC, Sasaki YF (2000) Single cell gel/comet assay: guidelines for in vitro and in vivo genetic toxicology testing. Environ Mol Mutagen 35:206–221
Collins AR, Ma AG, Duthie SJ (1995) The kinectics of repair of oxidative DNA damage (strand breaks and oxidised pyrimidies) in human cells. Mutat Res 30:69–77
Collins A, Dusinska M, Franklin M, Somorovska M, Petrovska H, Duthie S, Fillion L, Panayiutidis M, Raslova K, Vaughan N (1997) Comet assay in human biomonitoring studies: reliability, validation, and applications. Environ Mol Mutagen 30:139–146
Wajner M, Latini A, Wyse AT, Dutra-Filho CS (2004) The role of oxidative damage in the neuropathology of organic acidurias: insights from animal studies. J Inherit Metab Dis 27:427–448
Matte C, Monteiro SC, Calcagnotto T, Bavaresco CS, Netto CA, Wyse AT (2004) In vivo and in vitro effects of homocysteine on Na+, K+-ATPase activity in parietal, prefrontal and cingulate cortex of young rats. Int J Dev Neurosci 22:185–190
Delwing D, Cornelio AR, Wajner M, Wannmacher CM, Wyse AT (2007) Arginine administration reduces creatine kinase activity in rat cerebellum. Metab Brain Dis 22:13–23
Wyse AT, Stefanello FM, Chiarani F, Delwing D, Wannmacher CM, Wajner M (2004) Arginine administration decreases cerebral cortex acetylcholinesterase and serum butyrylcholinesterase probably by oxidative stress induction. Neurochem Res 29:385–389
Flora SJ (2007) Role of free radicals and antioxidants in health and disease. Cell Mol Biol 53:1–2
Halliwell B (2001) Role of free radicals in the neurodegenerative diseases. Therapeutic implications for antioxidant treatment. Drug Aging 18:685–716
Halliwell B, Gutteridge JC (2007) Free radicals in biology and medicine, 4th edn. Oxford University Press, New York, pp 1–851
Lissy E, Salim-Hanna M, Pascual C, Del Castillo MD (1995) Evaluation of total antioxidant potencial (TRAP) and total reactivity from luminol-enhanced chemiluminescence measurements. Free Rad Med 18:153–158
Evelson P, Travacio M, Repetto M, Escobar J, Llesuy S, Lissi EA (2001) Evaluation of total reactive antioxidant potencial (TRAP) of tissue homogenates and their cytosols. Arch Biochem Biophys 388:261–266
Konorev EA, Kalyanaraman B, Hogg N (2000) Modification of creatine kinase by S-nitrosothiols: S-nitrosation vs. S-thiolation. Free Radic Biol Med 28:1671–1678
Koufen P, Stark G, (2000) Free radical induced inactivation of creatine kinase: sites of interaction, protection, and recovery. Biochim Biophys Acta 1501:44–50
Carageorgio H, Tzotzes V, Pantos C, Mourouzis C, Zarros A, Tsakiris S (2004) In vivo and in vitro effects of cadmium on adult rat brain total antioxidant status, acetylcholinesterase, (Na+, K+)-ATPase and Mg2+-ATPase activities: protection by L-cysteine. Basic Clin Pharmacol Toxicol 94:112–118
Badisa VL, Latinwo LM, Odewumi CO, Ikediobi CO, Badisa RB, Ayuk-Takem LT, Nwoga J, West J (2007) Mechanism of DNA damage by cadmium and interplay of antioxidant enzymes and agents. Environ Toxicol 22:144–151
Tsutsui H, Ide T, Kinigawa S (2006) Mitochondrial oxidative stress, DNA damage, and heart failure. Antioxid Redox Signal 8:1737–1744
Xie J, Fan R, Meng Z (2007) Protein oxidation and DNA-protein crosslink induced by sulfur dioxide in lungs, livers, and hearts from mice. Inhal Toxicol 19:759–765
Frenzilli G, Sarcelli V, Fornai F, Paparelli A, Nigro M (2006) The comet assay as a method of assesment of neurotoxocity: usefulness for drugs of abuse. Ann NY Acad Sci 1074:478–481
Freeman B, Crapo JD (1982) Biology of disease: free radicals and tissue injury. Lab Invest 47:412–426
Lawer JM, Song W (2000) Specificity of antioxidant enzyme inhibition in skeletal muscle to reative nitrogen species donors. Biochem Biophys Res Commun 294:1093–1100
Schulz JB, Lindenau J, Seyfried J, Dichgans J (2000) Gluthatione, oxidative stress and neurodegeneration. Eur J Biochem 267:4904–4911
Hiramatsu M (2003) A role for guanidino compounds in the brain. Mol Cell Biochem 244:57–62
Acknowledgments
This work was supported in part by grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq—Brazil) and by the FINEP Research Grant “Rede Instituto Brasileiro de Neurociência (IBN-Net)—# 01.06.0842-00.”
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zugno, A.I., Stefanello, F.M., Scherer, E.B.S. et al. Guanidinoacetate Decreases Antioxidant Defenses and Total Protein Sulfhydryl Content in Striatum of Rats. Neurochem Res 33, 1804–1810 (2008). https://doi.org/10.1007/s11064-008-9636-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-008-9636-6