Abstract
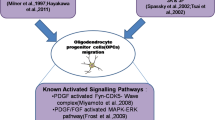
During CNS development, oligodendrocyte progenitor (OP) cells migrate from germinal zones to presumptive white matter tracts to generate myelinating oligodendrocytes. In vitro and in vivo studies indicate that platelet-derived growth factor-A (PDGF-A) is a potent chemoattractant for OP cells and important for normal distribution throughout the developing CNS. However, PDGF-A does not localize in concentration gradients corresponding to OP migratory pathways, as would be expected for a chemoattractant to direct migration. Therefore, the mechanism by which PDGF-A regulates OP distribution remains to be clarified. Here we show that PDGF-A induces OP migration and continuous exposure to PDGF-A is not required to maintain migration. Using pharmacological inhibitors, we show that a self-sustaining extracellular-regulated-kinase signaling pathway drives OP migration for up to 72 hours after the initial PDGF stimulus. These findings indicate PDGF-A may act to mobilize OP cells that then respond to distinct directional signals to distribute appropriately within the CNS.








Similar content being viewed by others
References
Davies JE, Miller RH (2001) Local sonic hedgehog signaling regulates oligodendrocyte precursor appearance in multiple ventricular zone domains in the chick metencephalon. Dev Biol 233(2):513–525
Levison SW, Chuang C, Abramson BJ et al (1993) The migrational patterns and developmental fates of glial precursors in the rat subventricular zone are temporally regulated. Development 119(3):611–622
Jarjour AA, Manitt C, Moore SW et al (2003) Netrin-1 Is a chemorepellent for oligodendrocyte precursor cells in the embryonic spinal cord. J Neurosci 23(9):3735–3744
Tsai HH, Frost E, To V et al (2002) The chemokine receptor CXCR2 controls positioning of oligodendrocyte precursors in developing spinal cord by arresting their migration. Cell 110(3):373–383
Frost E, Kiernan BW, Faissner A et al (1996) Regulation of oligodendrocyte precursor migration by extracellular matrix: evidence for substrate-specific inhibition of migration by tenascin-C. Dev Neurosci 18(4):266–273
Kakita A, Goldman JE (1999) Patterns and dynamics of SVZ cell migration in the postnatal forebrain: monitoring living progenitors in slice preparations. Neuron 23(3):461–472
Calver AR, Hall AC, Yu WP et al (1998) Oligodendrocyte population dynamics and the role of PDGF in vivo. Neuron 20(5):869–882
van Heyningen P, Calver AR, Richardson WD (2001) Control of progenitor cell number by mitogen supply and demand. Curr Biol 11(4):232–241
Fruttiger M, Karlsson L, Hall AC et al (1999) Defective oligodendrocyte development and severe hypomyelination in PDGF-A knockout mice. Development 126(3):457–467
Betsholtz C, Karlsson L, Lindahl P (2001) Developmental roles of platelet-derived growth factors. Bioessays 23(6):494–507
Tsai HH, Tessier-Lavigne M, Miller RH (2003) Netrin 1 mediates spinal cord oligodendrocyte precursor dispersal. Development 130(10):2095–2105
Ebner S, Dunbar M, McKinnon RD (2000) Distinct roles for PI3 K in proliferation and survival of oligodendrocyte progenitor cells. J Neurosci Res 62(3):336–345
Haines JD, Fragoso G, Hossain S et al (2008) p38 Mitogen-activated protein kinase regulates myelination. J Mol Neurosci 35:23–33
Bhalla US, Ram PT, Iyengar R (2002) MAP kinase phosphatase as a locus of flexibility in a mitogen-activated protein kinase signaling network. Science 297(5583):1018–1023
Armstrong RC (1998) Isolation and characterization of immature oligodendrocyte lineage cells. Methods 16(3):282–292
Bottenstein JE, Sato GH (1979) Growth of a rat neuroblastoma cell line in serum-free supplemented medium. Proc Natl Acad Sci USA 76(1):514–517
Frost EE, Milner R, Ffrench-Constant C (2000) Migration assays for oligodendrocyte precursor cells. In: Streuli C, Grant M (eds) Extracellular matrix protocols. Humana Press, Totowa, NJ, pp 265–278
Milner R, Edwards G, Streuli C et al (1996) A role in migration for the alpha V beta 1 integrin expressed on oligodendrocyte precursors. J Neurosci 16(22):7240–7252
Simpson PB, Armstrong RC (1999) Intracellular signals and cytoskeletal elements involved in oligodendrocyte progenitor migration. Glia 26(1):22–35
Frost EE, Nielsen JA, Le TQ et al (2003) PDGF and FGF2 regulate oligodendrocyte progenitor responses to demyelination. J Neurobiol 54(3):457–472
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(5259):680–685
Gallo V, Zhou JM, McBain CJ et al (1996) Oligodendrocyte progenitor cell proliferation and lineage progression are regulated by glutamate receptor-mediated K + channel block. J Neurosci 16(8):2659–2670
Tokumoto YM, Durand B, Raff MC (1999) An analysis of the early events when oligodendrocyte precursor cells are triggered to differentiate by thyroid hormone, retinoic acid, or PDGF withdrawal. Dev Biol 213(2):327–339
Vlahos CJ, Matter WF, Hui KY et al (1994) A specific inhibitor of phosphatidylinositol 3-kinase, 2-(4-morpholinyl)–8-phenyl-4H–1-benzopyran-4-one (LY294002). J Biol Chem 269(7):5241–5248
Pearson G, Robinson F, Beers Gibson T et al (2001) Mitogen-activated protein (MAP) kinase pathways:regulation and physiological functions. Endocr Rev 22(2):153–183
Alessi DR, Cuenda A, Cohen P et al (1995) PD 098059 is a specific inhibitor of the activation of mitogen-activated protein kinase kinase in vitro and in vivo. J Biol Chem 270(46):27489–27494
Newton R, Cambridge L, Hart LA et al (2000) The MAP kinase inhibitors, PD098059, UO126 and SB203580, inhibit IL-1beta-dependent PGE(2) release via mechanistically distinct processes. Br J Pharmacol 130(6):1353–1361
Larocca JN, Almazan G (1997) Acetylcholine agonists stimulate mitogen-activated protein kinase in oligodendrocyte progenitors by muscarinic receptors. J Neurosci Res 50(5):743–754
Favata MF, Horiuchi KY, Manos EJ et al (1998) Identification of a novel inhibitor of mitogen-activated protein kinase. J Biol Chem 273(29):18623–18632
Young PR, McLaughlin MM, Kumar S et al (1997) Pyridinyl imidazole inhibitors of p38 mitogen-activated protein kinase bind in the ATP site. J Biol Chem 272(18):12116–12121
Coffey ET, Smiciene G, Hongisto V et al (2002) c-Jun N-terminal protein kinase (jNK) 2/3 is specifically activated by stress, mediating c-Jun activation, in the presence of constitutive JNK1 activity in cerebellar neurons. J Neurosci 22(11):4335–4345
Nemenoff RA, Winitz S, Qian NX et al (1993) Phosphorylation and activation of a high molecular weight form of phospholipase A2 by p42 microtubule-associated protein 2 kinase and protein kinase C. J Biol Chem 268(3):1960–1964
Armstrong RC, Harvath L, Dubois-Dalcq ME (1990) Type 1 astrocytes and oligodendrocyte-type 2 astrocyte glial progenitors migrate toward distinct molecules. J Neurosci Res 27(3):400–407
Milner R, Anderson HJ, Rippon RF et al (1997) Contrasting effects of mitogenic growth factors on oligodendrocyte precursor cell migration. Glia 19(1):85–90
Rebay I (2002) Keeping the receptor tyrosine kinase signaling pathway in check: lessons from Drosophila. Dev Biol 251(1):1–17
Stork PJ (2002) ERK signaling: duration, duration, duration. Cell Cycle 1(5):315–317
Heldin CH, Ostman A, Ronnstrand L (1998) Signal transduction via platelet-derived growth factor receptors. Biochim Biophys Acta 1378(1):F79–113
Budihardjo I, Oliver H, Lutter M et al (1999) Biochemical pathways of caspase activation during apoptosis. Annu Rev Cell Dev Biol 15(1):269–290
Baron W, Shattil SJ, Ffrench-Constant C (2002) The oligodendrocyte precursor mitogen PDGF stimulates proliferation by activation of {alpha}v{beta}3 integrins. EMBO J. 21(8):1957–1966
McKinnon RD, Waldron S, Kiel ME (2005) PDGF alpha-receptor signal strength controls an RTK rheostat that integrates phosphoinositol 3’-kinase and phospholipase Cgamma pathways during oligodendrocyte maturation. J Neurosci 25(14):3499–3508
Heldin CH, Westermark B (1999) Mechanism of action and in vivo role of platelet-derived growth factor. Physiol Rev 79(4):1283–1316
Vemuri GS, McMorris FA (1996) Oligodendrocytes and their precursors require phosphatidylinositol 3-kinase signaling for survival. Development 122(8):2529–2537
Kato T, Ohtani-Kaneko R, Ono K, Okado N, Shiga T (2005) Developmental regulation of activated ERK expression in the spinal cord and dorsal root ganglion of the chick embryo. Neurosci Res 52(1):11–19
Zhou Q, Anderson DJ (2002) The bHLH transcription factors OLIG2 and OLIG1 couple neuronal and glial subtype specification. Cell 109(1):61–73
Masahira N, Takebayashi H, Ono K et al (2006) Olig2-positive progenitors in the embryonic spinal cord give rise not only to motoneurons and oligodendrocytes, but also to a subset of astrocytes and ependymal cells. Dev Biol 293(2):358–369
Bhat NR, Zhang P (1996) Activation of mitogen-activated protein kinases in oligodendrocytes. J Neurochem 66(5):1986–1994
Nishizuka Y (1992) Intracellular signaling by hydrolysis of phospholipids and activation of protein kinase C. Science 258(5082):607–614
Bhalla US, Iyengar R (1999) Emergent properties of networks of biological signaling pathways. Science 283(5400):381–387
Acknowledgements
This work was supported by National Institutes of Health Grant NS39293 (RCA), National Multiple Sclerosis Society Grant RG3515. (RCA), the Manitoba Children’s Hospital Foundation (EEF) and the Manitoba Health Research Council (EEF). We thank Dr. Joel Levine for reagents, Dr. Mary-Lou Cutler for reagents and helpful discussions. Jan Mustapha for technical assistance, and Dr. Katharine Blaschuk for helpful discussion and critical reading of the manuscript. Mrs. Nina Kelly for administrative assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special issue article in honor of Dr. George DeVries.
Rights and permissions
About this article
Cite this article
Frost, E.E., Zhou, Z., Krasnesky, K. et al. Initiation of Oligodendrocyte Progenitor Cell Migration by a PDGF-A Activated Extracellular Regulated Kinase (ERK) Signaling Pathway. Neurochem Res 34, 169–181 (2009). https://doi.org/10.1007/s11064-008-9748-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-008-9748-z