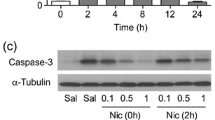
Abstract
Previous studies indicated that ethanol-induced neurodegeneration in postnatal day 7 (P7) mice, widely used as a model for the fetal alcohol spectrum disorders, was accompanied by glycogen synthase kinase-3β (GSK-3β) and caspase-3 activation. Presently, we examined whether tau, a microtubule associated protein, is modified by GSK-3β and caspase-3 in ethanol-treated P7 mouse forebrains. We found that ethanol increased phosphorylated tau recognized by the paired helical filament (PHF)-1 antibody and by the antibody against tau phosphorylated at Ser199. Ethanol also generated tau fragments recognized by an antibody against caspase-cleaved tau (C-tau). C-tau was localized in neurons bearing activated caspase-3 and fragmented nuclei. Over time, cell debris and degenerated projections containing C-tau appeared to be engulfed by activated microglia. A caspase-3 inhibitor partially blocked C-tau formation. Lithium, a GSK-3β inhibitor, blocked ethanol-induced caspase-3 activation, phosphorylated tau elevation, C-tau formation, and microglial activation. These results indicate that tau is phosphorylated by GSK-3β and cleaved by caspase-3 during ethanol-induced neurodegeneration in the developing brain.




Similar content being viewed by others
References
Brion JP, Octave JN, Couck AM (1994) Distribution of the phosphorylated microtubule-associated protein tau in developing cortical neurons. Neuroscience 63:895–909
Canu N, Dus L, Barbato C, Ciotti MT, Brancolini C, Rinaldi AM, Novak M, Cattaneo A, Bradbury A, Calissano P (1998) Tau cleavage and dephosphorylation in cerebellar granule neurons undergoing apoptosis. J Neurosci 18:7061–7074
Carloni S, Mazzoni E, Balduini W (2004) Caspase-3 and calpain activities after acute and repeated ethanol administration during the rat brain growth spurt. J Neurochem 89:197–203
Chakraborty G, Saito M, Mao RF, Wang R, Vadasz C, Saito M (2008) Lithium blocks ethanol-induced modulation of protein kinases in the developing brain. Biochem Biophys Res Commun 367:597–602
Chun W, Waldo GS, Johnson GV (2007) Split GFP complementation assay: a novel approach to quantitatively measure aggregation of tau in situ: effects of GSK3beta activation and caspase 3 cleavage. J Neurochem 103:2529–2539
Chung CW, Song YH, Kim IK, Yoon WJ, Ryu BR, Jo DG, Woo HN, Kwon YK, Kim HH, Gwag BJ, Mook-Jung IH, Jung YK (2001) Proapoptotic effects of tau cleavage product generated by caspase-3. Neurobiol Dis 8:162–172
El-Khodor BF, Burke RE (2002) Medial forebrain bundle axotomy during development induces apoptosis in dopamine neurons of the substantia nigra and activation of caspases in their degenerating axons. J Comp Neurol 452:65–79
Fasulo L, Ugolini G, Cattaneo A (2005) Apoptotic effect of caspase-3 cleaved tau in hippocampal neurons and its potentiation by tau FTDP-mutation N279 K. J Alzheimers Dis 7:3–13
Gabbita SP, Scheff SW, Menard RM, Roberts K, Fugaccia I, Zemlan FP (2005) Cleaved-tau: a biomarker of neuronal damage after traumatic brain injury. J Neurotrauma 22:83–94
Gamblin TC, Chen F, Zambrano A, Abraha A, Lagalwar S, Guillozet AL, Lu M, Fu Y, Garcia-Sierra F, LaPointe N, Miller R, Berry RW, Binder LI, Cryns VL (2003) Caspase cleavage of tau: linking amyloid and neurofibrillary tangles in Alzheimer’s disease. Proc Natl Acad Sci USA 100:10032–10037
Gendron TF, McCartney S, Causevic E, Ko LW, Yen SH (2008) Ethanol enhances tau accumulation in neuroblastoma cells that inducibly express tau. Neurosci Lett 443:67–71
Goedert M, Jakes R (1990) Expression of separate isoforms of human tau protein: correlation with the tau pattern in brain and effects on tubulin polymerization. EMBO J 9:4225–4230
Guo H, Albrecht S, Bourdeau M, Petzke T, Bergeron C, LeBlanc AC (2004) Active caspase-6 and caspase-6-cleaved tau in neuropil threads, neuritic plaques, and neurofibrillary tangles of Alzheimer’s disease. Am J Pathol 165:523–531
Han JY, Jeong JY, Lee YK, Roh GS, Kim HJ, Kang SS, Cho GJ, Choi WS (2006) Suppression of survival kinases and activation of JNK mediate ethanol-induced cell death in the developing rat brain. Neurosci Lett 398:113–117
Ikonomidou C, Bittigau P, Ishimaru MJ, Wozniak DF, Koch C, Genz K, Price MT, Stefovska V, Horster F, Tenkova T, Dikranian K, Olney JW (2000) Ethanol-induced apoptotic neurodegeneration and fetal alcohol syndrome. Science 287:1056–1060
Ishiguro K, Sato K, Takamatsu M, Park J, Uchida T, Imahori K (1995) Analysis of phosphorylation of tau with antibodies specific for phosphorylation sites. Neurosci Lett 202:81–84
Ieraci A, Herrera DG (2006) Nicontinamide protects against ethanol-induced apoptotic neurodegeneration in the developing mouse brain. PLoS Med 3(4):e101
Kaur C, Dheen ST, Ling EA (2007) From blood to brain: amoeboid microglial cell, a nascent macrophage and its functions in developing brain. Acta Pharmacol Sin 28:10087–11096
Kadowaki T, Nakadate K, Sakakibara S, Hirata K, Ueda S (2007) Expression of Iba1 protein in microglial cells of zitter mutant rat. Neurosci Lett 411:26–31
Kim D, Su J, Cotman CW (1999) Sequence of neurodegeneration and accumulation of phosphorylated tau in cultured neurons after okadaic acid treatment. Brain Res 839:253–262
Morishima-Kawashima M, Hasegawa M, Takio K, Suzuki M, Yoshida H, Watanabe A, Titani K, Ihara Y (1995) Hyperphosphorylation of tau in PHF. Neurobiol Aging 16:365–371 discussion 371–380
Nikolaev A, McLaughlin T, O’Leary DDM, Tessier-Lavigne M (2009) APP binds DR6 to trigger axon pruning and neuron death via distinct caspases. Nature 457:981–989
Noble W, Planel E, Zehr C, Olm V, Meyerson J, Suleman F, Gaynor K, Wang L, LaFrancois J, Feinstein B, Burns M, Krishnamurthy P, Wen Y, Bhat R, Lewis J, Dickson D, Duff K (2005) Inhibition of glycogen synthase kinase-3 by lithium correlates with reduced tauopathy and degeneration in vivo. Proc Natl Acad Sci USA 102:6990–6995
Olney JW, Tenkova T, Dikranian K, Muglia LJ, Jermakowicz WJ, D’Sa C, Roth KA (2002) Ethanol-induced caspase-3 activation in the in vivo developing mouse brain. Neurobiol Dis 9:205–219
Olney JW, Tenkova T, Dikranian K, Qin YQ, Labruyere J, Ikonomidou C (2002) Ethanol-induced apoptotic neurodegeneration in the developing C57BL/6 mouse brain. Brain Res Dev Brain Res 133:115–126
Park SY, Ferreira A (2005) The generation of a 17 kDa neurotoxic fragment: an alternative mechanism by which tau mediates beta-amyloid-induced neurodegeneration. J Neurosci 25:5365–5375
Park SY, Tournell C, Sinjoanu RC, Ferreira A (2007) Caspase-3- and calpain-mediated tau cleavage are differentially prevented by estrogen and testosterone in beta-amyloid-treated hippocampal neurons. Neuroscience 144:119–127
Rami A, Jansen S, Giesser I, Winckler J (2003) Post-ischemic activation of caspase-3 in the rat hippocampus: evidence of an axonal and dendritic localisation. Neurochem Int 43:211–223
Rissman RA, Poon WW, Blurton-Jones M, Oddo S, Torp R, Vitek MP, LaFerla FM, Rohn TT, Cotman CW (2004) Caspase-cleavage of tau is an early event in Alzheimer disease tangle pathology. J Clin Invest 114:121–130
Sadakata T, Washida M, Iwayama Y, Shoji S, Sato Y, Ohkura T, Katoh-Semba R, Nakajima M, Sekine Y, Tanaka M, Nakamura K, Iwata Y, Tsuchiya KJ, Mori N, Detera-Wadleigh SD, Ichikawa H, Itohara S, Yoshikawa T, Furuichi T (2007) Autistic-like phenotypes in Cadps2-knockout mice and aberrant CADPS2 splicing in autistic patients. J Clin Invest 117(4):931–943
Saito M, Saito M, Berg MJ, Guidotti A, Marks N (1999) Gangliosides attenuate ethanol-induced apoptosis in rat cerebellar granule neurons. Neurochem Res 24:1107–1115
Saito M, Mao RF, Wang R, Vadasz C, Saito M (2007) Effects of gangliosides on ethanol-induced neurodegeneration in the developing mouse brain. Alcohol Clin Exp Res 31:665–674
Sang H, Lu Z, Li Y, Ru B, Wang W, Chen J (2001) Phosphorylation of tau by glycogen synthase kinase 3beta in intact mammalian cells influences the stability of microtubules. Neurosci Lett 312:141–144
Takadera T, Ohyashiki T (2004) Glycogen synthase kinase-3 inhibitors prevent caspase-dependent apoptosis induced by ethanol in cultured rat cortical neurons. Eur J Pharmacol 499:239–245
Takahashi M, Tomizawa K, Ishiguro K (2000) Distribution of tau protein kinase I/glycogen synthase kinase-3beta, phosphatases 2A and 2B, and phosphorylated tau in the developing rat brain. Brain Res 857:193–206
Tashiro K, Hasegawa M, Ihara Y, Iwatsubo T (1997) Somatodendritic localization of phosphorylated tau in neonatal and adult rat cerebral cortex. Neuroreport 8:2797–2801
Utton MA, Vandecandelaere A, Wagner U, Reynolds CH, Gibb GM, Miller CC, Bayley PM, Anderton BH (1997) Phosphorylation of tau by glycogen synthase kinase 3beta affects the ability of tau to promote microtubule self-assembly. Biochem J 323(Pt 3):741–747
Young C, Klocke BJ, Tenkova T, Choi J, Labruyere J, Qin YQ, Holtzman DM, Roth KA, Olney JW (2003) Ethanol-induced neuronal apoptosis in vivo requires BAX in the developing mouse brain. Cell Death Differ 10:1148–1155
Zemlan FP, Mulchahey JJ, Gudelsky GA (2003) Quantification and localization of kainic acid-induced neurotoxicity employing a new biomarker of cell death: cleaved microtubule-associated protein-tau (C-tau). Neuroscience 121:399–409
Zemlan FP, Jauch EC, Mulchahey JJ, Gabbita SP, Rosenberg WS, Speciale SG, Zuccarello M (2002) C-tau biomarker of neuronal damage in severe brain injured patients: association with elevated intracranial pressure and clinical outcome. Brain Res 947:131–139
Zhang Q, Zhang X, Chen J, Miao Y, Sun A (2009) Role of caspase-3 in tau truncation at D421 is restricted in transgenic mouse models for tauopathies. J Neurochem 109:476–484
Zhong J, Yang X, Yao W, Lee W (2006) Lithium protects ethanol-induced neuronal apoptosis. Biochem Biophys Res Commun 350:905–910
Acknowledgments
We thank Dr. Peter Davies (Albert Einstein College of Medicine, New York, NY) for providing the PHF-1 antibody. This work was supported by National Institute on Alcohol Abuse and Alcoholism grant AA015355.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saito, M., Chakraborty, G., Mao, RF. et al. Tau Phosphorylation and Cleavage in Ethanol-Induced Neurodegeneration in the Developing Mouse Brain. Neurochem Res 35, 651–659 (2010). https://doi.org/10.1007/s11064-009-0116-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-009-0116-4