Abstract
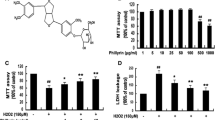
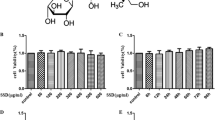
Oxidative stress is a major component of harmful cascades activated in neurodegenerative disorders. We sought to elucidate possible effects of forsythiaside, an active constituent isolated from the Chinese medicinal herb Forsythia suspense, on hydrogen peroxide (H2O2)-induced cell death and to determine the underlying molecular mechanisms in neuron-like PC12 cells. We found that forsythiaside treatment effectively protected PC12 cells against H2O2-induced cell damage and apoptosis. H2O2 exposure induced oxidative stress in PC12 cells, as revealed by increased ROS and lipid peroxidation (MDA), which were inhibited by forsythiaside pretreatment. Increased Bax/Bcl-2 ratio, mitochondrial membrane potential decrease, cytochrome c release, caspase-9/-3 activation, AIF/Endo G translocation were observed in H2O2-treated cells. Interestingly, forsythiaside effectively prevented these events. These results suggested that forsythiaside prevented H2O2-induced mitochondria-dependent apoptosis. Further, increased nuclear levels of Nrf2 and up-regulation of antioxidant enzymes (Mn/SOD and CAT) were detected in forsythiaside-treated cells, indicating the anti-oxidative effects of forsythiaside might be associated with activation of Nrf2 pathway. Moreover, forsythiaside was proved to be effective to prevent LPS-induced cell death and ROS generation. In conclusion, forsythiaside effectively inhibited H2O2-induced oxidative stress and subsequent apoptosis activation.







Similar content being viewed by others
References
Al-Ayadhi LY (2004) Oxidative stress and neurodegenerative disease. Neurosciences (Riyadh) 9:19–23
Zawia NH, Lahiri DK, Cardozo-Pelaez F (2009) Epigenetics, oxidative stress, and Alzheimer disease. Free Radic Biol Med 46:1241–1249
Simon HU, Haj-Yehia A, Levi-Schaffer F (2000) Role of reactive oxygen species (ROS) in apoptosis induction. Apoptosis 5:415–418
Leuner K, Pantel J, Frey C, Schindowski K, Schulz K, Wegat T, Maurer K, Eckert A, Muller WE (2007) Enhanced apoptosis, oxidative stress and mitochondrial dysfunction in lymphocytes as potential biomarkers for Alzheimer’s disease. J Neural Transm Suppl 72:207–215
Turunc Bayrakdar E, Uyanikgil Y, Kanit L, Koylu E, Yalcin A (2014) Nicotinamide treatment reduces the levels of oxidative stress, apoptosis, and PARP-1 activity in Abeta(1-42)-induced rat model of Alzheimer’s disease. Free Radic Res 48:146–158
Halliwell B, Aruoma OI (1991) DNA damage by oxygen-derived species. Its mechanism and measurement in mammalian systems. FEBS Lett 281:9–19
Heilmann J, Merfort I, Weiss M (1995) Radical scavenger activity of different 3′,4′-dihydroxyflavonols and 1,5-dicaffeoylquinic acid studied by inhibition of chemiluminescence. Planta Med 61:435–438
Wang HM, Wang LW, Liu XM, Li CL, Xu SP, Farooq AD (2013) Neuroprotective effects of forsythiaside on learning and memory deficits in senescence-accelerated mouse prone (SAMP8) mice. Pharmacol Biochem Behav 105:134–141
Qu H, Zhang Y, Wang Y, Li B, Sun W (2008) Antioxidant and antibacterial activity of two compounds (forsythiaside and forsythin) isolated from Forsythia suspensa. J Pharm Pharmacol 60:261–266
Cheng G, Zhao Y, Li H, Wu Y, Li X, Han Q, Dai C, Li Y (2014) Forsythiaside attenuates lipopolysaccharide-induced inflammatory responses in the bursa of Fabricius of chickens by downregulating the NF-kappaB signaling pathway. Exp Ther Med 7:179–184
Zhou L, Yang H, Ai Y, Xie Y, Fu Y (2014) [Protective effect of forsythiaside A on acute lung injure induced by lipopolysaccharide in mice], Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 30:151–154
Kim JM, Kim S, Kim DH, Lee CH, Park SJ, Jung JW, Ko KH, Cheong JH, Lee SH, Ryu JH (2011) Neuroprotective effect of forsythiaside against transient cerebral global ischemia in gerbil. Eur J Pharmacol 660:326–333
Naoi M, Maruyama W, Yi H, Inaba K, Akao Y, Shamoto-Nagai M (2009) Mitochondria in neurodegenerative disorders: regulation of the redox state and death signaling leading to neuronal death and survival. J Neural Transm 116:1371–1381
Satoh T, Sakai N, Enokido Y, Uchiyama Y, Hatanaka H (1996) Free radical-independent protection by nerve growth factor and Bcl-2 of PC12 cells from hydrogen peroxide-triggered apoptosis. J Biochem 120:540–546
Jang JH, Surh YJ (2001) Protective effects of resveratrol on hydrogen peroxide-induced apoptosis in rat pheochromocytoma (PC12) cells. Mutat Res 496:181–190
Benedi J, Arroyo R, Romero C, Martin-Aragon S, Villar AM (2004) Antioxidant properties and protective effects of a standardized extract of hypericum perforatum on hydrogen peroxide-induced oxidative damage in PC12 cells. Life Sci 75:1263–1276
Hong H, Liu GQ (2004) Protection against hydrogen peroxide-induced cytotoxicity in PC12 cells by scutellarin. Life Sci 74:2959–2973
Martinou JC, Youle RJ (2011) Mitochondria in apoptosis: Bcl-2 family members and mitochondrial dynamics. Dev Cell 21:92–101
Jacotot E, Costantini P, Laboureau E, Zamzami N, Susin SA, Kroemer G (1999) Mitochondrial membrane permeabilization during the apoptotic process. Ann NY Acad Sci 887:18–30
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES, Wang X (1997) Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91:479–489
Earnshaw WC, Martins LM, Kaufmann SH (1999) Mammalian caspases: structure, activation, substrates, and functions during apoptosis. Annu Rev Biochem 68:383–424
Tu WC, Wu CC, Hsieh HL, Chen CY, Hsu SL (2008) Honeybee venom induces calcium-dependent but caspase-independent apoptotic cell death in human melanoma A2058 cells. Toxicon 52:318–329
Vomhof-Dekrey EE, Picklo MJ Sr (2012) The Nrf2-antioxidant response element pathway: a target for regulating energy metabolism. J Nutr Biochem 23:1201–1206
Um HC, Jang JH, Kim DH, Lee C, Surh YJ (2011) Nitric oxide activates Nrf2 through S-nitrosylation of Keap1 in PC12 cells. Nitric Oxide 25:161–168
Li HY, Wu SY, Shi N (2007) Transcription factor Nrf2 activation by deltamethrin in PC12 cells: involvement of ROS. Toxicol Lett 171:87–98
Boesch-Saadatmandi C, Loboda A, Jozkowicz A, Huebbe P, Blank R, Wolffram S, Dulak J, Rimbach G (2008) Effect of ochratoxin A on redox-regulated transcription factors, antioxidant enzymes and glutathione-S-transferase in cultured kidney tubulus cells. Food Chem Toxicol 46:2665–2671
Li YX, Peng C, Zhang RQ, Li X, Jiang XH (2009) Pharmacokinetics of phillyrin and forsythiaside following iv administration to Beagle dog. Eur J Drug Metab Pharmacokinet 34:79–83
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Huang, C., Lin, Y., Su, H. et al. Forsythiaside Protects Against Hydrogen Peroxide-Induced Oxidative Stress and Apoptosis in PC12 Cell. Neurochem Res 40, 27–35 (2015). https://doi.org/10.1007/s11064-014-1461-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-014-1461-5