Abstract
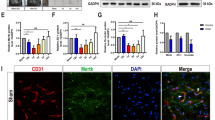
NF-κB is involved in the activation of microglia, which induces secondary spinal cord injury (SCI). This process involves the activation of NF-κB signaling pathway by TRAF6 through its polyubiquitination function. We know that deubiquitination of TRAF6 mediated by deubiquitinating enzyme (DUB) significantly inhibits activation of NF-κB pathway. The ubiquitin-specific protease 4 (USP4) belongs to the deubiquitinase family. Therefore, we hypothesize that USP4 is involved in the microglial activation and subsequent neuronal inflammation after SCI. In this study, we examined the expression and the role of USP4 after SCI. Western blot analysis showed that the expression of USP4 was downregulated and the expression of p-p65 was upregulated in the spinal cord after SCI. Immunohistochemical and immunofluorescence staining showed that USP4 was expressed in microglia but its expression decreased after SCI. In vitro LPS-induced activation of microglia showed decreased expression of USP4 and increased expression of p-p65 and TRAF6. USP4 silencing in LPS-induced activation of microglia promoted the expression of p-p65 and TRAF6 and the secretion of TNF-α and IL-1β. In conclusion, our study provides the first evidence that in microglial cells expression of USP4 decreases after SCI in rats. The downregulation of USP4 expression may promote microglial activation and subsequent neuronal inflammation through NF-κB by attenuating the deubiquitination of TRAF6. This mechanism is of great significance in the pathophysiology of secondary SCI.







Similar content being viewed by others
Abbreviations
- USP4:
-
Ubiquitin specific protease 4
- SCI:
-
Spinal cord injury
- CNS:
-
Central nervous system
- SD:
-
Sprague–Dawley
- NeuN:
-
Neuronal nuclear antigen
- GFAP:
-
Glial fibrillary acidic protein
- Iba-1:
-
Ionized calcium-binding adapter molecule-1
- LPS:
-
Lipopolysaccharide
- siRNA:
-
Short interferingRNA
References
Huang S, Liu X, Zhang J, Bao G, Xu G, Sun Y, Shen Q, Lian M, Huang Y, Cui Z (2015) Expression of peroxiredoxin 1 after traumatic spinal cord injury in rats. Cell Mol Neurobiol 35(8):1217–1226. doi:10.1007/s10571-015-0214-6
Ren J, Mao X, Chen M, Zhang W, Liu Y, Duan C, Zhang H, Sun C, Wu W, Zhu X, Ge J, Tao W, Wang Y, Lu H (2015) TCTP expression after rat spinal cord injury: implications for astrocyte proliferation and migration. J Mol Neurosci 57(3):366–375. doi:10.1007/s12031-015-0628-0
Sminkey L (2013) Spinal cord injury: as many as 500 000 people suffer each year. http://www.who.int/mediacentre/news/releases/2013/spinal-cord-injury-20131202/en/.
Sawada M, Kato K, Kunieda T, Mikuni N, Miyamoto S, Onoe H, Isa T, Nishimura Y (2015) Function of the nucleus accumbens in motor control during recovery after spinal cord injury. Science 350(6256):98–101. doi:10.1126/science.aab3825
Ambrozaitis KV, Kontautas E, Spakauskas B, Vaitkaitis D (2006) Pathophysiology of acute spinal cord injury. Medicina 42(3):255–261
Liu G, Fan G, Guo G, Kang W, Wang D, Xu B, Zhao J (2016) FK506 attenuates the inflammation in rat spinal cord injury by inhibiting the activation of NF-kappaB in microglia cells. Cell Mol Neurobiol. doi:10.1007/s10571-016-0422-8
Raha S, Lee HJ, Yumnam S, Hong GE, Saralamma VVG, Ha YL, Kim JO, Kim YS, Heo JD, Lee SJ, Kim EH, Kim GS (2016) Vitamin D2 suppresses amyloid-beta 25–35 induced microglial activation in BV2 cells by blocking the NF-kappaB inflammatory signaling pathway. Life Sci 161:37–44. doi:10.1016/j.lfs.2016.07.017
Yaguchi M, Ohta S, Toyama Y, Kawakami Y, Toda M (2008) Functional recovery after spinal cord injury in mice through activation of microglia and dendritic cells after IL-12 administration. J Neurosci Res 86(9):1972–1980. doi:10.1002/jnr.21658
Stirling DP, Cummins K, Mishra M, Teo W, Yong VW, Stys P (2014) Toll-like receptor 2-mediated alternative activation of microglia is protective after spinal cord injury. Brain 137(Pt 3):707–723. doi:10.1093/brain/awt341
Xiao N, Li H, Luo J, Wang R, Chen H, Chen J, Wang P (2012) Ubiquitin-specific protease 4 (USP4) targets TRAF2 and TRAF6 for deubiquitination and inhibits TNFalpha-induced cancer cell migration. Biochem J 441(3):979–986. doi:10.1042/bj20111358
Hadweh P, Habelhah H, Kieff E, Mosialos G, Hatzivassiliou E (2014) The PP4R1 subunit of protein phosphatase PP4 targets TRAF2 and TRAF6 to mediate inhibition of NF-kappaB activation. Cell Signal 26(12):2730–2737. doi:10.1016/j.cellsig.2014.08.001
Martinez-Forero I, Rouzaut A, Palazon A, Dubrot J, Melero I (2009) Lysine 63 polyubiquitination in immunotherapy and in cancer-promoting inflammation. Clin Cancer Res 15(22):6751–6757. doi:10.1158/1078-0432.ccr-09-1225
Yasunaga J, Lin FC, Lu X, Jeang KT (2011) Ubiquitin-specific peptidase 20 targets TRAF6 and human T cell leukemia virus type 1 tax to negatively regulate NF-kappaB signaling. J Virol 85(13):6212–6219. doi:10.1128/jvi.00079-11
Trompouki E, Hatzivassiliou E, Tsichritzis T, Farmer H, Ashworth A, Mosialos G (2003) CYLD is a deubiquitinating enzyme that negatively regulates NF-kappaB activation by TNFR family members. Nature 424(6950):793–796. doi:10.1038/nature01803
Wijnhoven P, Konietzny R, Blackford AN, Travers J, Kessler BM, Nishi R, Jackson SP (2015) USP4 auto-deubiquitylation promotes homologous recombination. Mol Cell 60(3):362–373. doi:10.1016/j.molcel.2015.09.019
Zhang J, Cui Z, Feng G, Bao G, Xu G, Sun Y, Wang L, Chen J, Jin H, Liu J, Yang L, Li W (2015) RBM5 and p53 expression after rat spinal cord injury: implications for neuronal apoptosis. Int J Biochem Cell Biol 60:43–52. doi:10.1016/j.biocel.2014.12.020
Baskar Jesudasan SJ, Todd KG, Winship IR (2014) Reduced inflammatory phenotype in microglia derived from neonatal rat spinal cord versus brain. PLoS ONE 9(6):e99443. doi:10.1371/journal.pone.0099443
Nikodemova M, Watters JJ (2012) Efficient isolation of live microglia with preserved phenotypes from adult mouse brain. J Neuroinflamm 9:147. doi:10.1186/1742-2094-9-147
Stein DM, Pineda JA, Roddy V, Knight WAt (2015) Emergency neurological life support: traumatic spine injury. Neurocrit Care 23(Suppl 2):S155–S164. doi:10.1007/s12028-015-0169-y
Kingwell K (2011) A new approach to respiratory recovery after spinal cord injury? Nat Rev Neurol 7(9):473. doi:10.1038/nrneurol.2011.124
Jin X, Yamashita T (2016) Microglia in central nervous system repair after injury. J Biochem 159(5):491–496. doi:10.1093/jb/mvw009
Liu N, Zang KK, Zhang YQ (2015) Activation of microglia and astrocytes in different spinal segments after peripheral nerve injury in mice. Sheng Li Xue Bao 67(6):571–582
Antar V, Baran O, Yuceli S, Erdogan H, Altintas O, Eryigit Baran G, Tasdemiroglu E (2015) Assessment of the neuroprotective effects of the acetylcholinesterase inhibitor Huperzine A in an experimental spinal cord trauma model. J Neurosurg Sci
Cartier N, Lewis CA, Zhang R, Rossi FM (2014) The role of microglia in human disease: therapeutic tool or target? Acta Neuropathol 128(3):363–380. doi:10.1007/s00401-014-1330-y
Ziv Y, Avidan H, Pluchino S, Martino G, Schwartz M (2006) Synergy between immune cells and adult neural stem/progenitor cells promotes functional recovery from spinal cord injury. Proc Natl Acad Sci USA 103(35):13174–13179. doi:10.1073/pnas.0603747103
Nakajima K, Kohsaka S (2004) Microglia: neuroprotective and neurotrophic cells in the central nervous system. Curr Drug Targets Cardiovasc Haematol Disord 4(1):65–84
Magni P, Ruscica M, Dozio E, Rizzi E, Beretta G, Facino RM (2012) Parthenolide inhibits the LPS-induced secretion of IL-6 and TNF-alpha and NF-kappaB nuclear translocation in BV-2 microglia. Phytother Res 26(9):1405–1409. doi:10.1002/ptr.3732
Li Z, Hao Q, Luo J, Xiong J, Zhang S, Wang T, Bai L, Wang W, Chen M, Wang W, Gu L, Lv K, Chen J (2016) USP4 inhibits p53 and NF-kappaB through deubiquitinating and stabilizing HDAC2. Oncogene 35(22):2902–2912. doi:10.1038/onc.2015.349
Fan YH, Yu Y, Mao RF, Tan XJ, Xu GF, Zhang H, Lu XB, Fu SB, Yang J (2011) USP4 targets TAK1 to downregulate TNFalpha-induced NF-kappaB activation. Cell Death Differ 18(10):1547–1560. doi:10.1038/cdd.2011.11
Tao Z, Fusco A, Huang DB, Gupta K, Young Kim D, Ware CF, Van Duyne GD, Ghosh G (2014) p100/IkappaBdelta sequesters and inhibits NF-kappaB through kappaBsome formation. Proc Natl Acad Sci USA 111(45):15946–15951. doi:10.1073/pnas.1408552111
Funding
Funding was provided by The Biomechanical and Clinical Research of Lumbar Cortical Bone Tract Screw System (Grant No. MS22015074).
Author information
Authors and Affiliations
Corresponding author
Additional information
Xingjie Jiang and Mingchen Yu have contributed equally.
Rights and permissions
About this article
Cite this article
Jiang, X., Yu, M., Ou, Y. et al. Downregulation of USP4 Promotes Activation of Microglia and Subsequent Neuronal Inflammation in Rat Spinal Cord After Injury. Neurochem Res 42, 3245–3253 (2017). https://doi.org/10.1007/s11064-017-2361-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-017-2361-2