Abstract
Purpose
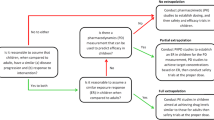
Bilastine is an H1 antagonist whose pharmacokinetics (PK) and pharmacodynamics (PD) have been resolved in adults with a therapeutic oral dose of 20 mg/day. Bilastine has favorable characteristics for use in pediatrics but the PK/PD and the optimal dose in children had yet to be clinically explored. The purpose is to: (1) Develop an ontogenic predictive model of bilastine PK linked to the PD in adults by integrating current knowledge; (2) Use the model to design a PK study in children; (3) Confirm the selected dose and the study design through the evaluation of model predictability in the first recruited children; (4) Consider for inclusion the group of younger children (< 6 years).
Methods
A semi-mechanistic approach was applied to predict bilastine PK in children assuming the same PD as described in adults. The model was used to simulate the time evolution of plasma levels and wheal and flare effects after several doses and design an adaptive PK trial in children that was then confirmed using data from the first recruits by comparing observations with model predictions.
Results
PK/PD simulations supported the selection of 10 mg/day in 2 to <12 year olds. Results from the first interim analysis confirmed the model predictions and design hence trial continuation.
Conclusion
The model successfully predicted bilastine PK in pediatrics and optimally assisted the selection of the dose and sampling scheme for the trial in children. The selected dose was considered suitable for younger children and the forthcoming safety study in children aged 2 to <12 years.







Similar content being viewed by others
Abbreviations
- %:
-
Percentage
- ADME:
-
Absorption distribution metabolism excretion
- AEs:
-
Adverse Events
- AR:
-
Allergic Rhinoconjuctivitis
- AUC:
-
Area under the curve
- CI:
-
Confidence interval
- CL:
-
Clearance
- CLr:
-
Renal clearance
- CLu:
-
Unbound clearance
- Cmax:
-
Maximum plasma concentration
- CO:
-
Cardiac output
- CYP450:
-
Cytochrome P450
- EMA:
-
European medicine agency
- F:
-
Bioavailability
- FDA:
-
Food and drug administration
- fu:
-
Unbound plasma fraction
- g/mol:
-
Grams per mol
- GFR:
-
Glomerular filtration rate
- h:
-
Hour
- IC50:
-
Half maximal inhibitory concentration
- ICH:
-
International conference on harmonization
- Ka:
-
Absorption rate constant
- L:
-
Liter
- L/h:
-
Liter per hour
- Log P:
-
Computational logarithm of the partition coefficient between n-octanol and water
- M:
-
Albumin molar concentration
- M&S:
-
Modeling & simulation
- mg:
-
Milligram
- mg/day:
-
Milligram per day
- mg/ml:
-
Milligram per milliliter
- MIDD:
-
Model informed drug development
- ng.h/ml:
-
Nanogram. hour/ml
- ng/ml:
-
Nanogram per milliliter
- OATP:
-
Organic anion transporting peptide
- PD:
-
Pharmacodynamics
- PDCO:
-
Pediatric committee of the EMA
- P-gp:
-
P-glycoprotein
- PIP:
-
Pediatric investigation plan
- PK:
-
Pharmacokinetics
- pka:
-
Negative base-10 logarithm of the acid dissociation constant
- Q:
-
Distribution or intercompartmental clearance
- QD:
-
Once a day (from the Latin quaque die)
- SEE:
-
Standard error of estimates
- TBW:
-
Total body water
- U:
-
Urticaria
- Vc:
-
Central volume of distribution
- Vp:
-
Peripheral volume of distribution
- VPC:
-
Visual predictive check
- Vss:
-
Steady state volume of distribution
- WT:
-
Body weight
- yr:
-
Year
- yrs:
-
Years
References
Pozas M, Rodriguez M, Valls I, Soler A, Vozmediano V. Considerations about paediatric dosing: from birth to adulthood, pharmacokinetics and pharmacodynamics. Int J Pharm [Internet]. 2014;469(2):238–9. Available from: http://www.sciencedirect.com/science/article/pii/S0378517314002853
Zisowsky J, Krause A, Dingemanse J. Drug development for pediatric populations: regulatory aspects. Pharmaceutics. 2010;2(4):364–88.
Field MJ, Behrman RE, Children I of M (US) C on CRI. Ethical conduct of clinical research involving children. [Internet]. Washington, D.C.: National Academies Press (US); 2004. Available from: http://www.ncbi.nlm.nih.gov/books/NBK25542/. Accessed 31 Aug 2010.
European Medicines Agency ICH Topic E11: Note for Guidance on Clinical Investigation of Medicinal Products in the Paediatric Population (CPMP/ICH/2711/99). Available from: http://www.ema.europa.eu/docs/en_GB/document_library/Scientific_guideline/2009/09/WC500003066.pdf. Accessed 31 Aug 2010.
European Commission. Ethical considerations for clinical trials on medicinal products with the paediatric population (Recommendations of the ad hoc group for the development of implementing guidelines for Directive 2001/20/EC relating to good clinical practice in the conduct. 2008. p. 1–34. Available from: http://ec.europa.eu/health//sites/health/files/files/eudralex/vol-10/ethical_considerations_en.pdf.
ICH Topic E 11: clinical investigation of medicinal products in the pediatric population. Step 5 note for guidance on investigation of medicinal products in the paediatric population (CPMP/ICH/2711/99). Prescrire Int 2001:1–13.
Regulation (EC) No 1901/2006 of the European Parliament and of the Council of 12 December 2006 on medicinal products for paediatric use and amending Regulation (EEC) No 1768/92, Directive 2001/20/EC, directive 2001/. Off J Eur Union [Internet] Available from: http://ec.europa.eu/health/files/eudralex/vol-1/reg_2006_1901/reg_2006_1901_en.pdf.
Bellanti F, Della PO. Modelling and simulation as research tools in paediatric drug development. Eur J Clin Pharmacol [Internet]. 2011;67(Suppl 1):75–86. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21246352
Vozmediano V, Ortega I, Lukas JC, Gonzalo A, Rodriguez M, Lucero ML. Integration of preclinical and clinical knowledge to predict intravenous PK in human: bilastine case study. Eur J Drug Metab Pharmacokinet. 39(1):33–41. Available from: https://doi.org/10.1007/s13318-013-0131-3.
Miller R, Ewy W, Corrigan BW, Ouellet D, Hermann D, Kowalski KG, et al. How modeling and simulation have enhanced decision making in new drug development. J Pharmacokinet Pharmacodyn [Internet]. Springer Netherlands; 2005;32(2):185–97. Available from: http://www.springerlink.com/content/f380345010x42k35/.
Lalonde RL, Kowalski KG, Hutmacher MM, Ewy W, Nichols DJ, Milligan PA, et al. Model-based drug development. Clin Pharmacol Ther [Internet]. 2007;82(1):21–32. Available from: https://doi.org/10.1038/sj.clpt.6100235
Meibohm B, Läer S, Panetta JC, Barrett JS. Population pharmacokinetic studies in pediatrics: issues in design and analysis. AAPS J [Internet]. 2005;7(2):E475–87. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2750985&tool=pmcentrez&rendertype=abstract
Bachert C, Kuna P, Zuberbier T. Bilastine in allergic rhinoconjunctivitis and urticaria. Allergy [Internet]. 2010;65(8):1–13. Available from: http://doi.wiley.com/10.1111/j.1398-9995.2010.02404.x
Pampura AN, Papadopoulos NG, Spičák V, Kurzawa R. Evidence for clinical safety, efficacy, and parent and physician perceptions of Levocetirizine for the treatment of children with allergic disease. Int Arch Allergy Immunol [Internet]. 2011;155(4):367–78. Available from: http://www.ncbi.nlm.nih.gov/pubmed/21346367
Carr WW. Pediatric allergic rhinitis: current and future state of the art. Allergy Asthma Proc [Internet]. 2008;29(1):14–23. Available from: http://pubget.com/paper/18302833/Pediatric_allergic_rhinitis__current_and_future_state_of_the_art
Druce HM, Kaliner MA. Allergic rhinitis. JAMA J Am Med Assoc [Internet]. 1988;259(2):260–3. Available from: http://jama.jamanetwork.com/article.aspx?articleid=370187
Pearlman D. Chronic rhinitis in children. J Allergy Clin Immunol [Internet]. 1988;81(5):962–6. Available from: https://doi.org/10.1016/0091-6749(88)90161-3
Jauregizar N, de la Fuente L, Lucero ML, Sologuren A, Leal N, Rodríguez M. Pharmacokinetic-pharmacodynamic modelling of the antihistaminic (H1) effect of bilastine. Clin Pharmacokinet [Internet]. 2009;48(8):543–54. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19705924
Kuna P, Bachert C, Nowacki Z, van Cauwenberge P, Agache I, Fouquert L, et al. Efficacy and safety of bilastine 20 mg compared with cetirizine 10 mg and placebo for the symptomatic treatment of seasonal allergic rhinitis: a randomized, double-blind, parallel-group study. Clin Exp Allergy [Internet]. 2009;39(9):1338–47. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19438584
ICH Topic E 14. Guidance on the clinical evaluation of QT / QTc interval prolongation and proarrhythmic potential. 2005.
Tyl B, Kabbaj M, Azzam S, Sologuren A, Valiente R, Reinbolt E, et al. Lack of significant effect of bilastine administered at therapeutic and supratherapeutic doses and concomitantly with ketoconazole on ventricular repolarization: results of a thorough QT study (TQTS) with QT-concentration analysis. J Clin Pharmacol. 2011;52(6):893–903. Available from: https://doi.org/10.1177/0091270011407191.
Simons FER, Simons KJ. Levocetirizine: pharmacokinetics and pharmacodynamics in children age 6 to 11 years. J Allergy Clin Immunol [Internet]. 2005;116(2):355–61. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16083790
Simons FER, Johnston L, Simons KJ. Clinical pharmacology of the H1-receptor antagonists cetirizine and loratadine in children. Pediatr Allergy Immunol [Internet]. 2000;11(2):116–9. Available from: http://www.blackwell-synergy.com/links/doi/10.1034%2Fj.1399-3038.2000.00045.x
EMEA. Guideline on the clinical development of medicinal products for the treatment of allergic rhino-conjunctivitis (CHMP/EWP/2455/02). 2004. p. 1–9. Available from: http://www.ema.europa.eu/ema/index.jsp?curl=pages/regulation/general/general_content_000525.jsp&mid=WC0b01ac0580511aff%5Cnpapers2://publication/uuid/2D38B7FC-8B5A-4E73-A85D-CB8E78E230C7.
FDA Draft Guidance for Industry. Allergic rhinitis : developing drug products for treatment. 2016. Available from: http://www.fda.gov/downloads/drugs/guidancecomplianceregulatoryinformation/guidances/ucm071293.pdf.
Mentré F, Dubruc C, Thénot J-P. Population pharmacokinetic analysis and optimization of the experimental design for mizolastine solution in children. J Pharmacokinet Pharmacodyn [Internet]. Springer Netherlands; 2001;28(3):299–319–319. Available from: http://www.springerlink.com/content/u18008015p34677u/.
Karafoulidou A, Suarez E, Anastasopoulou I, Katsarou O, Kouramba A, Kotsi P, et al. Population pharmacokinetics of recombinant factor VIII:C (ReFacto®) in adult HIV-negative and HIV-positive haemophilia patients. Eur J Clin Pharmacol. 2009;65(11):1121–30.
Encinas E, Calvo R, Lukas JC, Vozmediano V, Rodriguez M, Suarez E. A Predictive pharmacokinetic/pharmacodynamic model of fentanyl for analgesia/sedation in neonates based on a semi-physiologic approach. Paediatr Drugs [Internet]. 2013. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23657896.
Edginton AN, Schmitt W, Voith B, Willmann S. A mechanistic approach for the scaling of clearance in children. Clin Pharmacokinet. 2006;45(7):683–704.
Bouzom F, Walther B. Pharmacokinetic predictions in children by using the physiologically based pharmacokinetic modelling. Fundam Clin Pharmacol. 2008;22(6):579–87.
Sadaba B, Azanza JR, Gomez-Guiu A, Rodil R. Critical appraisal of bilastine for the treatment of allergic rhinoconjunctivitis and urticaria. Ther Clin Risk Manag. 2013;9(1):197–205.
Van Sassenbroeck DK, De Paepe P, Belpaire FM, Boon PA, Buylaert WA. Influence of hypovolemia on the pharmacokinetics and electroencephalographic effect of gamma-hydroxybutyrate in the rat. Anesthesiology. 2002;97(5):1218–26.
Björkman S. Prediction of drug disposition in infants and children by means of physiologically based pharmacokinetic (PBPK) modelling: theophylline and midazolam as model drugs. Br J Clin Pharmacol [Internet]. 2005;59(6):691–704. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1884855&tool=pmcentrez&rendertype=abstract
Johnson TN, Thomson M. Intestinal metabolism and transport of drugs in children: the effects of age and disease. J Pediatr Gastroenterol Nutr [Internet]. 2008;47(1):3–10. Available from: http://www.ncbi.nlm.nih.gov/pubmed/18607262
Batchelor HK, Fotaki N, Klein S. Paediatric oral biopharmaceutics: key considerations and current challenges. Adv Drug Deliv Rev [Internet]. Elsevier B.V.; 2014;73:102–26. Available from: https://doi.org/10.1016/j.addr.2013.10.006
Fakhoury M, Litalien C, Medard Y, Cavé H, Ezzahir N, Peuchmaur M, et al. Localization and mRNA expression of CYP3A and P-glycoprotein in human duodenum as a function of age. Drug Metab Dispos [Internet]. 2005;33(11):1603–7. Available from: http://dmd.aspetjournals.org/content/33/11/1603
Anderson BJ, Holford NHG. Mechanism-based concepts of size and maturity in pharmacokinetics. Annu Rev Pharmacol Toxicol [Internet]. 2008;48:303–32. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17914927
Zhang L, Pfister M, Meibohm B. Concepts and challenges in quantitative pharmacology and model-based drug development. AAPS J [Internet]. 2008;10(4):552–9. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2628212&tool=pmcentrez&rendertype=abstract
U.S. Department of Health and Human Services Food and Drug Administration, Center for Drug Evaluation and Research (CDER), Center for Biologics Evaluation and Research (CBER). E14 – Clinical Evaluation of QT/QTc Interval Prolongation and Proarrhythmic Potential for Non-Antiarrhythmic Drugs.2005; Guidance for industry. Available from: https://www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/UCM073153.pdf.
Dingemanse J, Appel-Dingemanse S. Integrated pharmacokinetics and pharmacodynamics in drug development. Clin Pharmacokinet [Internet]. 2007;46(9):713–37. Available from: http://www.ncbi.nlm.nih.gov/pubmed/17713971
Zhang L, Sinha V, Forgue ST, Callies S, Ni L, Peck R, et al. Model-based drug development: the road to quantitative pharmacology. J Pharmacokinet Pharmacodyn [Internet]. 2006;33(3):369–93. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16770528
Peck CC. Quantitative clinical pharmacology is transforming drug regulation. J Pharmacokinet Pharmacodyn [Internet]. Springer Netherlands; 2010;37(6):617–628. Available from: http://www.springerlink.com/content/r54x30m0j0gk5k37/.
Alcorn J, McNamara PJ. Ontogeny of hepatic and renal systemic clearance pathways in infants: part II. Clin Pharmacokinet [Internet]. 2002;41(13):1077–94. Available from: http://www.ncbi.nlm.nih.gov/pubmed/12222995
Allegra Uses, Dosage & Side Effects - Drugs.com [Internet]. Available from: http://www.drugs.com/allegra.html.
Krishna R, Krishnaswami S, Kittner B, Sankoh AJ, Jensen BK. The utility of mixed-effects covariate analysis in rapid selection of doses in pediatric subjects: a case study with fexofenadine hydrochloride. Biopharm Drug Dispos [Internet]. 2004;25(9):373–87. Available from: http://www.ncbi.nlm.nih.gov/pubmed/15517550
Edginton AN, Willmann S, Schmitt W. Predicting pharmacokinetics in children using PK-Sim ®. Water. 2005:51368–8.
Hayton WL. Maturation and growth of renal function: dosing renally cleared drugs in children. AAPS PharmSci [Internet]. 2002;2(1):E3. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2750998&tool=pmcentrez&rendertype=abstract.
McNamara PJ, Alcorn J. Protein binding predictions in infants. AAPS PharmSci [Internet]. 2002;4(1):E4. Available from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=2751289&tool=pmcentrez&rendertype=abstract
Davies B, Morris T. Physiological parameters in laboratory animals and humans. Pharm Res [Internet]. 1993;10(7):1093–5. Available from: http://www.ncbi.nlm.nih.gov/pubmed/8378254
ICRP. International comission of radiological protection (ICRP): reference values [Internet]. 89. 2003. Available from: http://www.icrp.org/publication.asp?id=ICRPPublication 89.
Mahmood I. Interspecies pharmacokinetic scaling: Principles and Application of Allometric Scaling. Rockville: Pine House Publishers; 2005. p. 27.
Mahmood I. Pediatric pharmacology and pharmacokinetics. Rockville: Pine House Publishers; 2008. p. 52–67.
Acknowledgments and Disclosures
One of the authors (VV) became involved thanks to support from the Department of Industry, Commerce and Tourism of the Basque Government (Ikertu). This work is also part of the doctoral thesis of the corresponding author (directed by Dr. Rosario Calvo). Finally, the authors of this manuscript would also like to thank Maria Luisa Lucero for her contribution during the management of FAES FARMA, S.A. Research Department, as well as Roman Valiente for his contribution as Director of the Clinical Research Department of FAES FARMA, S.A. Dr. Ander Sologuren is employee of FAES FARMA, S.A., Spain. None of the other authors have any conflicts of interest other than receiving funding from FAES FARMA, S.A., for designing the trial and conducting the modeling & simulation analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vozmediano, V., Sologuren, A., Lukas, J.C. et al. Model Informed Pediatric Development Applied to Bilastine: Ontogenic PK Model Development, Dose Selection for First Time in Children and PK Study Design. Pharm Res 34, 2720–2734 (2017). https://doi.org/10.1007/s11095-017-2248-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-017-2248-6