Abstract
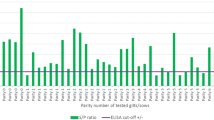
The present study aimed to determine the prevalence of porcine circovirus-2 (PCV-2) DNA-positive ovarian and uterine tissues in gilts culled due to reproductive disturbance in Thailand. Tissues (70 ovaries and 102 uteri) and serum (n = 102) samples from 102 gilts were included. PCV-2 DNA was detected by using polymerase chain reactions. The localisation of PCV-2 antigen was determined by immunohistochemistry, and PCV-2 antibody was evaluated by ELISA. PCV-2 DNA was detected in 30.0 % (21/70) of the ovaries and in 45.1 % (46/102) of the uteri. Age did not influence the frequency of PCV-2 DNA detection in these reproductive organs of gilts (P > 0.05). The prevalence of PCV-2 DNA-positive uterine tissue in gilts culled due to non-reproductive problems (20.0 %) was lower than gilts culled due to abortion (85.0 %), abnormal vaginal discharge (47.5 %) and anoestrus (53.5 %) (P < 0.05). The prevalence of PCV-2 DNA-positive uterine tissue in the gilts with high antibody titres (23.0 %) was lower than in gilts with low antibody titres (57.6 %) and seronegative gilts (64.5 %) (P < 0.05). PCV-2 immunostaining was detected in the endometrial cells, lymphocytes and macrophages of the uteri and in oocytes and granulosa cells of the ovaries. In conclusion, the detection of PCV-2 in the reproductive organs reveals an important potential impact of this virus on the reproductive apparatus in gilts.


Similar content being viewed by others
References
Bielanski, A., Larochelle, R., Algire, J. and Magar, R., 2004. Distribution of PCV-2 DNA in the reproductive tract, oocytes and embryos of PCV-2 antibody-positive pigs. The Veterinary Records, 155, 597–598.
Bolin, S.R., Stoffregen, W.C., Nayar, G.P. and Hamel, A.L., 2001. Postweaning multisystemic wasting syndrome induced after experimental inoculation of cesarean-derived, colostrum-deprived piglets with type 2 porcine circovirus. Journal of Veterinary Diagnostic Investigation, 13, 185–194.
Gauger, P.C., Lager, K.M., Vincent, A.L., Opriessnig, T., Cheung, A.K., Butler, J.E., Kehrli, M.E. Jr., 2011. Leukogram abnormalities in gnotobiotic pigs infected with porcine circovirus type 2. Veterinary Microbiology, 154, 185–190.
Kim, H.B., Lyoo, K.S. and Joo, H.S., 2009. Efficacy of different disinfectants in vitro against porcine circovirus type 2. Veterinary Record, 164, 599–600.
Kim, J., Jung, K. and Chae, C., 2004. Prevalence of porcine circovirus type 2 in aborted foetuses and stillborn piglets. Veterinary Record, 155, 489–492.
Langohr, I.M., Stevenson, G.W., Nelson, E.A., Lenz, S.D., HogenEsch, H., Wei, H. and Pogranichniy, R.M., 2010. Vascular lesions in pigs experimentally infected with porcine circovirus type 2 serogroup B. Veterinary Pathology, 47, 140–147.
Madson, D.M. and Opriessnig, T., 2011. Effect of porcine circovirus type 2 (PCV2) infection on reproduction: disease, vertical transmission, diagnostics and vaccination. Animal Health Research Reviews, 12, 47–65.
Madson, D.M., Patterson, A.R., Ramamoorthy, S., Pal, N., Meng, X.J. and Opriessnig, T., 2009. Reproductive failure experimentally induced in sows via artificial insemination with semen spiked with porcine circovirus type 2. Veterinary Pathology, 46, 707–716.
Paphavasit, T., Lehrbach, P., Navasakuljinda, W., Kedkovid, R., Lacharoje, S., Thanawongnuwech, R. and Teankum, K., 2009. Efficacy of a chimeric PCV2 vaccine: a field trial. Thai Journal of Veterinary Medicine, 39, 145–155.
Patterson, A.R., Baker, R.B., Madson, D.M., Pintar, A.L., Opriessnig, T., 2011. Disinfection protocols reduce the amount of porcine circovirus type 2 in contaminated 1:61 scale model livestock transport vehicles. Journal of Swine Health and Production, 19, 156–164.
Pejsak, Z., Kusior, G., Pomorska-Mol, M. and Podgorska, K., 2012. Influence of long-term vaccination of a breeding herd of pigs against PCV2 on reproductive parameters, Polish Journal of Veterinary Science, 15, 37–42.
Prickett, J.R., Johnson, J., Murtaugh, M.P., Puvanendiran, S., Wang, C., Zimmerman, J.J., Opriessnig, T., 2011. Prolonged detection of PCV2 and anti-PCV2 antibody in oral fluids following experimental inoculation. Transboundary and Emerging Diseases, 58, 121–127.
Ritterbusch, G.A., Rocha, C.A., Mores, N., Simon, N.L., Zanella, E.L., Coldebella, A. and Ciacci-Zanella, J.R. 2012. Natural co-infection of torque teno virus and porcine circovirus 2 in the reproductive apparatus of swine. Research in Veterinary Science, 92, 519–523.
Roongsitthichai, A., Suwimonteerabutr, J., Kaeoket, K., Koonjaenak, S. and Tummaruk, P., 2011. Association between serum cortisol and progesterone concentrations and the infiltration of immune cells in the endometrium of gilts with vaginal discharge. Comparative Clinical Pathology, 20, 563–571.
Rose, N., Opriessnig, T., Grasland, B. and Jestin, A., 2012. Epidemiology and transmission of porcine circovirus type 2 (PCV2). Virus Research, 164, 78–89.
Sarli, G., Morandi, F., Panarese, S., Bacci, B., Ferrara, D., Bianco, C., Fusaro, L., Bacci, M.L., Galeati, G., Dottori, M., Bonilauri, P., Lelli, D., Leotti, G., Vila, T., Joisel, F., Allan, G., Benazzi, C., Ostanello, F., 2012. Reproduction in porcine circovirus type 2 (PCV2) seropositive gilts inseminated with PCV2b spiked semen. Acta Veterinaria Scandinavica, 54, 51.
Segalés, J., 2012. Porcine circovirus type 2 (PCV2) infections: clinical signs, pathology and laboratory diagnosis. Virus Research, 164, 10–19.
Segalés, J., Calsamiglia, M., Olvera, A. Sibila, M., Badiella, L., Domingo, M., 2005. Quantification of porcine circovirus type 2 (PCV2) DNA in serum and tonsillar, nasal, tracheo-bronchial, urinary and faecal swabs of pigs with and without postweaning multisystemic wasting syndrome (PMWS). Veterinary Microbiology, 111, 223–229.
Silva, F.M., Silva, Junior A., Vidigal, P.M., Oliveira, C.R., Viana, V.W., Silva, C.H., Vargas, M.I., Fietto, J.L. and Almeida, M.R., 2011. Porcine circovirus-2 viral load versus lesions in pigs: perspectives for post-weaning multisystemic wasting syndrome. Journal of Comparative Pathology, 144, 296–302.
Teamsuwan, Y., Kaeoket, K., Tienthai, P. and Tummaruk, P. 2010. Morphological changes and infiltration of immune cells in the endometrium of anoestrus gilt in relation to the ovarian appearance and serum progesterone. Thai Journal of Veterinary Medicine, 40, 31–40.
Tummaruk, P., Kesdangsakonwut, S. and Kunavongkrit, A. 2009. Relationships among specific reasons for culling, reproductive data, and gross morphology of the genital tracts in gilts culled due to reproductive failure in Thailand. Theriogenology, 71, 369–375.
Tummaruk, P., Tantasuparuk, W., Techakumphu, A. and Kunavongkrit, A. 2007. Age, body weight and backfat thickness at first observed oestrus in crossbred Landrace x Yorkshire gilts, seasonal variations and their influence on subsequence reproductive performance. Animal Reproduction Science, 99, 167–181.
Tummaruk, P., Tantasuparuk, W., Techakumphu, M. and Kunavongkrit, A. 2010. Influence of repeat-service and weaning-to-first-service interval on farrowing proportion of gilts and sows. Preventive Veterinary Medicine, 96, 194–200.
Acknowledgments
Financial support for the present study was provided by grant for International Research Integration: Chula Research Scholar, Ratchadaphiseksomphot Endowment Fund.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pearodwong, P., Srisuwatanasagul, S., Teankum, K. et al. Prevalence of porcine circovirus-2 DNA-positive ovarian and uterine tissues in gilts culled due to reproductive disturbance in Thailand. Trop Anim Health Prod 47, 833–840 (2015). https://doi.org/10.1007/s11250-015-0796-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-015-0796-5