Abstract
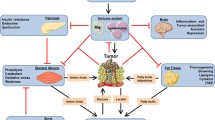
Despite recent consensus definitions, lack of specific biomarkers remains a hurdle towards a more accurate and efficient diagnosis of cancer cachexia, distinguishing cachexia as a separate entity from other wasting syndromes. In a previous pilot study, we have shown that cancer-cachectic mice have a unique metabolic fingerprint with distinct glucose and lipid alterations compared to healthy controls. Further metabolomics studies were carried out to investigate differences in metabolic profiles of cancer-cachectic mice to tumor-bearing non-cachectic mice, calorie-restricted mice, and surgically treated cancer-cachectic mice. CD2F1 mice were divided into: (1) Cachexia Group received cachexia-inducing C26 undifferentiated colon carcinoma cells; (2) Tumor-Burden Group received, non-cachectic, P388 lymphoma cells; (3) Caloric-Restriction Group, remaining cancer-free, but subjected to caloric-restriction; (4) Surgery Group, similar to Cachexia Group, but tumors resected mid-experiment; and (5) Control Group aged intact. Baseline, mid-experiment and final serum samples were collected for 1H NMR spectroscopic analysis. After data reduction, unsupervised principal component analysis and orthogonal projections to latent structures analyses demonstrate that the unique metabolic fingerprint is independent of tumor-burden and distinct from profiles of caloric-restriction and aging. Hyperlipidemia, hyperglycemia, and reduced branched-chain amino acids distinguish cachexia from other groups. Furthermore, the profile of surgically treated mice differs from that of cachectic mice, reverting to a profile more congruent with healthy controls indicating cachexia is amenable to correction where surgical cure is possible. That metabolomic analysis of murine serum is able to differentiate cachexia from tumor-burden and caloric-restriction warrants similar translational investigations in patients to explore cancer cachexia’s unique biomarkers.





Similar content being viewed by others
References
Anthony, J. C., Yoshizawa, F., Anthony, T. G., Vary, T. C., Jefferson, L. S., & Kimball, S. R. (2000). Leucine stimulates translation initiation in skeletal muscle of postabsorptive rats via a rapamycin-sensitive pathway. Journal of Nutrition, 130, 2413–2419.
Beck, S. A., & Tisdale, M. J. (1989). Nitrogen excretion in cancer cachexia and its modification by a high fat diet in mice. Cancer Research, 49, 3800–3804.
Bennani-Baiti, N., & Walsh, D. (2009). What is cancer anorexia-cachexia syndrome? A historical perspective. Journal of the Royal College of Physicians of Edinburgh, 39, 257–262.
Busquets, S., Alvarez, B., Lopez-Soriano, F. J., & Argiles, J. M. (2002). Branched-chain amino acids: A role in skeletal muscle proteolysis in catabolic states? Journal of Cellular Physiology, 191, 283–289.
Busquets, S., Carbo, N., Almendro, V., Figueras, M., Lopez-Soriano, F. J., & Argiles, J. M. (2001). Hyperlipemia: A role in regulating UCP3 gene expression in skeletal muscle during cancer cachexia? FEBS Letters, 505, 255–258.
Conti, F., Manganaro, M., & Miccheli, A. (2006). Metabolomics and medical practice. Clinica Terapeutica, 157, 549–552.
Darlington, G. J., Wilson, D. R., & Lachman, L. B. (1986). Monocyte-conditioned medium, interleukin-1, and tumor necrosis factor stimulate the acute phase response in human hepatoma cells in vitro. Journal of Cell Biology, 103, 787–793.
Delano, M. J., & Moldawer, L. L. (2006). The origins of cachexia in acute and chronic inflammatory diseases. Nutrition in Clinical Practice: Official Publication of the American Society for Parenteral and Enteral Nutrition, 21, 68–81.
Eley, H. L., Russell, S. T., & Tisdale, M. J. (2007). Effect of branched-chain amino acids on muscle atrophy in cancer cachexia. Biochemical Journal, 407, 113–120.
Evans, W. J., et al. (2008). Cachexia: A new definition. Clinical Nutrition, 27, 793–799.
Fearon, K. C., Glass, D. J., & Guttridge, D. C. (2012). Cancer cachexia: Mediators, signaling, and metabolic pathways. Cell Metabolism, 16, 153–166.
Fearon, K. C., & Moses, A. G. (2002). Cancer cachexia. International Journal of Cardiology, 85, 73–81.
Fearon, K., et al. (2011). Definition and classification of cancer cachexia: An international consensus. Lancet Oncology, 12, 489–495.
Feingold, K. R., Soued, M., Serio, M. K., Moser, A. H., Dinarello, C. A., & Grunfeld, C. (1989). Multiple cytokines stimulate hepatic lipid synthesis in vivo. Endocrinology, 125, 267–274.
Grunfeld, C., et al. (1989). Persistence of the hypertriglyceridemic effect of tumor necrosis factor despite development of tachyphylaxis to its anorectic/cachectic effects in rats. Cancer Research, 49, 2554–2560.
Holecek, M. (2011). Branched-chain amino acid oxidation in skeletal muscle—physiological and clinical importance of its modulation by reactant availability. Current Nutrition & Food Science, 7, 50–56.
Holm, E., et al. (1995). Substrate balances across colonic carcinomas in humans. Cancer Research, 55, 1373–1378.
Kotler, D. P. (2000). Cachexia. Annals of Internal Medicine, 133, 622–634.
Lawton, K. A., et al. (2008). Analysis of the adult human plasma metabolome. Pharmacogenomics, 9, 383–397.
Lecker, S. H., et al. (2004). Multiple types of skeletal muscle atrophy involve a common program of changes in gene expression. FASEB Journal, 18, 39–51.
Lee, S. W., Dai, G., Hu, Z., Wang, X., Du, J., & Mitch, W. E. (2004). Regulation of muscle protein degradation: Coordinated control of apoptotic and ubiquitin-proteasome systems by phosphatidylinositol 3 kinase. Journal of the American Society of Nephrology, 15, 1537–1545.
Lowenstein, J. M., & Goodman, M. N. (1978). The purine nucleotide cycle in skeletal muscle. Federation Proceedings, 37, 2308–2312.
Mahmoud, F. A., & Rivera, N. I. (2002). The role of C-reactive protein as a prognostic indicator in advanced cancer. Current Oncology Reports, 4, 250–255.
Mantovani, G., et al. (2010). Phase II nonrandomized study of the efficacy and safety of COX-2 inhibitor celecoxib on patients with cancer cachexia. Journal of Molecular Medicine, 88, 85–92.
Martinez-Outschoorn, U. E., et al. (2011). Energy transfer in “parasitic” cancer metabolism: Mitochondria are the powerhouse and Achilles’ heel of tumor cells. Cell Cycle, 10, 4208–4216.
Nelson, K. A., Walsh, D., & Sheehan, F. A. (1994). The cancer anorexia-cachexia syndrome. Journal of Clinical Oncology, 12, 213–225.
Norton, J. A., Gorschboth, C. M., Wesley, R. A., Burt, M. E., & Brennan, M. F. (1985). Fasting plasma amino acid levels in cancer patients. Cancer, 56, 1181–1186.
O’Connell, T., et al. (2008). Metabolomic analysis of cancer cachexia reveals distinct lipid and glucose alterations. Metabolomics, 4, 216–225.
Perlmutter, D. H., Dinarello, C. A., Punsal, P. I., & Colten, H. R. (1986). Cachectin/tumor necrosis factor regulates hepatic acute-phase gene expression. Journal of Clinical Investigation, 78, 1349–1354.
Pickering, W. P., Price, S. R., Bircher, G., Marinovic, A. C., Mitch, W. E., & Walls, J. (2002). Nutrition in CAPD: Serum bicarbonate and the ubiquitin-proteasome system in muscle. Kidney International, 61, 1286–1292.
Rieu, I., et al. (2006). Leucine supplementation improves muscle protein synthesis in elderly men independently of hyperaminoacidaemia. Journal of Physiology, 575, 305–315.
Rofe, A. M., Bourgeois, C. S., Coyle, P., Taylor, A., & Abdi, E. A. (1994). Altered insulin response to glucose in weight-losing cancer patients. Anticancer Research, 14, 647–650.
Shaw, J. H., & Wolfe, R. R. (1987). Glucose and urea kinetics in patients with early and advanced gastrointestinal cancer: The response to glucose infusion, parenteral feeding, and surgical resection. Surgery, 101, 181–191.
Strasser, F. (2008). Diagnostic criteria of cachexia and their assessment: Decreased muscle strength and fatigue. Current Opinion in Clinical Nutrition and Metabolic Care, 11, 417–421.
Tan, B. H., & Fearon, K. C. (2008). Cachexia: Prevalence and impact in medicine. Current Opinion in Clinical Nutrition and Metabolic Care, 11, 400–407.
Tisdale, M. J. (1997). Cancer cachexia: Metabolic alterations and clinical manifestations. Nutrition, 13, 1–7.
Tisdale, M. J. (2002). Cachexia in cancer patients. Nature Reviews Cancer, 2, 862–871.
van Ravenzwaay, B., et al. (2007). The use of metabolomics for the discovery of new biomarkers of effect. Toxicology Letters, 172, 21–28.
Wiklund, S., et al. (2008). Visualization of GC/TOF-MS-based metabolomics data for identification of biochemically interesting compounds using OPLS class models. Analytical Chemistry, 80, 115–122.
Yoshizawa, F. (2004). Regulation of protein synthesis by branched-chain amino acids in vivo. Biochemical and Biophysical Research Communications, 313, 417–422.
Zhou, Q., Du, J., Hu, Z., Walsh, K., & Wang, X. H. (2007). Evidence for adipose-muscle cross talk: Opposing regulation of muscle proteolysis by adiponectin and fatty acids. Endocrinology, 148, 5696–5705.
Acknowledgments
J. Walter Juckett postdoctoral fellowship (H.D.); National Institutes of Health T32 training grant (S.A.); University of North Carolina Program in Translational Science grant (M.C.); General Clinical Research Center grant #RR000046 (T.O.); and National Institute of Environmental Health Sciences grant P30ES10126 (T.O.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Der-Torossian, H., Asher, S.A., Winnike, J.H. et al. Cancer cachexia’s metabolic signature in a murine model confirms a distinct entity. Metabolomics 9, 730–739 (2013). https://doi.org/10.1007/s11306-012-0485-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11306-012-0485-6