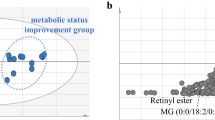
Abstract
Background
Menopause is associated with increased abdominal fat and increased risk of developing diabetes and cardiovascular disease.
Objectives
The present study evaluated the plasma metabolic response in relation to insulin sensitivity after weight loss via diet intervention.
Methods
This work includes two studies; i) Ten women on a 5 weeks Paleolithic-type diet (PD, 30 energy percent (E%) protein, 40 E% fat, 30 E% carbohydrates), ii) 55 women on 6 months of either PD or Nordic Nutrition Recommendations diet (NNR, 15 E% protein, 30 E% fat, and 55 E% carbohydrates). Plasma metabolic profiles were acquired at baseline and post diet using gas chromatography time-of-flight/mass spectrometry and investigated in relation to insulin sensitivity using multivariate bioinformatics.
Results
Both the PD and NNR diet resulted in significant weight loss, reduced waist circumference, improved serum lipid profiles, and improved insulin sensitivity. We detected a baseline metabolic profile that correlated significantly with insulin sensitivity, and of which components increased significantly in the PD group compared to NNR. Specifically, a significant increase in myo-inositol (MI), a second messenger of insulin action, and β-hydroxybutyric acid (β-HB) increased while dihomo-gamma-linoleic acid (DGLA) decreased in PD compared to NNR, which correlated with improved insulin sensitivity. We also detected a significant decrease in tyrosine and tryptophan, potential markers of insulin resistance when elevated in the circulation, with the PD but not the NNR.
Conclusions
Using metabolomics, we detected changes in the plasma metabolite profiles associated with weight loss in postmenopausal women by different diets. The metabolic profiles following 6 months of PD were linked to beneficial effects on insulin sensitivity compared to NNR.
Graphical Abstract




Similar content being viewed by others
References
Becker, W. (2005). New Nordic nutrition recommendations 2004. Physical activity as important as good nourishing food. Lakartidningen, 102, 2757-8–2760-2.
Brennan, L. (2013). Metabolomics in nutrition research: Current status and perspectives. Biochemical Society Transactions, 41, 670–673.
Clements, R. S., & Darnell, B. (1980). Myoinositol content of common foods—development of a high-myo-inositol diet. American Journal of Clinical Nutrition, 33, 1954–1967.
Corrado, F., D’Anna, R., Di Vieste, G., Giordano, D., Pintaudi, B., Santamaria, A., & Di Benedetto, A. (2011). The effect of myoinositol supplementation on insulin resistance in patients with gestational diabetes. Diabetic Medicine, 28(8), 972–975.
Dunaif, A. (1997). Insulin resistance and the polycystic ovary syndrome: Mechanism and implications for pathogenesis. Endocrine Reviews, 18, 774–800.
EFRON, B. A. G., G. 1983. A Leisurely Look at the Bootstrap, the Jack-knife, and Cross-validation. The American Statistician. http://www.jstor.org/stable/2685844: American Statistical Association.
Fiehn, O., Kopka, J., Dormann, P., Altmann, T., Trethewey, R. N., & Willmitzer, L. (2000). Metabolite profiling for plant functional genomics. Nature Biotechnology, 18, 1157–1161.
Genazzani, A. D., Lanzoni, C., Ricchieri, F., & Jasonni, V. M. (2008). Myo-inositol administration positively affects hyperinsulinemia and hormonal parameters in overweight patients with polycystic ovary syndrome. Gynecological Endocrinology, 24, 139–144.
Giordano, D., Corrado, F., Santamaria, A., Quattrone, S., Pintaudi, B., di Benedetto, A., & D’Anna, R. (2011). Effects of myo-inositol supplementation in postmenopausal women with metabolic syndrome: A perspective, randomized, placebo-controlled study. Menopause-the Journal of the North American Menopause Society, 18, 102–104.
Jönsson, T., Granfeldt, Y., Ahrén, B., Branell, U. C., Pålsson, G., Hansson, A., et al. (2009). Beneficial effects of a Paleolithic diet on cardiovascular risk factors in type 2 diabetes: A randomized cross-over pilot study. Cardiovasc Diabetol, 8(35), 1–14.
Jönsson, T., Granfeldt, Y., Erlanson-Albertsson, C., Ahrén, B., & Lindeberg, S. (2010). A paleolithic diet is more satiating per calorie than a mediterranean-like diet in individuals with ischemic heart disease. Nutrition & metabolism, 7(1), 1.
Jonsson, P., Johansson, A. I., Gullberg, J., Trygg, J., Grung, B., Marklund, S., et al. (2005). High-throughput data analysis for detecting and identifying differences between samples in GC/MS-based metabolomic analyses. Analytical Chemistry, 77(17), 5635–5642.
Jonsson, P., Johansson, E. S., Wuolikainen, A., Lindberg, J., Schuppe-Koistinen, I., Kusano, M., et al. (2006). Predictive metabolite profiling applying hierarchical multivariate curve resolution to GC-MS datas - A potential tool for multi-parametric diagnosis. Journal of Proteome Research, 5, 1407–1414.
Kapoor, R., & Huang, Y.-S. (2006). Gamma linolenic acid: An antiinflammatory omega-6 fatty acid. Current Pharmaceutical Biotechnology, 7, 531–534.
Kell, D. B. (2004). Metabolomics and systems biology: Making sense of the soup. Current Opinion in Microbiology, 7, 296–307.
Lemieux, M., Al-Jawadi, A., Wang, S., & Moustaid-Moussa, N. (2013). Metabolic Profiling in Nutrition and Metabolic Disorders. Advances in Nutrition, 4, 548–550.
Lindeberg, S., Jonsson, T., Granfeldt, Y., Borgstrand, E., Soffman, J., Sjostrom, K., & Ahren, B. (2007). A Palaeolithic diet improves glucose tolerance more than a Mediterranean-like diet in individuals with ischaemic heart disease. Diabetologia, 50, 1795–1807.
Lisabeth, L. D., Beiser, A. S., Brown, D. L., Murabito, J. M., Kelly-Hayes, M., & Wolf, P. A. (2009). Age at natural menopause and risk of ischemic stroke The Framingham Heart Study. Stroke, 40, 1044–1049.
McNiven, E. M. S., German, J. B., & Slupsky, C. M. (2011). Analytical metabolomics: Nutritional opportunities for personalized health. Journal of Nutritional Biochemistry, 22, 995–1002.
Mellberg, C., Sandberg, S., Ryberg, M., Eriksson, M., Brage, S., Larsson, C., et al. (2014). Long-term effects of a Palaeolithic-type diet in obese postmenopausal women: A 2-year randomized trial. European Journal of Clinical Nutrition, 68, 350–357.
Nestler, J. E., Jakubowicz, D. J., Reamer, P., Gunn, R. D., & Allan, G. (1999). Ovulatory and metabolic effects of D-chiro-inositol in the polycystic ovary syndrome. New England Journal of Medicine, 340, 1314–1320.
Newgard, C. B., An, J., Bain, J. R., Muehlbauer, M. J., Stevens, R. D., Lien, L. F., et al. (2009). A branched-chain amino acid-related metabolic signature that differentiates obese and lean humans and contributes to insulin resistance. Cell Metabolism, 9(4), 311–326.
Peppa, M., Koliaki, C., Papaefstathiou, A., Garoflos, E., Katsilambros, N., Raptis, S. A., et al. (2013). Body composition determinants of metabolic phenotypes of obesity in nonobese and obese postmenopausal women. Obesity, 21, 1807–1814.
Redestig, H., Fukushima, A., Stenlund, H., Moritz, T., Arita, M., Saito, K., & Kusano, M. (2009). Compensation for systematic cross-contribution improves normalization of mass spectrometry based metabolomics data. Analytical Chemistry, 81, 7974–7980.
Ren, J., & Ceylan-Isik, A. F. (2004). Diabetic cardiomyopathy—Do women differ from men? Endocrine, 25, 73–83.
Rexrode, K. M., Carey, V. J., Hennekens, C. H., Walters, E. E., Colditz, G. A., Stampfer, M. J., et al. (1998). Abdominal adiposity and coronary heart disease in women. Jama-Journal of the American Medical Association, 280, 1843–1848.
Russo, G. L. (2009). Dietary n-6 and n-3 polyunsaturated fatty acids: From biochemistry to clinical implications in cardiovascular prevention. Biochemical Pharmacology, 77, 937–946.
Ryberg, M., Sandberg, S., Mellberg, C., Stegle, O., Lindahl, B., Larsson, C., et al. (2013). A Palaeolithic-type diet causes strong tissue-specific effects on ectopic fat deposition in obese postmenopausal women. Journal of Internal Medicine, 274, 67–76.
Sakuma, M., Kametani, S., & Akanuma, H. (1998). Purification and some properties of a hepatic NADPH-dependent reductase that specifically acts on 1,5-anhydro-D-fructose. Journal of Biochemistry, 123, 189–193.
Saltiel, A. R., & Kahn, C. R. (2001). Insulin signalling and the regulation of glucose and lipid metabolism. Nature, 414, 799–806.
Suhre, K. (2014). Metabolic profiling in diabetes. Journal of Endocrinology, 221, R75–R85.
Trygg, J., Gullberg, J., Johansson, A. I., Jonsson, P., Antti, H., Marklund, S. L., & Moritz, T. (2005). Extraction and GC/MS analysis of the human blood plasma metabolome. Analytical Chemistry, 77, 8086–8094.
Virtanen, J. K., Mursu, J., Voutilainen, S., Uusitupa, M., & Tuomainen, T.-P. (2014). Serum omega-3 polyunsaturated fatty acids and risk of incident type 2 diabetes in men: The Kuopio Ischemic Heart Disease Risk Factor study. Diabetes Care, 37, 189–196.
Wang, T. J., Larson, M. G., Vasan, R. S., Cheng, S., Rhee, E. P., McCabe, E., et al. (2011). Metabolite profiles and the risk of developing diabetes. Nature Medicine, 17, 448–453.
Wishart, D. S., Tzur, D., Knox, C., Eisner, R., Guo, A. C., Young, N., et al. (2007). HMDB: The human metabolome database. Nucleic Acids Research, 35, D521–D526.
Yamanouchi, T., Inoue, T., Ichiyanagi, K., Sakai, T., & Ogata, N. (2003). 1,5-Anhydroglucitol stimulates insulin release in insulinoma cell lines. Biochimica Et Biophysica Acta-General Subjects, 1623, 82–87.
Yamanouchi, T., Ogata, N., Tagaya, T., Kawasaki, T., Sekino, N., Funato, H., et al. (1996). Clinical usefulness of serum 1,5-anhydroglucitol in monitoring glycaemic control. Lancet, 347, 1514–1518.
Zeisel, S. H., Freake, H. C., Bauman, D. E., Bier, D. M., Burrin, D. G., German, J. B., et al. (2005). The nutritional phenotype in the age of metabolomics. Journal of Nutrition, 135, 1613–1616.
Acknowledgments
We are grateful to all of the women who participated in this study. Krister Lundgren and Inga-Britt Carlsson assisted during the GC-TOF/MS analysis, and Inger Arnesjö made important contributions during the dietary intervention studies. This work was supported by grants from the Swedish Research Council (VR), the Swedish Council for Working Life and Social Research, the Swedish Strategic Research Foundation (SSF), Wallenberg Consortium North (WCN), the Kempe Foundation, the Swedish Heart and Lung Foundation, the Medical Faculty at Umeå University, and the Västerbotten County Council.
Financial support
This study was supported by grants from The Swedish Council for Working Life and Social Research (2006-0699 and 2010-0398), the Swedish Research Council (K2011-12237-15-6), the Swedish Heart and Lung Foundation, the Swedish Diabetes Foundation, King Gustaf V and Drottning Victorias Foundation, the County Council of Västerbotten, and Umeå University, Sweden.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Henrik Antti and Tommy Olsson have shared senior authorship.
Rights and permissions
About this article
Cite this article
Chorell, E., Ryberg, M., Larsson, C. et al. Plasma metabolomic response to postmenopausal weight loss induced by different diets. Metabolomics 12, 85 (2016). https://doi.org/10.1007/s11306-016-1013-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-016-1013-x