Abstract
Objectives
Determination of lymphatic metastasis is of great importance for both treatment planning and patient prognosis. We aim to distinguish tumor metastatic lymph nodes (TLNs) and reactive lymph nodes (RLNs) with diffusion-weighted and superparamagnetic iron oxide (SPIO)-enhanced magnetic resonance imaging (MRI).
Materials and Methods
Ipsilateral popliteal lymph node metastasis or lymphadenitis model was established by hock injection of either luciferase-expressing 4T1 murine breast cancer cells or complete Freund's adjuvant in male BALB/c mice. At different time points after inoculation, bioluminescence imaging and T2-weighted, diffusion-weighted, and SPIO-enhanced MRI were performed. Imaging findings were confirmed by histopathological staining.
Results
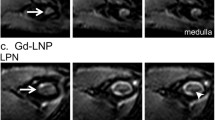
Size enlargement was observed in both TLNs and RLNs. At day 28, TLNs showed strong bioluminescence signal and bigger size than RLNs (p < 0.01). At early stages up to day 21, both TLNs and RLNs appeared homogeneous on diffusion-weighted imaging. At day 28, TLNs showed heterogeneous apparent diffusion coefficient (ADC) map with significantly higher average ADC value of 0.41 ± 0.03 × 10−3mm2/s than that of RLNs (0.34 ± 0.02 × 10−3mm2/s; p < 0.05). On SPIO-enhanced MRI, both TLNs and RLNs showed distinct T2 signal reduction at day 21 after inoculation. At day 28, TLNs demonstrated partial uptake of the iron oxide particles, which was confirmed by Prussian blue staining.
Conclusions
Both diffusion-weighted and SPIO-enhanced MRI can distinguish tumor metastatic lymph nodes from reactive lymph nodes. However, neither method is able to detect tumor metastasis to the draining lymph nodes at early stages.




Similar content being viewed by others
References
Kim SH, Kim SC, Choi BI, Han MC (1994) Uterine cervical carcinoma: evaluation of pelvic lymph node metastasis with MR imaging. Radiology 190:807–811
Oyen RH, Van Poppel HP, Ameye FE et al (1994) Lymph node staging of localized prostatic carcinoma with CT and CT-guided fine-needle aspiration biopsy: prospective study of 285 patients. Radiology 190:315–322
Lei J, Xue HD, Li Z, Li S, Jin ZY (2010) Possible pathological basis for false diagnoses of lymph nodes by USPIO-enhanced MRI in rabbits. J Magn Reson Imaging 31:1428–1434
King AD, Ahuja AT, Yeung DK et al (2007) Malignant cervical lymphadenopathy: diagnostic accuracy of diffusion-weighted MR imaging. Radiology 245:806–813
Klerkx WM, Geldof AA, Heintz AP et al (2011) Longitudinal 3.0T MRI analysis of changes in lymph node volume and apparent diffusion coefficient in an experimental animal model of metastatic and hyperplastic lymph nodes. J Magn Reson Imaging 33:1151–1159
Lin G, Ho KC, Wang JJ et al (2008) Detection of lymph node metastasis in cervical and uterine cancers by diffusion-weighted magnetic resonance imaging at 3T. J Magn Reson Imaging 28:128–135
Le Bihan D (1995) Molecular diffusion, tissue microdynamics and microstructure. NMR Biomed 8:375–386
Roy C, Bierry G, Matau A, Bazille G, Pasquali R (2010) Value of diffusion-weighted imaging to detect small malignant pelvic lymph nodes at 3 T. Eur Radiol 20:1803–1811
Patterson DM, Padhani AR, Collins DJ (2008) Technology insight: water diffusion MRI—a potential new biomarker of response to cancer therapy. Nat Clin Pract Oncol 5:220–233
Kwee TC, Takahara T, Ochiai R, Nievelstein RA, Luijten PR (2008) Diffusion-weighted whole-body imaging with background body signal suppression (DWIBS): features and potential applications in oncology. Eur Radiol 18:1937–1952
Abdel Razek AA, Soliman NY, Elkhamary S, Alsharaway MK, Tawfik A (2006) Role of diffusion-weighted MR imaging in cervical lymphadenopathy. Eur Radiol 16:1468–1477
Eiber M, Beer AJ, Holzapfel K et al (2010) Preliminary results for characterization of pelvic lymph nodes in patients with prostate cancer by diffusion-weighted MR-imaging. Invest Radiol 45:15–23
Sumi M, Sakihama N, Sumi T et al (2003) Discrimination of metastatic cervical lymph nodes with diffusion-weighted MR imaging in patients with head and neck cancer. AJNR Am J Neuroradiol 24:1627–1634
Sumi M, Van Cauteren M, Nakamura T (2006) MR microimaging of benign and malignant nodes in the neck. AJR Am J Roentgenol 186:749–757
Anzai Y, Piccoli CW, Outwater EK et al (2003) Evaluation of neck and body metastases to nodes with ferumoxtran 10-enhanced MR imaging: phase III safety and efficacy study. Radiology 228:777–788
Harisinghani MG, Barentsz J, Hahn PF et al (2003) Noninvasive detection of clinically occult lymph-node metastases in prostate cancer. N Engl J Med 348:2491–2499
Pultrum BB, van der Jagt EJ, van Westreenen HL et al (2009) Detection of lymph node metastases with ultrasmall superparamagnetic iron oxide (USPIO)-enhanced magnetic resonance imaging in oesophageal cancer: a feasibility study. Cancer Imaging 9:19–28
Yu MK, Park J, Jon S (2012) Targeting strategies for multifunctional nanoparticles in cancer imaging and therapy. Theranostics 2:3–44
Huang J, Zhang X, Wang L, Yang L, Mao H (2012) Improving the magnetic resonance imaging contrast and detection methods with engineered magnetic nanoparticles. Theranostics 2:86–102
Lim SW, Kim HW, Jun HY et al (2011) TCL-SPION-enhanced MRI for the detection of lymph node metastasis in murine experimental model. Acad Radiol 18:504–511
Vassallo P, Matei C, Heston WD, McLachlan SJ, Koutcher JA, Castellino RA (1994) AMI-227-enhanced MR lymphography: usefulness for differentiating reactive from tumor-bearing lymph nodes. Radiology 193:501–506
Aslakson CJ, Miller FR (1992) Selective events in the metastatic process defined by analysis of the sequential dissemination of subpopulations of a mouse mammary tumor. Cancer Res 52:1399–1405
Krzystyniak K, Kozlowska E, Desjardins R et al (1995) Different T-cell activation by streptozotocin and Freund's adjuvant in popliteal lymph node (PLN). Int J Immunopharmacol 17:189–196
Zhang F, Niu G, Lin X et al (2011) Imaging tumor-induced sentinel lymph node lymphangiogenesis with LyP-1 peptide. Amino Acids. doi:10.1007/s00726-011-0976-1
de Bondt RB, Hoeberigs MC, Nelemans PJ et al (2009) Diagnostic accuracy and additional value of diffusion-weighted imaging for discrimination of malignant cervical lymph nodes in head and neck squamous cell carcinoma. Neuroradiology 51:183–192
Holzapfel K, Duetsch S, Fauser C, Eiber M, Rummeny EJ, Gaa J (2009) Value of diffusion-weighted MR imaging in the differentiation between benign and malignant cervical lymph nodes. Eur J Radiol 72:381–387
Vandecaveye V, De Keyzer F, Vander Poorten V et al (2009) Head and neck squamous cell carcinoma: value of diffusion-weighted MR imaging for nodal staging. Radiology 251:134–146
Kwee TC, Takahara T, Ochiai R et al (2010) Complementary roles of whole-body diffusion-weighted MRI and 18F-FDG PET: the state of the art and potential applications. J Nucl Med 51:1549–1558
Dietrich O, Heiland S, Benner T, Sartor K (2000) Reducing motion artefacts in diffusion-weighted MRI of the brain: efficacy of navigator echo correction and pulse triggering. Neuroradiology 42:85–91
Unger JB, Ivy JJ, Ramaswamy MR, Charrier A, Connor P (2005) Whole-body [18F]fluoro-2-deoxyglucose positron emission tomography scan staging prior to planned radical hysterectomy and pelvic lymphadenectomy. Int J Gynecol Cancer 15:1060–1064
Kim JK, Kim KA, Park BW, Kim N, Cho KS (2008) Feasibility of diffusion-weighted imaging in the differentiation of metastatic from nonmetastatic lymph nodes: early experience. J Magn Reson Imaging 28:714–719
Weissleder R, Elizondo G, Wittenberg J, Lee AS, Josephson L, Brady TJ (1990) Ultrasmall superparamagnetic iron oxide: an intravenous contrast agent for assessing lymph nodes with MR imaging. Radiology 175:494–498
Weissleder R, Elizondo G, Josephson L et al (1989) Experimental lymph node metastases: enhanced detection with MR lymphography. Radiology 171:835–839
Herborn CU, Lauenstein TC, Vogt FM, Lauffer RB, Debatin JF, Ruehm SG (2002) Interstitial MR lymphography with MS-325: characterization of normal and tumor-invaded lymph nodes in a rabbit model. AJR Am J Roentgenol 179:1567–1572
Oghiso Y, Matsuoka O (1983) Time-dependent changes of microscopic localization of intravenously administered colloidal carbon particles in mouse lymph nodes. J Toxicol Sci 8:291–300
Acknowledgments
This project was supported in part by the Intramural Research Program of the National Institute of Biomedical Imaging and Bioengineering (NIBIB), National Institutes of Health (NIH), and the International Cooperative Program of the National Science Foundation of China (NSFC) (81028009).
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Zhang, F., Zhu, L., Huang, X. et al. Differentiation of Reactive and Tumor Metastatic Lymph Nodes with Diffusion-weighted and SPIO-Enhanced MRI. Mol Imaging Biol 15, 40–47 (2013). https://doi.org/10.1007/s11307-012-0562-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-012-0562-2