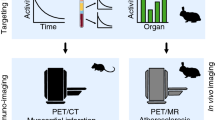
Abstract
Purpose
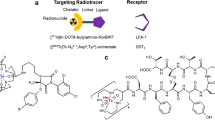
Rupture-prone atherosclerotic plaques are characterized by accumulation of macrophages, which have shown to express somatostatin type 2 receptors. We aimed to investigate whether somatostatin receptor-targeting positron emission tomography (PET) tracers, [68Ga]DOTANOC, [18F]FDR-NOC, and [68Ga]DOTATATE, can detect inflamed atherosclerotic plaques.
Procedures
Atherosclerotic IGF-II/LDLR−/−ApoB100/100 mice were studied in vivo and ex vivo for tracer uptake into atherosclerotic plaques. Furthermore, [68Ga]DOTANOC and [68Ga]DOTATATE were compared in a head-to-head setting for in vivo PET/X-ray computed tomography (CT) imaging characteristics.
Results
Ex vivo uptake of [68Ga]DOTANOC and [68Ga]DOTATATE in the aorta was higher in atherosclerotic mice compared to control C57Bl/6N mice, while the aortic uptake of [18F]FDR-NOC showed no genotype difference. Unlike [18F]FDR-NOC, [68Ga]DOTANOC and [68Ga]DOTATATE showed preferential binding to atherosclerotic plaques with plaque-to-wall ratio of 1.7 ± 0.3 and 2.1 ± 0.5, respectively. However, the aortic uptake and aorta-to-blood ratio of [68Ga]DOTANOC were higher compared to [68Ga]DOTATATE in in vivo PET/CT imaging.
Conclusion
Our results demonstrate superior applicability for [68Ga]DOTANOC and [68Ga]DOTATATE in the detection of atherosclerotic plaques compared to [18F]FDR-NOC.






Similar content being viewed by others
References
Weber C, Noels H (2011) Atherosclerosis: current pathogenesis and therapeutic options. Nat Med 17:1410–1422
Hansson GK, Libby P (2006) The immune response in atherosclerosis: a double-edged sword. Nat Rev Immunol 6:508–519
Moore KJ, Sheedy FJ, Fisher EA (2013) Macrophages in atherosclerosis: a dynamic balance. Nat Rev Immunol 13:709–721
Swirski FK, Nahrendorf M (2013) Leukocyte behavior in atherosclerosis, myocardial infarction, and heart failure. Science 339:161–166
Langer HF, Haubner R, Pichler BJ, Gawaz M (2008) Radionuclide imaging: a molecular key to the atherosclerotic plaque. J Am Coll Cardiol 52:1–12
Tarkin JM, Joshi FR, Rudd JH (2014) PET imaging of inflammation in atherosclerosis. Nat Rev Cardiol 11:443–457
Rudd JH, Narula J, Strauss HW et al (2010) Imaging atherosclerotic plaque inflammation by fluorodeoxyglucose with positron emission tomography: ready for prime time? J Am Coll Cardiol 55:2527–2535
Ogawa M, Ishino S, Mukai T et al (2004) 18F-FDG accumulation in atherosclerotic plaques: immunohistochemical and PET imaging study. J Nucl Med 45:1245–1250
Zhang Z, Machac J, Helft G et al (2006) Non-invasive imaging of atherosclerotic plaque macrophage in a rabbit model with F-18 FDG PET: a histopathological correlation. BMC Nucl Med 6:3
Tahara N, Kai H, Ishibashi M et al (2006) Simvastatin attenuates plaque inflammation: evaluation by fluorodeoxyglucose positron emission tomography. J Am Coll Cardiol 48:1825–1831
Aziz K, Berger K, Claycombe K et al (2008) Noninvasive detection and localization of vulnerable plaque and arterial thrombosis with computed tomography angiography/positron emission tomography. Circulation 117:2061–2070
Paulmier B, Duet M, Khayat R et al (2008) Arterial wall uptake of fluorodeoxyglucose on PET imaging in stable cancer disease patients indicates higher risk for cardiovascular events. J Nucl Cardiol 15:209–217
Buettner C, Rudd JH, Fayad ZA (2011) Determinants of FDG uptake in atherosclerosis. JACC Cardiovasc Imaging 4:1302–1304
Rominger A, Saam T, Vogl E et al (2010) In vivo imaging of macrophage activity in the coronary arteries using 68Ga-DOTATATE PET/CT: correlation with coronary calcium burden and risk factors. J Nucl Med 51:193–197
Li X, Bauer W, Kreissl MC et al (2013) Specific somatostatin receptor II expression in arterial plaque: 68Ga-DOTATATE autoradiographic, immunohistochemical and flow cytometric studies in apoE-deficient mice. Atherosclerosis 230:33–39
Li X, Samnick S, Lapa C et al (2012) 68Ga-DOTATATE PET/CT for the detection of inflammation of large arteries: correlation with 18F-FDG, calcium burden and risk factors. EJNMMI Res 2:52
Wild D, Mäcke HR, Waser B et al (2005) 68Ga-DOTANOC: a first compound for PET imaging with high affinity for somatostatin receptor subtypes 2 and 5. Eur J Nucl Med Mol Imaging 32:724
Wild D, Schmitt JS, Ginj M et al (2003) DOTA-NOC, a high-affinity ligand of somatostatin receptor subtypes 2, 3 and 5 for labelling with various radiometals. Eur J Nucl Med Mol Imaging 30:1338–1347
Kiviniemi A, Gardberg M, Autio A et al (2014) Feasibility of experimental BT4C glioma models for somatostatin receptor 2-targeted therapies. Acta Oncol 53:1125–1134
Heinonen SE, Leppänen P, Kholová I et al (2007) Increased atherosclerotic lesion calcification in a novel mouse model combining insulin resistance, hyperglycemia, and hypercholesterolemia. Circ Res 101:1058–1067
Silvola JM, Saraste A, Laitinen I et al (2011) Effects of age, diet, and type 2 diabetes on the development and FDG uptake of atherosclerotic plaques. JACC Cardiovasc Imaging 4:1294–1301
Körner M, Waser B, Schonbrunn A et al (2012) Somatostatin receptor subtype 2A immunohistochemistry using a new monoclonal antibody selects tumors suitable for in vivo somatostatin receptor targeting. Am J Surg Pathol 36:242–252
Belosi F, Cicoria G, Lodi F et al (2013) Generator breakthrough and radionuclidic purification in automated synthesis of 68Ga-DOTANOC. Curr Radiopharm 6:72–77
Li XG, Autio A, Ahtinen H et al (2013) Translating the concept of peptide labeling with 5-deoxy-5-[18F]fluororibose into preclinical practice: 18F-labeling of Siglec-9 peptide for PET imaging of inflammation. Chem Commun (Camb) 49:3682–3684
Li XG, Helariutta K, Roivainen A et al (2014) Using 5-deoxy-5-[18F]fluororibose to glycosylate peptides for positron emission tomography. Nat Protoc 9:138–145
Rinne P, Silvola JM, Hellberg S et al (2014) Pharmacological activation of the melanocortin system limits plaque inflammation and ameliorates vascular dysfunction in atherosclerotic mice. Arterioscler Thromb Vasc Biol 34:1346–1354
Silvola JM, Laitinen I, Sipilä HJ et al (2011) Uptake of 68gallium in atherosclerotic plaques in LDLR−/−ApoB100/100 mice. EJNMMI Res 1:14
Patel YC (1999) Somatostatin and its receptor family. Front Neuroendocrinol 20:157–198
Adams RL, Adams IP, Lindow SW et al (2005) Somatostatin receptors 2 and 5 are preferentially expressed in proliferating endothelium. Br J Cancer 92:1493–1498
Armani C, Catalani E, Balbarini A et al (2007) Expression, pharmacology, and functional role of somatostatin receptor subtypes 1 and 2 in human macrophages. J Leukoc Biol 81:845–855
Schatka I, Wollenweber T, Haense C et al (2013) Peptide receptor-targeted radionuclide therapy alters inflammation in atherosclerotic plaques. J Am Coll Cardiol 62:2344–2345
Yudkin JS, Kumari M, Humphries SE, Mohamed-Ali V (2000) Inflammation, obesity, stress and coronary heart disease: is interleukin-6 the link? Atherosclerosis 148:209–214
Schuett H, Oestreich R, Waetzig GH et al (2012) Transsignaling of interleukin-6 crucially contributes to atherosclerosis in mice. Arterioscler Thromb Vasc Biol 32:281–290
Sheikine Y, Hansson GK (2004) Chemokines and atherosclerosis. Ann Med 36:98–118
Wallace AM, McMahon AD, Packard CJ et al (2001) Plasma leptin and the risk of cardiovascular disease in the west of Scotland coronary prevention study (WOSCOPS). Circulation 104:3052–3056
Koh KK, Park SM, Quon MJ (2008) Leptin and cardiovascular disease: response to therapeutic interventions. Circulation 117:3238–3249
Ducimetiere P, Eschwege E, Papoz L et al (1980) Relationship of plasma insulin levels to the incidence of myocardial infarction and coronary heart disease mortality in a middle-aged population. Diabetologia 19:205–210
Stout RW (1990) Insulin and atheroma. 20-yr perspective. Diabetes Care 13:631–654
Acknowledgments
The authors wish to thank Riikka Siitonen and Aake Honkaniemi for assistance in mouse experiments and Erica Nyman and Sinikka Collanus for technical assistance in tissue sectioning and immunohistochemistry. This study was conducted within the Finnish Center of Excellence in Cardiovascular and Metabolic Diseases supported by the Academy of Finland, the University of Turku, the Turku University Hospital, and the Åbo Akademi University.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Petteri Rinne and Sanna Hellberg contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 129 kb).
Rights and permissions
About this article
Cite this article
Rinne, P., Hellberg, S., Kiugel, M. et al. Comparison of Somatostatin Receptor 2-Targeting PET Tracers in the Detection of Mouse Atherosclerotic Plaques. Mol Imaging Biol 18, 99–108 (2016). https://doi.org/10.1007/s11307-015-0873-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-015-0873-1