Abstract
Purpose
Tryptophan metabolism via indoleamine 2,3-dioxygenase (IDO)-mediated kynurenine pathway plays a role in immunomodulation and has been emerging as a plausible target for cancer immunotherapy. Imaging IDO-mediated kynurenine pathway of tryptophan metabolism with positron emission tomography (PET) could provide valuable information for noninvasive assessment of cancer immunotherapy response. In this work, radiotracer 1-(2-[18F]fluoroethyl)-L-tryptophan (1-L-[18F]FETrp) and its enantioisomer 1-D-[18F]FETrp were synthesized and evaluated for PET imaging of IDO-mediated kynurenine pathway of tryptophan metabolism.
Procedures
Enantiopure 1-L-[18F]FETrp and 1-D-[18F]FETrp were prepared by a nucleophilic reaction of N-boc-1-(2-tosylethyl) tryptophan tert-butyl ester with [18F]Fluoride, followed by acid hydrolysis in a GE Tracerlab FX-N module. In vitro cell uptake assays were performed with a breast cancer cell line MDA-MB-231. Small animal PET/computed tomography (CT) imaging was carried out in a mouse model bearing MDA-MB-231 xenografts.
Results
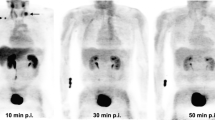
Automatic radiosynthesis of 1-L-[18F]FETrp and 1-D-[18F]FETrp was achieved by a one-pot two-step approach in 19.0 ± 7.0 and 9.0 ± 3.0 % (n = 3) decay-corrected yield with radiochemical purity over 99 %, respectively. In vitro cell uptake study indicated the uptake of 1-D-[18F]FETrp in MDA-MB-231 cells was 0.73 ± 0.07 %/mg of protein at 60 min, while, the corresponding uptake of 1-L-[18F]FETrp was 6.60 ± 0.77 %/mg. Further mechanistic assays revealed that amino acid transport systems L-tpye amino acid transporter (LAT) and alanine-, serine-, and cysteine-preferring (ASC), and enzyme IDO expression were involved in cell uptake of 1-L-[18F]FETrp. Small animal PET/CT imaging study showed the tumor uptake of 1-L-[18F]FETrp was 4.6 ± 0.4 % ID/g, while, the tumor uptake of 1-D-[18F]FETrp was low to 1.0 ± 0.2 % ID/g, which were confirmed by ex vivo biodistribution study.
Conclusions
We have developed a practical method for the automatic radiosynthesis of 1-L-[18F]FETrp and 1-D-[18F]FETrp. Our biological evaluation results suggest that 1-L-[18F]FETrp is a promising radiotracer for PET imaging of IDO-mediated kynurenine pathway of tryptophan metabolism in cancer.






Similar content being viewed by others
References
Vécsei L, Szalárdy L, Fülöp F, Toldi J (2013) Kynurenines in the CNS: recent advances and new questions. Nat Rev Drug Discov 12:64–82
Juhász C, Nahleh Z, Zitron I, Chugani DC et al (2012) Tryptophan metabolism in breast cancers: molecular imaging and immunohistochemistry studies. Nucl Med Biol 39:926–932
Uyttenhove C, Pilotte L, Théate I et al (2003) Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nat Med 9:1269–1274
Karanikas V, Zamanakou M, Kerenidi T et al (2007) Indoleamine 2,3-dioxygenase (IDO) expression in lung cancer. Cancer Biol Ther 6:1258–1262
Yoshikawa T, Hara T, Tsurumi H et al (2010) Serum concentration of L-kynurenine predicts the clinical outcome of patients with diffuse large B-cell lymphoma treated with R-CHOP. Eur J Haematol 84:304–309
Godin-Ethier J, Hanafi LA, Piccirillo CA, Lapointe R (2011) Indoleamine 2,3-dioxygenase expression in human cancers: clinical and immunologic perspectives. Clin Cancer Res 17:6985–6991
Liu XQ, Lu K, Feng LL et al (2014) Up-regulated expression of indoleamine 2,3-dioxygenase 1 in non-Hodgkin lymphoma correlates with increased regulatory T-cell infiltration. Leuk Lymphoma 55:405–414
Munn DH, Melloe AL (2016) IDO in the tumor microenvironment: inflammation, counter-regulation, and tolerance. Trends Immunol 37:193–207
Ametamey SM, Honer M, Schubiger PA (2008) Molecular imaging with PET. ChemRev 108:1501–1516
Batista CEA, Juhász C, Muzik O et al (2009) Imaging correlates of differential expression of indoleamine 2,3-dioxygenase in human brain tumors. Mol Imaging Biol 11:460–466
Chakruborty PK, Mangner TJ, Chugani DC et al (1996) A high-yield and simplified procedure for the synthesis of α-[11C]methyl-L-tryptophan. Nucl Med Biol 23:1005–1008
Huang X, Xiao X, Gillies RJ, Tian H (2016) Design and automated production of 11C-alpha-methyl-L-tryptophan (11C-AMT). Nucl Med Biol 43:303–308
Chiotellis A, Mu L, Müller A et al (2013) Synthesis and biological evaluation of 18F-labeled fluoropropyl tryptophan analogs as potential PET probes for tumor imaging. Eur J Med Chem 70:768–780
Sun T, Tang G, Tian H et al (2012) Radiosynthesis of 1-[18F]fluoroethyl-L-tryptophan as a novel potential amino acid PET tracer. Appl Radiat Isot 70:676–680
Henrottin J, Zervosen A, Lemaire C et al (2015) N1-Fluoroalkyltryptophan analogues: synthesis and in vitro study as potential substrates for indoleamine 2,3-dioxygenase. ACS Med Chem Lett 6:260–265
Burger IA, Zitzmann-Kolbe S, Pruim J et al (2014) First clinical results of (D)-18F-fluoromethyltyrosine (BAY 86-9596) PET/CT in patients with non-small cell lung cancer and head and neck squamous cell carcinoma. J Nucl Med 55:1778–1785
Langen KJ, Hamacher K, Bauer D et al (2005) Preferred stereoselective transport of the D-isomer of cis-4-[18F]fluoro-proline at the blood-brain barrier. J Cereb Blood Flow Metab 25:607–616
Tsukada H, Sato K, Fukumoto D et al (2006) Evaluation of D-isomers of O-11C-methyl tyrosine and O-18F-fluoromethyl tyrosine as tumor-imaging agents in tumor-bearing mice. J Nucl Med 47:679–688
Henrottin J, Lemaire C, Egrise D et al (2016) Fully automated radiosynthesis of 1-[18F]FETrp, a potential substrate for indoleamine 2,3-dioxygenase PET imaging. Nucl Med Biol 43:379–389
Zimnitsky DS, Yurkshtovich TL, Bychkovsky PM (2006) Adsorption of Zwitterionic drugs on oxidized cellulose from aqueous solutions. React Funct Polym 295:33–40
Travers MT, Gow IF, Barber MC et al (2004) Indoleamine 2,3-dioxygenase activity and L-tryptophan transport in human breast cancer cells. Biochim Biophys Acta 1661:106–112
Adams JL, Smothers J, Srinivasan R, Hoos A (2015) Big opportunities for small molecules in immuno-oncology. Nat Rev Drug Discov 14:603–622
Wang Q, Holst J (2015) L-type amino acid transport and cancer: targeting the mTORC1 pathway to inhibit neoplasia. Am J Cancer Res 5:1281–1294
Zhao Y, Wang L, Pan J (2015) The role of L-type amino acid transporter 1 in human tumors. Intractable Rare Dis Res 4:165–169
McConathy J, Goodman MM (2008) Non-natural amino acids for tumor imaging using positron emission tomography and single photon emission computed tomography. Cancer Metastasis Rev 27:555–573
Kramer SD, Mu L, Muller A et al (2012) 5-(2-18F-Fluoroethoxy)-L-tryptophan as a substrate of system L transport for tumor imaging by PET. J Nucl Med 53:434–442
Fuchs BC, Bode BP (2005) Amino acid transporters ASCT2 and LAT1 in cancer: partners in crime? Semin Cancer Biol 15:254–266
Wander SA, Hennessy BT, Slingerland JM (2011) Next-generation mTOR inhibitors in clinical oncology: how pathway complexity informs therapeutic strategy. J Clin Invest 121:1231–1241
Xie L, Maeda J, Kumata K et al (2016) Development of 1-N-11C-methyl-L- and -D-tryptophan for pharmacokinetic imaging of the immune checkpoint inhibitor 1-methyl-tryptophan. Sci Rep 5:1–9
Acknowledgments
We would like to thank Prof. Xiankai Sun for providing insight that greatly assisted our work, Robert Hallgren for producing 18F for radiochemistry study, and Hussein Diab for acquiring and analyzing the micro-PET/CT imaging study. We also thank Drs. William Silvers, Guiyang Hao, and Aditi Mulgaonkar for their assistance in this study. This work was supported by Cancer Prevention Research Institute of Texas (CPRIT) Grant (RP110771), the Simmons Cancer Center Grant (NIH 5P30 CA 142543), and American Cancer Society and the Simmons Cancer Center (ACS-IRG-02-196).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 1712 kb)
Rights and permissions
About this article
Cite this article
Xin, Y., Cai, H. Improved Radiosynthesis and Biological Evaluations of L- and D-1-[18F]Fluoroethyl-Tryptophan for PET Imaging of IDO-Mediated Kynurenine Pathway of Tryptophan Metabolism. Mol Imaging Biol 19, 589–598 (2017). https://doi.org/10.1007/s11307-016-1024-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11307-016-1024-z