Abstract
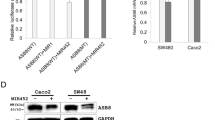
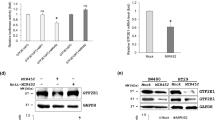
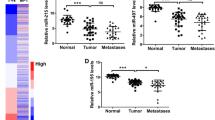
MicroRNAs are small single-stranded RNA molecules consisting of approximately 22 nucleotides (nt), and have post-transcriptional regulatory functions. By gene chip screening, we previously showed that miR-142-3p was significantly upregulated in colorectal cancer tissue and was associated with clinicopathological features, compared with matched non-tumor tissue. In this study, we confirmed significant upregulation of miR-142-3p in 60 colorectal cancer samples and three colorectal cancer cell lines by quantitative real-time PCR (qRT-PCR). Using software and network resources, we predicted TCF7 as a target of miR-142-3p, which we confirmed with dual-luciferase assays. By RT-PCR and Western blot analysis, we found miR-142-3p negatively regulates TCF7 expression post-transcriptionally. CCK8 assays and growth curves indicated that overexpression of miR-142-3p in SW480 colorectal cancer cells potently inhibited cell proliferation in vitro. The expression of TCF7 mRNA and protein was upregulated in both colorectal cancer tissues and colorectal cancer cell lines, closely correlating with its function as an oncogene in promoting tumor cell proliferation and inhibiting apoptosis. With TCF7 as a direct target, our results suggested that miR-142-3p may be involved in the regulation of cell proliferation in colorectal cancer.
Article PDF
Similar content being viewed by others
References
Rebecca S, Deepa N, Ahmedin J. Cancer Statistics, 2012. CA Cancer J Clin, 2012, 62: 10–29
Liu J. Control of protein synthesis and mRNA degradation by microRNAs. Curr Opin Cell Biol, 2008, 20: 214–221
Mattick J S, Makunin I V. Non-coding RNA. Hum Mol Genet, 2006, 15: R17–29
Lee R C, Feinbaum R L, Ambros V. The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell, 1993, 75: 843–854
Ambros V. The functions of animal microRNAs. Nature, 2004, 431: 350–355
Hwang H W, Mendell J T. MicroRNAs in cell proliferation, cell death, and tumorig-enesis. Br J Cancer, 2006, 94: 776–780
Bartel D P. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell, 2004, 116: 281–297
Wan H Y, Guo L M, Liu T, et al. Regulation of the transcription factor NF-kappaB1 by microRNA-9 in human gastric adenocarcinoma. Mol Cancer, 2010, 9: 16
Calin G A, Dumitru C D, Shimizu M, et al. Frequent deletions and down-regulation of micro-RNA genes miR15 and miR16 at 13q14 in chronic lymphocytic leukemia. Proc Natl Acad Sci USA, 2002, 99: 15524–15529
Bandi N, Zbinden S, Gugger M, et al. MiR-15a and miR-16 are implicated in cell cycle regulation in a Rb-dependent manner and are frequently deleted or down-regulated in non-small cell lung cancer. Cancer Res, 2009, 69: 5553–5559
Bonci D, Coppola V, Musumeci M, et al. The miR-15a-miR-16-1 cluster controls prostate cancer by targeting multiple oncoge-nic activities. Nat Med, 2008, 14: 1271–1277
Dyrskjøt L, Ostenfeld M S, Bramsen J B, et al. Genomic profiling of microRNAs in bladder cancer: miR-129 is associated with poor outcome and promotes cell death in vitro. Cancer Res, 2009, 69: 4851–486
Su H, Yang J R, Xu T, et al. Down-regulated in hepatocellular carcinoma, promotes apoptosis and suppresses tumorigenicity. Cancer Res, 2009, 69: 1135–1142
Foley N H, Bray I M, Tivnan A, et al. MicroRNA-184 inhibits neuroblastoma cell survival through targeting the serine/threonine kinase AKT2. Mol Cancer, 2010, 9: 83
Tazawa H, Tsuchiya N, Izumiya M, et al. Tumorsuppressive miR-34a induces senescence-like growth arrest through modulation of the E2F pathway in human colon cancer cells. Proc Natl Acad Sci USA, 2007, 104: 15472–15477
Bonci D, Coppola V, Musumeci M, et al. The miR-15a-miR-16-1 cluster controls prostate cancer by targeting multiple oncogenic activities. Nat Med, 2008, 14: 1271–1277
Nasser M W, Datta J, Nuovo G, et al. Down-regulation of micro-RNA-1 (miR-1) in lung cancer. Suppression of tumorigenic property of lung cancer cells and their sensitization to doxorubicin-induced apoptosis by miR-1. J Biol Chem, 2008, 283: 33394–33405
Ma L, Teruya-Feldstein J, Weinberg R A. Tumour invasion and metastasis initiated by microRNA-10b in breast cancer. Nature, 2007, 449: 682–688
Yang H, Kong W, He L, et al. MicroRNA expression profiling in human ovarian cancer: MiR-214 induces cell survival and cisplatin resistance by targeting PTEN. Cancer Res, 2008, 68: 425–433
Yao J, Liang L, Huang S, et al. MicroRNA-30d promotes tumor invasion and metastasis by targeting Galphai2 in hepatocellular carcinoma. Hepatology, 2010, 51: 846–856
Chen W C, Lin M S, Huang J X, et al. Clinical significance of aberrant microRNAs expression in human colorectal cancer. World J Gastroenterol, 2010, 18: 3187–3194
Lv M, Zhang X, Jia H, et al. An oncogenic role of miR-142-3p in human T-cell acute lymphoblastic leukemia (T-ALL) by targeting glucocorticoid receptor-α and cAMP/PKA pathways. Leukemia, 2012, 26: 769–777
Wu L, Cai C, Wang X, et al. MicroRNA-142-3p, a new regulator of RAC1, suppresses the migration and invasion of hepatocellular carcinoma cells. FEBS Lett, 2011, 585: 1322–1330
Lin R J, Xiao D W, Liao L D, et al. MiR-142-3p as a potential prognostic biomarker for esophageal squamous cell carcinoma. J Surg Oncol, 2012, 105: 175–182
Griffiths-Jones S, Grocock R J, van Dongen S, et al. MiRBase: MicroRNA sequences, targets and gene nomenclature. Nucleic Acids Res, 2006, 34: D140–144
Lewis B P, Shih I H, Jones-Rhoades M W, et al. Prediction of mammalian microRNA targets. Cell, 2003, 115: 787–798
Krek A, Grün D, Poy M N, et al. Combinatorial microRNA target predictions. Nat Genet, 2005, 37: 495–500
Hosack D A, Dennis G Jr, Sherman B T, et al. Identifying biological themes within lists of genes with EASE. Genome Biol, 2003, 4: 70–77
Yan L X, Huang X F, Shao Q, et al. MicroRNA miR-21 overexpression in human breast cancer is associated with advanced clinical stage, lymph node metastasis and patient poor prognosis. RNA, 2008, 14: 2348–2360
Chekulaeva M, Filipowicz W. Mechanisms of miRNA-mediated post-transcriptional regulation in animal cells. Curr Opin Cell Biol, 2009, 21: 452–460
Sethupathy P, Megraw M, Hatzigeorgiou A G. A guide through present computational approaches for the identification of mammalian microRNA targets. Nat Methods, 2006, 3: 881–886
Logan C Y, Nusse R. The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol, 2004, 20: 781–810
Taketo M M. Shutting down Wnt signal-activated cancer. Nat Genet, 2004, 36: 320–322
MacDonald B T, Tamai K, He X. Wnt/beta-catenin signaling: Components, mechanisms, and diseases. Dev Cell, 2009, 17: 9–26
Author information
Authors and Affiliations
Corresponding author
Additional information
These authors contributed equally to this work.
This article is published with open access at Springerlink.com
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Zhou, J., Jiang, Z., Wang, Z. et al. MicroRNA-142-3p is frequently upregulated in colorectal cancer and may be involved in the regulation of cell proliferation. Chin. Sci. Bull. 58, 2836–2845 (2013). https://doi.org/10.1007/s11434-013-5937-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11434-013-5937-5