Abstract
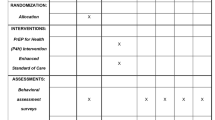
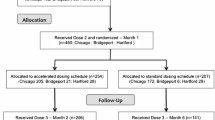
Hepatitis C virus (HCV) is hyperendemic among injection drug users (IDUs). However, few scientifically proven interventions to prevent secondary transmission of HCV from infected IDUs to others exist. This report describes the design, feasibility, and baseline characteristics of participants enrolled in the Study to Reduce Intravenous Exposure (STRIVE). STRIVE was a multisite, randomized-control trial to test a behavioral intervention developed to reduce distribution of used injection equipment (needles, cookers, cottons, and rinse water) and increase health-care utilization among antibody HCV (anti-HCV) positive IDUs. STRIVE enrolled anti-HCV positive IDU in Baltimore, New York City, and Seattle; participants completed behavioral assessments and venipuncture for HIV, HCV-RNA, and liver function tests (LFTs) and were randomized to attend either a six-session, small-group, peer-mentoring intervention workshop or a time-matched, attention-control condition. Follow-up visits were conducted at 3 and 6 months. At baseline, of the 630 HCV-positive IDUs enrolled (mean age of 26 years, 60% white, 76% male), 55% reported distributive needle sharing, whereas 74, 69, and 69% reported sharing cookers, cottons, and rinse water, respectively. Health-care access was low, with 41% reporting an emergency room as their main source of medical care. Among those enrolled, 66% (418/630) were randomized: 53% (222/418) and 47% (196/418) to the intervention and control conditions, respectively. Follow-up rates were 70 and 73% for the 3- and 6-month visits, respectively. As distributive sharing of used injection equipment was common while reports of receiving HCV care were low, these findings indicate an urgent need for HCV-related interventions with IDUs and demonstrate the acceptability and feasibility to do so.

Similar content being viewed by others
References
NIH consensus statement on management of hepatitis C: 2002. NIH Consens State Sci Statements. 2002;19:1–46.
Williams I. Epidemiology of hepatitis C in the United States. Am J Med. 1999;107:2S–9S.
Yen T, Keeffe EB, Ahmed A. The epidemiology of hepatitis C virus infection. J Clin Gastroenterol. 2003;36:47–53.
Alter MJ, Mast EE, Moyer LA, Margolis HS. Hepatitis C. Infect Dis Clin North Am. 1998;12:13–26.
Thorpe LE, Ouellet LJ, Hershow R, et al. Risk of hepatitis C virus infection among young adult injection drug users who share injection equipment. Am J Epidemiol. 2002;155:645–653.
Garfein RS, Doherty MC, Monterroso ER, Thomas DL, Nelson KE, Vlahov D. Prevalence and incidence of hepatitis C virus infection among young adult injection drug users. J Acquir Immune Defic Syndr Human Retrovirol. 1998;18(Suppl 1):S11–S19.
Thorpe L, Ouellet L, Hershow R, Bailey S, Williams I, Monerosso E. The multiperson use of non-syringe injection equipment and risk of hepatitis c infection in a cohort of young adult injection drug users in Chicago, 1997–1999. Ann Epidemiol. 2000;10:472–473.
Hagan H, Thiede H, Weiss NS, Hopkins SG, Duchin JS, Alexander ER. Sharing of drug preparation equipment as a risk factor for hepatitis C. Am J Public Health. 2001;91:42–46.
Gerberding JL. Management of occupational exposures to blood-borne viruses. N Engl J Med. 1995;332:444–451.
Hagan H, Des Jarlais DC. HIV and HCV infection among injecting drug users. Mt Sinai J Med. 2000;67:423–428.
Stoove MA, Gifford SM, Dore GJ. The impact of injecting drug use status on hepatitis C-related referral and treatment. Drug Alcohol Depend. 2005;77:81–86.
Stephenson J. Former addicts face barriers to treatment for HCV. JAMA. 2001;285:1003–1005.
Strauss SM, Astone JM, Jarlais DD, Hagan H. A comparison of HCV antibody testing in drug-free and methadone maintenance treatment programs in the United States. Drug Alcohol Depend. 2004;73:227–236.
Ompad DC, Fuller CM, Vlahov D, Thomas D, Strathdee SA. Lack of behavior change after disclosure of hepatitis C virus infection among young injection drug users in Baltimore, Maryland. Clin Infect Dis. 2002;35:783–788.
Hagan H, McGough JP, Thiede H, Weiss NS, Hopkins S, Alexander ER. Syringe exchange and risk of infection with hepatitis B and C viruses. Am J Epidemiol. 1999;149:203–213.
Crofts N, Nigro L, Oman K, Stevenson E, Sherman J. Methadone maintenance and hepatitis C virus infection among injecting drug users. Addiction. 1997;92:999–1005.
Strauss SM, Astone JM, Hagan H, Des J. The content and comprehensiveness of hepatitis C education in methadone maintenance and drug-free treatment units. J Urban Health. 2004;81:38–47.
Garfein RS, Latka M, Hagan H, et al. CIDUS-III/Drug Users Intervention Trial (DUIT): A Randomized, Controlled Trial of a Peer-Education Behavioral Intervention to Prevent HIV and Hepatitis C Virus Infection Among 15–30 Year Old Injection Drug Users. Paper Presented at: National HIV Prevention Conference; June 12, 2005; Atlanta, GA.
Alter MJ, Kuhnert WL, Finelli L. Guidelines for laboratory testing and result reporting of antibody to hepatitis C virus. MMWR Morb Mortal Wkly Rep. 2003;52:1–13.
Fishbein M, Ajzen F. Belief, Attitude and Behavior: An Introduction to Theory and Research. Reading, MA: Addison-Wesley; 1975.
Montano DE, Kasprzyk D, Taplin SH. The Theory of Reasoned Action and the Theory of Planned Behavior. In: Glanz K, Lewis FM, Rimer BK, eds. Health Behavior and Health Education. San Francisco, CA: Jossey-Bass Publishers; 1997.
Bandura A. Self-efficacy mechanism in human agency. Am Psychol. 1986;40:359–373.
Bandura A. A Social-Cognitive Approach to the Exercise of Control over AIDS Infection. In: Albee GW, Schneider SF, eds. Primary Prevention of AIDS: Psychological Approaches. Newbury Park, CA: Sage Publications; 1991.
Rothman KJ, Greenland S. Modern Epidemiology, 2nd edition. Philadelphia, PA: Lippincott-Raven; 1998.
Hagan H, Campbell JV, Thiede H, et al. Self-reported hepatitis C virus antibody status and risk behavior in young injectors. Public Health Rep. 2006;121:710–719.
Rosenblum A, Nuttbrock L, McQuistion HL, Magura S, Joseph H. Hepatitis C and substance use in a sample of homeless people in New York City. J Addict Dis. 2001;20:15–25.
Klinkenberg WD, Caslyn RJ, Morse GA, et al. Prevalence of human immunodeficiency virus, hepatitis B, and hepatitis C among homeless persons with co-occurring severe mental illness and substance use disorders. Compr Psychiatry. 2003;44:293–302.
Stein JA, Nyamathi A. Correlates of hepatitis C virus infection in homeless men: a latent variable approach. Drug Alcohol Depend. 2004;75:89–95.
Macalino GE, Vlahov D, Sanford-Colby S, et al. Prevalence and incidence of HIV, hepatitis B virus, and hepatitis C virus infections among males in Rhode Island prisons. Am J Public Health. 2004;94:1218–1223.
Reindollar RW. Hepatitis C and the correctional population. Am J Med. 1999;107:100S–103S.
Samuel MC, Doherty PM, Bulterys M, Jenison SA. Association between heroin use, needle sharing and tattoos received in prison with hepatitis B and C positivity among street-recruited injecting drug users in New Mexico, USA. Epidemiol Infect. 2001;127:475–484.
Gebo KA, Diener-West M, Moore RD. Hospitalization rates differ by hepatitis C status in an urban HIV cohort. J Acquir Immune Defic Syndr. 2003;34:165–173.
Wiegand J, Buggisch P, Boecher W, et al. Early monotherapy with pegylated interferon alpha-2b for acute hepatitis C infection: the HEP-NET acute-HCV-II study. Hepatology. 2006;43:250–256.
Jaeckel E, Cornberg M, Wedemeyer H, et al. Treatment of acute hepatitis C with interferon alfa-2b. N Engl J Med. 2001;345:1452–1457.
Hagan H, Latka MH, Campbell JV, et al. Eligibility for treatment of hepatitis C virus infection among young injection drug users in 3 US cities. Clin Infect Dis. 2006;42:669–672.
Turner CF, Ku L, Rogers SM, Lindberg LD, Pleck JH, Sonenstein FL. Adolescent sexual behavior, drug use, and violence: increased reporting with computer survey technology. Science. 1998;280:867–873.
Des Jarlais DC, Paone D, Milliken J, et al. Audio-computer interviewing to measure risk behaviour for HIV among injecting drug users: a quasi-randomised trial. Lancet. 1999;353:1657–1661.
Metzger DS, Koblin B, Turner C, et al. Randomized controlled trial of audio computer-assisted self-interviewing: utility and acceptability in longitudinal studies. HIVNET Vaccine Preparedness Study Protocol Team. Am J Epidemiol. 2000;152:99–106.
Acknowledgements
The authors gratefully acknowledge the investigators, staff, and participants of the STRIVE project for their total commitment to the success of this project. Additionally, we thank David Purcell from the U.S. Centers for Disease Control for consulting on study design and the investigators of the DUIT study for generously sharing the DUIT study protocol and instruments.
The STRIVE group includes the following individuals:
Steffanie A. Strathdee, Elizabeth T. Golub, David Thomas, Marie Bailey-Kloch, Yvette Bowser, Peter O’Driscoll, Janet Reeves, Marcella Sapun, Dale Netski, McCay Moiforay, Fleesie Hubbard, Coralee Meslin, Karen Yen-Hobelmann, Marie Bailey-Kloch, Eddie Poole, David Hudson, Gina Gant, Eric Hendren from Johns Hopkins University, Baltimore, MD; Mary Latka, Farzana Kapadia, David Vlahov, Danielle Ompad, Micaela Coady, Sebastian Bonner, Joanna Cruz, Sandra DelVecchio, Dirk Jackson, Gregory Malave, Joan Monserrate, Clarisse Miller O’Shea, Manny Yonko from The New York Academy of Medicine, New York, NY; Holly Hagan, Jennifer V. Campbell, Eileen Hough, Hanne Thiede, Rong Lee, Susan Nelson, Jef St. De Lore, Kimberly Houk, Sarah Brooks, Carrie Shriver, Jeanette Frazier, Jean Pass, Paul Swenson from Seattle-King County Public Health, Seattle, WA; and Richard S. Garfein from the University of Califormia at San Diego, San Diego, CA.
Funding for this project was provided by the National Institute on Drug Abuse through grant DA14499. Trial registration: Study to Reduce Intravenous Exposures, NCT00391482, http://clinicaltrials.gov/ct/show/NCT00391482?order=1.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kapadia, F., Latka, M.H., Hagan, H. et al. Design and Feasibility of a Randomized Behavioral Intervention to Reduce Distributive Injection Risk and Improve Health-Care Access Among hepatitisC virus Positive Injection Drug Users: The Study to Reduce Intravenous Exposures (STRIVE). J Urban Health 84, 99–115 (2007). https://doi.org/10.1007/s11524-006-9133-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11524-006-9133-7