Abstract
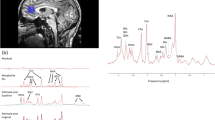
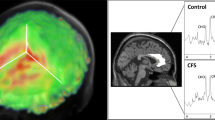
Persistent cancer-related fatigue (PCRF) is one of the most troubling side-effects of breast cancer (BC) treatment. One explanatory model for PCRF is sickness behavior, which is a set of adaptive responses including sleepiness and depressed mood in reaction to an inflammatory trigger. Prior research has investigated differences in inflammatory cytokines between fatigued and non-fatigued BC survivors, but no study has examined differences in brain metabolites. Differences in inflammatory markers, and brain metabolites using proton magnetic resonance spectroscopy were evaluated within 16 fatigued and 13 non-fatigued BC survivors. Fatigued BC survivors had significantly higher ratios of two markers derived from brain metabolites; namely (a) creatine, normalized to total creatine (creatine + phosphocreatine (Cr/tCr)) ratio (P = 0.03) and (b) glutamate + glutamine (Glx) to N-acetyl-aspartate (NAA) ratio (P = 0.01) in the posterior insula compared to non-fatigued breast cancer survivor. Further, serum IL-6 was increased in fatigued women compared to non-fatigued women (P = 0.03), Using receiver operator curves (ROC) we determined that the posterior insula Glx/NAA ratio was the best predictor of fatigue with an overall area under the receiver operating characteristic curve (AUROC) of 79 %, with a sensitivity of 81 % and a specificity of 69 %. However, posterior insula Glx/NAA, Cr/tCr and serum IL-6 were not significantly correlated with one another implying the possibility of independent biological mechanisms for PCRF rather than an interrelated mechanism as represented by the sickness behavior model. This study provides novel preliminary evidence of several distinct neurobiological changes in the posterior insula associated with PCRF in BC survivors. Future, longitudinal studies are needed to explore these distinct biological phenomena where changes through time in peripheral immune markers and brain metabolites are examined to determine if they correlate with changes in fatigue.



Similar content being viewed by others
References
Alexander, S., Minton, O., Andrews, P., & Stone, P. (2009). A comparison of the characteristics of disease-free breast cancer survivors with or without cancer-related fatigue syndrome. European Journal of Cancer, 45(3), 384–392.
Allen, P. J. (2012). Creatine metabolism and psychiatric disorders: does creatine supplementation have therapeutic value? Neuroscience and Biobehavioral Reviews, 36(5), 1442–1462.
American Cancer Society. (2012). Cancer treatment and survivorsip facts & figures 2012–2013. In A. C. Society (Ed.), Atlanta, Georgia.
Benveniste, H., Zhang, S., Reinsel, R. A., Li, H., Lee, H., Rebecchi, M., et al. (2012). Brain metabolomic profiles of lung cancer patients prior to treatment characterized by proton magnetic resonance spectroscopy. International Journal of Clinical and Experimental Medicine, 5(2), 154–164.
Berger, A. M., Kuhn, B. R., Farr, L. A., Lynch, J. C., Agrawal, S., Chamberlain, J., et al. (2009). Behavioral therapy intervention trial to improve sleep quality and cancer-related fatigue. Psychooncology, 18(6), 634–646.
Bjelland, I., Dahl, A. A., Haug, T. T., & Neckelmann, D. (2002). The validity of the hospital anxiety and depression scale. An updated literature review. Journal of Psychosomatic Research, 52(2), 69–77.
Bokemeyer, M., Ding, X. Q., Goldbecker, A., Raab, P., Heeren, M., Arvanitis, D., et al. (2011). Evidence for neuroinflammation and neuroprotection in HCV infection-associated encephalopathy. Gut, 60(3), 370–377.
Boksem, M. A., & Tops, M. (2008). Mental fatigue: costs and benefits. Brain Research Reviews, 59(1), 125–139.
Bower, J. E. (2005). Prevalence and causes of fatigue after cancer treatment: the next generation of research. Journal of Clinical Oncology, 23(33), 8280–8282.
Bower, J. E. (2008). Behavioral symptoms in patients with breast cancer and survivors. Journal of Clinical Oncology, 26(5), 768–777.
Bower, J. E., Ganz, P. A., Aziz, N., & Fahey, J. L. (2002). Fatigue and proinflammatory cytokine activity in breast cancer survivors. Psychosomatic Medicine, 64(4), 604–611.
Bower, J. E., Ganz, P. A., Aziz, N., Fahey, J. L., & Cole, S. W. (2003). T-cell homeostasis in breast cancer survivors with persistent fatigue. Journal of the National Cancer Institute, 95(15), 1165–1168.
Bower, J. E., Ganz, P. A., Irwin, M. R., Kwan, L., Breen, E. C., & Cole, S. W. (2011). Inflammation and behavioral symptoms after breast cancer treatment: do fatigue, depression, and sleep disturbance share a common underlying mechanism? Journal of Clinical Oncology, 29(26), 3517–3522.
Brooks, J. C., Roberts, N., Whitehouse, G., & Majeed, T. (2000). Proton magnetic resonance spectroscopy and morphometry of the hippocampus in chronic fatigue syndrome. British Journal of Radiology, 73(875), 1206–1208.
Cameron, B. A., Bennett, B., Li, H., Boyle, F., Desouza, P., Wilcken, N., et al. (2012). Post-cancer fatigue is not associated with immune activation or altered cytokine production. Annals of Oncology. doi:10.1093/annonc/mds108.
Castillo, M. (2007). Spectroscopy evidence of diffuse brain abnormalities in patients with epileptogenic foci. American Journal of Neuroradiology, 28(6), 1076–1077.
Chang, Y. J., Lee, J. S., Lee, C. G., Lee, W. S., Lee, K. S., Bang, S. M., et al. (2007). Assessment of clinical relevant fatigue level in cancer. Support Care Cancer, 15(7), 891–896.
Chaudhuri, A., Condon, B. R., Gow, J. W., Brennan, D., & Hadley, D. M. (2003). Proton magnetic resonance spectroscopy of basal ganglia in chronic fatigue syndrome. Neuroreport, 14(2), 225–228.
Collado-Hidalgo, A., Bower, J. E., Ganz, P. A., Cole, S. W., & Irwin, M. R. (2006). Inflammatory biomarkers for persistent fatigue in breast cancer survivors. Clinical Cancer Research, 12(9), 2759–2766.
Craig, A. D. (2010). The sentient self. Brain Structure and Function, 214(5–6), 563–577.
Craig, A. D. (2011). Significance of the insula for the evolution of human awareness of feelings from the body. Annals of the New York Academy of Sciences, 1225, 72–82.
Dantzer, R. (2001). Cytokine-induced sickness behavior: mechanisms and implications. Annals of the New York Academy of Sciences, 933, 222–234.
Dantzer, R., & Kelley, K. W. (2007). Twenty years of research on cytokine-induced sickness behavior. Brain, Behavior, and Immunity, 21(2), 153–160.
Filosa, A., Paixao, S., Honsek, S. D., Carmona, M. A., Becker, L., Feddersen, B., et al. (2009). Neuron-glia communication via EphA4/ephrin-A3 modulates LTP through glial glutamate transport. Nature Neuroscience, 12(10), 1285–1292.
Fletcher, M. A., Zeng, X. R., Barnes, Z., Levis, S., & Klimas, N. G. (2009). Plasma cytokines in women with chronic fatigue syndrome. Journal of Translational Medicine, 7, 96.
Friedman, S. D., Shaw, D. W., Ishak, G., Gropman, A. L., & Saneto, R. P. (2010). The use of neuroimaging in the diagnosis of mitochondrial disease. Developmental Disabilities Research Reviews, 16(2), 129–135.
Gelinas, C., & Fillion, L. (2004). Factors related to persistent fatigue following completion of breast cancer treatment. Oncology Nursing Forum, 31(2), 269–278.
Harris, R. E., Sundgren, P. C., Craig, A. D., Kirshenbaum, E., Sen, A., Napadow, V., et al. (2009). Elevated insular glutamate in fibromyalgia is associated with experimental pain. Arthritis and Rheumatism, 60(10), 3146–3152.
Hart, B. L. (1988). Biological basis of the behavior of sick animals. Neuroscience and Biobehavioral Reviews, 12(2), 123–137.
Hawkins, R. A. (2009). The blood–brain barrier and glutamate. American Journal of Clinical Nutrition, 90(3), 867S–874S.
Hedberg, T. G., & Stanton, P. K. (1996). Long-term plasticity in cingulate cortex requires both NMDA and metabotropic glutamate receptor activation. European Journal of Pharmacology, 310(1), 19–27.
Kesler, S. R., Watson, C., Koovakkattu, D., Lee, C., O’Hara, R., Mahaffey, M. L., et al. (2013). Elevated prefrontal myo-inositol and choline following breast cancer chemotherapy. Brain Imaging and Behavior. doi:10.1007/s11682-013-9228-1.
Kim, S. H., Son, B. H., Hwang, S. Y., Han, W., Yang, J. H., Lee, S., et al. (2008). Fatigue and depression in disease-free breast cancer survivors: prevalence, correlates, and association with quality of life. Journal of Pain and Symptom Management, 35(6), 644–655.
Lee, B. N., Dantzer, R., Langley, K. E., Bennett, G. J., Dougherty, P. M., Dunn, A. J., et al. (2004). A cytokine-based neuroimmunologic mechanism of cancer-related symptoms. Neuroimmunomodulation, 11(5), 279–292.
Liu, L., Mills, P. J., Rissling, M., Fiorentino, L., Natarajan, L., Dimsdale, J. E., et al. (2012). Fatigue and sleep quality are associated with changes in inflammatory markers in breast cancer patients undergoing chemotherapy. Brain, Behavior, and Immunity, 26(5), 706–713.
Lopez Zunini, R. A., Scherling, C., Wallis, N., Collins, B., Mackenzie, J., Bielajew, C., et al. (2012). Differences in verbal memory retrieval in breast cancer chemotherapy patients compared to healthy controls: a prospective fMRI study. Brain Imaging and Behavior. doi:10.1007/s11682-012-9213-0.
Maddock, R. J., & Buonocore, M. H. (2012). MR spectroscopic studies of the brain in psychiatric disorders. Current Topics in Behavioral Neurosciences. doi:10.1007/7854_2011_197.
Maier, S. F. (2003). Bi-directional immune-brain communication: implications for understanding stress, pain, and cognition. Brain, Behavior, and Immunity, 17(2), 69–85.
Mathew, S. J., Mao, X., Keegan, K. A., Levine, S. M., Smith, E. L., Heier, L. A., et al. (2009). Ventricular cerebrospinal fluid lactate is increased in chronic fatigue syndrome compared with generalized anxiety disorder: an in vivo 3.0 T (1)H MRS imaging study. NMR in Biomedicine, 22(3), 251–258.
Matsui, K., Jahr, C. E., & Rubio, M. E. (2005). High-concentration rapid transients of glutamate mediate neural-glial communication via ectopic release. Journal of Neuroscience, 25(33), 7538–7547.
Mehta, A., Prabhakar, M., Kumar, P., Deshmukh, R., & Sharma, P. L. (2013). Excitotoxicity: bridge to various triggers in neurodegenerative disorders. European Journal of Pharmacology, 698(1–3), 6–18.
Mendoza, T. R., Wang, X. S., Cleeland, C. S., Morrissey, M., Johnson, B. A., Wendt, J. K., et al. (1999). The rapid assessment of fatigue severity in cancer patients: Use of the Brief Fatigue Inventory. Cancer, 85(5), 1186–1196.
Moffett, J. R., Ross, B., Arun, P., Madhavarao, C. N., & Namboodiri, A. M. (2007). N-Acetylaspartate in the CNS: from neurodiagnostics to neurobiology. Progress in Neurobiology, 81(2), 89–131.
Montazeri, A. (2008). Health-related quality of life in breast cancer patients: a bibliographic review of the literature from 1974 to 2007. Journal of Experimental & Clinical Cancer Research, 27, 32.
Myers, J. S. (2008). Proinflammatory cytokines and sickness behavior: implications for depression and cancer-related symptoms. Oncology Nursing Forum, 35(5), 802–807.
Nakanishi, S. (1992). Molecular diversity of glutamate receptors and implications for brain function. Science, 258(5082), 597–603.
Provencher, S. W. (1993). Estimation of metabolite concentrations from localized in vivo proton NMR spectra. Magnetic Resonance in Medicine, 30(6), 672–679.
Puri, B. K., Counsell, S. J., Zaman, R., Main, J., Collins, A. G., Hajnal, J. V., et al. (2002). Relative increase in choline in the occipital cortex in chronic fatigue syndrome. Acta Psychiatrica Scandinavica, 106(3), 224–226.
Rawson, E. S., & Venezia, A. C. (2011). Use of creatine in the elderly and evidence for effects on cognitive function in young and old. Amino Acids, 40(5), 1349–1362.
Reid-Arndt, S. A., Hsieh, C., & Perry, M. C. (2010). Neuropsychological functioning and quality of life during the first year after completing chemotherapy for breast cancer. Psychooncology, 19(5), 535–544.
Reuter-Lorenz, P. A., & Cimprich, B. (2013). Cognitive function and breast cancer: promise and potential insights from functional brain imaging. Breast Cancer Research and Treatment, 137(1), 33–43.
Reyngoudt, H., Achten, E., & Paemeleire, K. (2012). Magnetic resonance spectroscopy in migraine: what have we learned so far? Cephalalgia. doi:10.1177/0333102412452048.
Ronnback, L., & Hansson, E. (2004). On the potential role of glutamate transport in mental fatigue. Journal of Neuroinflammation, 1(1), 22.
Savic, I., Thomas, A. M., Ke, Y., Curran, J., Fried, I., & Engel, J., Jr. (2000). In vivo measurements of glutamine + glutamate (Glx) and N-acetyl aspartate (NAA) levels in human partial epilepsy. Acta Neurologica Scandinavica, 102(3), 179–188.
Schifitto, G., Deng, L., Yeh, T. M., Evans, S. R., Ernst, T., Zhong, J., et al. (2011). Clinical, laboratory, and neuroimaging characteristics of fatigue in HIV-infected individuals. Journal for Neurovirology, 17(1), 17–25.
Schubert, C., Hong, S., Natarajan, L., Mills, P. J., & Dimsdale, J. E. (2007). The association between fatigue and inflammatory marker levels in cancer patients: a quantitative review. Brain, Behavior, and Immunity, 21(4), 413–427.
Segerstrom, S. C., & Miller, G. E. (2004). Psychological stress and the human immune system: a meta-analytic study of 30 years of inquiry. Psychological Bulletin, 130(4), 601–630.
Starkweather, A. R., Lyon, D. E., & Schubert, C. M. (2011). Pain and Inflammation in women with early-stage breast cancer prior to induction of chemotherapy. Biological Research for Nursing. doi:10.1177/1099800411425857.
Tittle, M. B., McMillan, S. C., & Hagan, S. (2003). Validating the brief pain inventory for use with surgical patients with cancer. Oncology Nursing Forum, 30(2), 325–330.
Wallimann, T., Wyss, M., Brdiczka, D., Nicolay, K., & Eppenberger, H. M. (1992). Intracellular compartmentation, structure and function of creatine kinase isoenzymes in tissues with high and fluctuating energy demands: the ‘phosphocreatine circuit’ for cellular energy homeostasis. Biochemistry Journal, 281(Pt 1), 21–40.
Acknowledgments
This research was supported by grants from the Ronald P. and Joan M. Nordgren Cancer Research Fund and the University of Michigan National Institutes of Health Clinical and Translational Awards (CTSA) grant number UL1RR024986.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standards
The experiments described in this manuscript comply with the current laws of the country in which they were performed.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zick, S.M., Zwickey, H., Wood, L. et al. Preliminary differences in peripheral immune markers and brain metabolites between fatigued and non-fatigued breast cancer survivors: a pilot study. Brain Imaging and Behavior 8, 506–516 (2014). https://doi.org/10.1007/s11682-013-9270-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11682-013-9270-z