Abstract
Background
Bariatric surgery leads to remission of several obesity-related comorbidities, including hypertension. Although antihypertensive medication use is decreased after bariatric surgery, the exact time course of decrease in blood pressure after surgery is not known.
Methods
A database of patients undergoing bariatric surgery at our institute was used to study the effect of surgery on time course of blood pressure changes. Data from surgeries performed between January 2010 and December 2012 were used.
Results
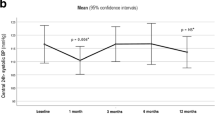
Maximum blood pressure and body weight decreases were observed at 2 weeks and 1 year after surgery, respectively. Average decrease in the mean arterial pressure (MAP) was 4.46 mmHg (61.5 ± 17.1% of maximal decrease) and 7.17 mmHg (maximum decrease) at 1 and 2 weeks after surgery, when the decrease in body weight is 22.8 ± 1.6 and 28 ± 1.4% of maximal weight loss, respectively. In hypertensive patients, MAP decreased from 98.5 ± 0.78 to 92.3 ± 1.76 and 93.1 ± 0.92 mmHg at 1 and 2 weeks post-surgery, respectively. In normotensive patients, the MAP decreased from 96.2 ± 0.79 to 88.7 ± 1.25, 90.0 ± 0.94, 86.5 ± 1.35, 88.0 ± 1.13, and 86.4 ± 2.13 mmHg at 2 weeks, 3 and 6 months, and 1 and 3 years after surgery, respectively.
Conclusions
These data demonstrate that significant decrease in MAP occurs within 2 weeks after bariatric surgery in hypertensive as well as normotensive patients. Future studies are required to investigate the weight-independent mechanisms of blood pressure decreases after bariatric surgery.



Similar content being viewed by others
References
Eldar S, Heneghan HM, Brethauer SA, et al. Bariatric surgery for treatment of obesity. Int J Obes. 2011;35(Suppl 3):S16–21. https://doi.org/10.1038/ijo.2011.142.
Puzziferri N, Roshek 3rd TB, Mayo HG, et al. Long-term follow-up after bariatric surgery: a systematic review. JAMA. 2014;312(9):934–42. https://doi.org/10.1001/jama.2014.10706.
Payne JH, DeWind LT. Surgical treatment of obesity. Am J Surg. 1969;118(2):141–7. https://doi.org/10.1016/0002-9610(69)90113-5.
Benaiges D, Mas-Lorenzo A, Goday A, et al. Laparoscopic sleeve gastrectomy: more than a restrictive bariatric surgery procedure? World J Gastroenterol. 2015;21(41):11804–14. https://doi.org/10.3748/wjg.v21.i41.11804.
Courcoulas AP, Yanovski SZ, Bonds D, et al. Long-term outcomes of bariatric surgery: a National Institutes of Health symposium. JAMA Surg. 2014;149(12):1323–9. https://doi.org/10.1001/jamasurg.2014.2440.
Muller-Stich BP, Senft JD, Warschkow R, et al. Surgical versus medical treatment of type 2 diabetes mellitus in nonseverely obese patients: a systematic review and meta-analysis. Ann Surg. 2015;261(3):421–9. https://doi.org/10.1097/SLA.0000000000001014.
Breen DM, Rasmussen BA, Kokorovic A, et al. Jejunal nutrient sensing is required for duodenal-jejunal bypass surgery to rapidly lower glucose concentrations in uncontrolled diabetes. Nat Med. 2012;18(6):950–5. https://doi.org/10.1038/nm.2745.
Saeidi N, Meoli L, Nestoridi E, et al. Reprogramming of intestinal glucose metabolism and glycemic control in rats after gastric bypass. Science. 2013;341(6144):406–10. https://doi.org/10.1126/science.1235103.
Munzberg H, Laque A, Yu S, et al. Appetite and body weight regulation after bariatric surgery. Obes Rev. 2015;16(Suppl 1):77–90. https://doi.org/10.1111/obr.12258.
Mingrone G. Role of the incretin system in the remission of type 2 diabetes following bariatric surgery. Nutr Metab Cardiovasc Dis. 2008;18(8):574–9. https://doi.org/10.1016/j.numecd.2008.07.004.
Hatoum IJ, Blackstone R, Hunter TD, et al. Clinical factors associated with remission of obesity-related comorbidities after bariatric surgery. JAMA Surg. 2016;151(2):130–7. https://doi.org/10.1001/jamasurg.2015.3231.
Neff KJ, Baud G, Raverdy V, et al. Renal function and remission of hypertension after bariatric surgery: a 5-year prospective cohort study. Obes Surg. 2017;27(3):613–9. https://doi.org/10.1007/s11695-016-2333-7.
Ahmed AR, Rickards G, Coniglio D, et al. Laparoscopic Roux-en-Y gastric bypass and its early effect on blood pressure. Obes Surg. 2009;19(7):845–9. https://doi.org/10.1007/s11695-008-9671-z.
Bland CM, Tritsch AM, Bookstaver DA, et al. Hypertension and diabetes mellitus medication management in sleeve gastrectomy patients. Am J Health Syst Pharm. 2013;70(12):1018–20. https://doi.org/10.2146/ajhp120607.
Bonfils PK, Taskiran M, Damgaard M, et al. Roux-en-Y gastric bypass alleviates hypertension and is associated with an increase in mid-regional pro-atrial natriuretic peptide in morbid obese patients. J Hypertens. 2015;33(6):1215–25. https://doi.org/10.1097/HJH.0000000000000526.
Hamdy O, Porramatikul S, Al-Ozairi E. Metabolic obesity: the paradox between visceral and subcutaneous fat. Curr Diabetes Rev. 2006;2(4):367–73.
Matsuzawa Y, Funahashi T, Nakamura T. The concept of metabolic syndrome: contribution of visceral fat accumulation and its molecular mechanism. J Atheroscler Thromb. 2011;18(8):629–39. https://doi.org/10.5551/jat.7922.
Vaneckova I, Maletinska L, Behuliak M, et al. Obesity-related hypertension: possible pathophysiological mechanisms. J Endocrinol. 2014;223(3):R63–78. https://doi.org/10.1530/JOE-14-0368.
Prior LJ, Davern PJ, Burke SL, et al. Exposure to a high-fat diet during development alters leptin and ghrelin sensitivity and elevates renal sympathetic nerve activity and arterial pressure in rabbits. Hypertension. 2014;63(2):338–45. https://doi.org/10.1161/HYPERTENSIONAHA.113.02498.
Wofford MR, Anderson Jr DC, Brown CA, et al. Antihypertensive effect of alpha- and beta-adrenergic blockade in obese and lean hypertensive subjects. Am J Hypertens. 2001;14(7 Pt 1):694–8. https://doi.org/10.1016/S0895-7061(01)01293-6.
Hall JE, do Carmo JM, da Silva AA, et al. Obesity-induced hypertension: interaction of neurohumoral and renal mechanisms. Circ Res. 2015;116(6):991–1006. https://doi.org/10.1161/CIRCRESAHA.116.305697.
Seven E, Husemoen LL, Wachtell K, et al. Overweight, adipocytokines and hypertension: a prospective population-based study. J Hypertens. 2014;32(7):1488–1494; discussion 94. https://doi.org/10.1097/HJH.0000000000000207.
Jorgensen NB, Dirksen C, Bojsen-Moller KN, et al. Exaggerated glucagon-like peptide 1 response is important for improved beta-cell function and glucose tolerance after Roux-en-Y gastric bypass in patients with type 2 diabetes. Diabetes. 2013;62(9):3044–52. https://doi.org/10.2337/db13-0022.
Ryan D, Acosta A. GLP-1 receptor agonists: nonglycemic clinical effects in weight loss and beyond. Obesity (Silver Spring). 2015;23(6):1119–29. https://doi.org/10.1002/oby.21107.
Blonde L, Klein EJ, Han J, et al. Interim analysis of the effects of exenatide treatment on A1C, weight and cardiovascular risk factors over 82 weeks in 314 overweight patients with type 2 diabetes. Diabetes Obes Metab. 2006;8(4):436–47. https://doi.org/10.1111/j.1463-1326.2006.00602.x.
Buse JB, Drucker DJ, Taylor KL, et al. DURATION-1: exenatide once weekly produces sustained glycemic control and weight loss over 52 weeks. Diabetes Care. 2010;33(6):1255–61. https://doi.org/10.2337/dc09-1914.
Aghamohammadzadeh R, Greenstein AS, Yadav R, et al. Effects of bariatric surgery on human small artery function: evidence for reduction in perivascular adipocyte inflammation, and the restoration of normal anticontractile activity despite persistent obesity. J Am Coll Cardiol. 2013;62(2):128–35. https://doi.org/10.1016/j.jacc.2013.04.027.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethical Statement
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent Statement
For this type of study, formal consent is not required.
Electronic Supplementary Material
ESM 1
(DOCX 128 KB)
Rights and permissions
About this article
Cite this article
Hawkins, D.N., Faler, B.J., Choi, Y.U. et al. Time Course of Blood Pressure Decrease After Bariatric Surgery in Normotensive and Hypertensive Patients. OBES SURG 28, 1845–1851 (2018). https://doi.org/10.1007/s11695-017-3091-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11695-017-3091-x