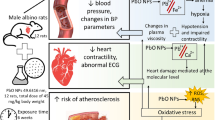
Abstract
Lead exposure has been considered to be a risk factor for hypertension and cardiovascular disease. Our purpose was to evaluate the effects of low plasma lead concentration on cardiac contractility in isolated papillary muscles. Wistar rats were divided in control group or group treated with 100 ppm of lead acetate in the drinking water for 15 days. Blood pressure (BP) was measured weekly. At the end of the treatment period, the animals were anesthetized and euthanized, and parameters related to isolated papillary muscle contractility were recorded. The lead concentrations in the blood reached 12.3 ± 2 μg/dL. The BP was increased in the group treated with 100 ppm of lead acetate. Lead treatment did not alter force and time derivatives of the force of left ventricular papillary muscles. In addition, the inotropic response induced by an increase in the extracellular Ca2+ concentration was reduced in the Pb2+ group. However, the uptake of Ca2+ by the sarcoplasmic reticulum and the protein expression of SERCA and phospholamban remained unchanged. Postrest contraction was similar in the both groups, and tetanic peak and plateau tension were reduced in lead group. These results demonstrated that the reduction in the inotropic response to calcium does not appear to be caused by changes in the trans-sarcolemmal calcium flux but suggest that an impairment of the contractile machinery might be taking place. Our results demonstrate that even at a concentration below the limit considered to be safe, lead exerts deleterious effects on the cardiac contractile machinery.






Similar content being viewed by others
References
Glenn BS, Stewart WF, Links JM et al (2003) The longitudinal association of lead with blood pressure. Epidemiology 14:30–36
Kosnett MJ (2003) Intoxicação por Metais Pesados & Quelantes. In:Katzung, Bertram G (Guanabara koogan) Farmacol Básica Clínica 8Ed Rio de Janeiro, 867–870
Navas-Acien A, Guallar E, Silbergeld EK, Rothenberg SJ (2007) Lead exposure and cardiovascular disease–a systematic review. Environ Health Perspect 115:472–482
Vaziri ND, Ding Y, Ni Z, Gonick HC (1997) Altered nitric oxide metabolism and increased oxygen free radical activity in lead-induced hypertension: effect of lazaroid therapy. Kidney Int 52:1042–1046
Vaziri ND, Ding Y, Ni Z (1999) Nitric oxide synthase expression in the course of lead-induced hypertension. Hypertension 34:558–562. doi:10.1161/01.HYP.34.4.558
Vaziri ND, Lin C-Y, Farmand F, Sindhu RK (2003) Superoxide dismutase, catalase, glutathione peroxidase and NADPH oxidase in lead-induced hypertension. Kidney Int 63:186–194. doi:10.1046/j.1523-1755.2003.00711.x
Farmand F, Ehdaie A, Roberts CK, Sindhu RK (2005) Lead-induced dysregulation of superoxide dismutases, catalase, glutathione peroxidase, and guanylate cyclase. Environ Res 98:33–39
Heydari A, Norouzzadeh A, Khoshbaten A et al (2006) Effects of short-term and subchronic lead poisoning on nitric oxide metabolites and vascular responsiveness in rat. Toxicol Lett 166:88–94
Fiorim J, Ribeiro RF, Silveira EA et al (2011) Low-level lead exposure increases systolic arterial pressure and endothelium-derived vasodilator factors in rat aortas. PLoS One. doi:10.1371/journal.pone.0017117
Silveira EA, Siman FDM, de Oliveira FT et al (2014) Low-dose chronic lead exposure increases systolic arterial pressure and vascular reactivity of rat aortas. Free Radic Biol Med 67:366–376
Fioresi M, Simões MR, Furieri LB et al (2014) Chronic lead exposure increases blood pressure and myocardial contractility in rats. PLoS One 9:e96900
Lustberg M, Silbergeld E (2002) Blood lead levels and mortality. Arch Intern Med 162:2443–2449. doi:10.1001/archinte.162.21.2443
Agency for Toxic Substances and Disease Registry (ATSDR) (2007) Toxocological profile for lead (Update). Department of Public Health and Human Services, Public Health Service. Atlanta, GA: US
Carmignani M, Boscolo P, Poma A, Volpe AR (1999) Kininergic system and arterial hypertension following chronic exposure to inorganic lead. Immunopharmacology 44:105–110
Carmignani M, Volpe AR, Boscolo P et al (2000) Catcholamine and nitric oxide systems as targets of chronic lead exposure in inducing selective functional impairment. Life Sci 68:401–415
Nhanes, III [National Health and Nutrition Examination Survey], Hond ED, Nawrot TSJ (2002) The relationship between blood pressure and blood lead in NHANES III. J Hum Hypertens 16:563–568
Vassallo DV, Lebarch EC, Moreira CM et al (2008) Lead reduces tension development and the myosin ATPase activity of the rat right ventricular myocardium. Braz J Med Biol Res 41:789–795
Fioresi M, Furieri LB, Simões MR et al (2013) Acute exposure to lead increases myocardial contractility independent of hypertension development. Braz J Med Biol Res 46:178–185
Korecková-Sysalová J (1997) Determination of cadmium and lead levels in human blood of a general Czech population by GFAAS. Biol Trace Elem Res 56:321–329
Grizzo LT, Cordellini S (2008) Perinatal lead exposure affects nitric oxide and cyclooxygenase pathways in aorta of weaned rats. Toxicol Sci 103:207–214. doi:10.1093/toxsci/kfn018
Leite CM, Vassallo DV, Mill JG (1995) Characteristics of tetanic contractions in caffeine-treated rat myocardium. Can J Physiol Pharmacol 73:638–643
BRASIL (2006) Atenção à saúde dos trabalhadores expostos ao chumbo metálico. Ministério da http://bvsms.saude.gov.br/bvs/publicacoes/protocolo_atencao_saude_trab_exp_chumbo_met.pdf Accessed 10 December 2014
(WHO) WHO (2011) Prüss-Ustün, A; Vickers, C; Haefliger, P. Bertollini, R. Knowns and unknowns on burden of disease due to chemicals: a systematic review. Department of Public Health and Environment, World Health Organization. Geneva, Switzerland. Environ Heal 10:2–15
Glenn BS, Bandeen-Roche K, Lee B-K et al (2006) Changes in systolic blood pressure associated with lead in blood and bone. Epidemiology 17:538–544
Simões MR, Ribeiro Júnior RF, Vescovi MV a et al (2011) Acute lead exposure increases arterial pressure: role of the renin-angiotensin system. PLoS One 6:e18730. doi:10.1371/journal.pone.0018730
Khalil-Manesh F, Gonick HC, Weiler EW et al (1993) Lead-induced hypertension: possible role of endothelial factors. Am J Hypertens 6:723–729
Vaziri ND, Ding Y (2001) Effect of lead on nitric oxide synthase expression in coronary endothelial cells: role of superoxide. Hypertension 37:223–226
Tsao DA, Yu HS, Cheng JT et al (2000) The change of beta-adrenergic system in lead-induced hypertension. Toxicol Appl Pharmacol 164:127–133
Boscolo P, Carmignani M (1988) Neurohumoral blood pressure regulation in lead exposure. Environ Health Perspect 78:101–106
Prentice RC, Kopp SJ (1985) Cardiotoxicity of lead at various perfusate calcium concentrations: functional and metabolic responses of the perfused rat heart. Toxicol Appl Pharmacol 81:491–501
Bernal J, Lee JH, Cribbs LL, Perez-Reyes E (1997) Full reversal of Pb++ block of L-type Ca++ channels requires treatment with heavy metal antidotes. J Pharmacol Exp Ther 282:172–180
Dabrowska-Bouta B, Struzyńska L, Rafałowska U (1996) Effect of acute and chronic lead exposure on the level of sulfhydryl groups in rat brain. Acta Neurobiol Exp (Wars) 56:233–236
Acknowledgments
This project was supported by CAPES (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) and CNPq/FAPES (Conselho Nacional de Desenvolvimento Científico e Tecnológico and Fundação de Amparo à Pesquisa do Espírito Santo) (54668662/2011). The funders had no role in the study’s design, data collection, or analysis nor were they involved in the manuscript’s preparation or the decision to publish it.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Silva, M.A.S.C., de Oliveira, T.F., Almenara, C.C.P. et al. Exposure to a Low Lead Concentration Impairs Contractile Machinery in Rat Cardiac Muscle. Biol Trace Elem Res 167, 280–287 (2015). https://doi.org/10.1007/s12011-015-0300-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12011-015-0300-0