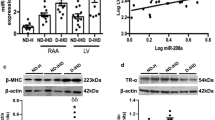
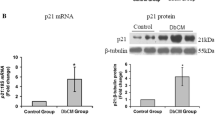
Abstract
Oxidative stress is involved in the etiology of diabetes-induced cardiac dysfunction while microRNAs (miRNAs) are known as regulators for genes involved in cardiac remodeling. However, a functional link between miRNAs and diabetes-induced cardiac dysfunction remains to be investigated. Here, we aimed to identify whether the expression levels of miRNAs are associated with oxidative stress/diabetic heart and if proteins responsible from contractile activity during diabetes might be directly modulated by miRNAs. Diabetic cardiomyopathy developed with streptozotocin, is characterized with marked changes in sarcomere and mitochondria, depressed left ventricular developed pressure, and a massive oxidative stress that is particularly evident in the heart. miRNA profiling was performed in freshly isolated left ventricular cells from diabetic rats. Using microarray analysis, we identified marked changes in the expression of 43 miRNAs (37 of them were downregulated while 6 miRNAs were upregulated) out of examined total of 351 miRNAs. Among them, 6 miRNAs were further validated by real-time PCR. The expression levels of miR-1, miR-499, miR-133a, and miR-133b were markedly depressed in the diabetic cardiomyocytes while miR-21 level increased and miR-16 level was unchanged. Notably, normalization of cardiac function and oxidant/antioxidant level after N-acetylcysteine (NAC)-treatment of diabetic rats resulted with a significant restoration in the expression levels of miR-499, miR-1, miR-133a, and miR-133b in the myocardium. Since changes in the level of muscle-specific miR-1 has been implicated in cardiac diseases and its specific molecular targets involved in its action, in part, associated with oxidative stress are limited, we first examined the protein levels of some SR-associated proteins such as junctin and triadin. Junctin but not triadin is markedly overexpressed in diabetic cardiomyocytes while its level was normalized in NAC-treated diabetics. Luciferase reporter assay showed that junctin is targetted by miR-1. Taken together, our data demonstrates that intervention with an antioxidant treatment for 4-week leads to significant cardioprotection against diabetes-induced injury, controlling oxidant/antioxidant level, which may directly control the levels of some miRNAs including miR-1 and its target protein junctin, which is involved in the development of diabetic cardiomyopathy.





Similar content being viewed by others
References
Rubler, S., Dlugash, J., Yuceoglu, Y. Z., Kumral, T., Branwood, A. W., & Grishman, A. (1972). New type of cardiomyopathy associated with diabetic glomerulosclerosis. American Journal of Cardiology, 30, 595–602.
Fein, F. S., & Sonnenblick, E. H. (1994). Diabetic cardiomyopathy. Cardiovascular Drugs and Therapy, 8, 65–73.
Pierce, G. N., & Russel, J. C. (1997). Regulation of intracellular Ca2+ in the heart during diabetes. Cardiovascular Research, 34, 41–47.
Ayaz, M., Ozdemir, S., Ugur, M., Vassort, G., & Turan, B. (2004). Effects of selenium on altered mechanical and electrical cardiac activities of diabetic rat. Archives of Biochemistry and Biophysics, 426(1), 83–90.
Rahangdale, S., Yeh, S. Y., Malhotra, A., & Veves, A. (2009). Therapeutic interventions and oxidative stress in diabetes. Frontiers in Bioscience, 14, 192–209.
Vassort, G., & Turan, B. (2010). Protective role of antioxidants in diabetes-induced cardiac dysfunction. Cardiovascular Toxicology, 10(2), 73–86.
Turan, B. (2010). Role of antioxidants in redox regulation of diabetic cardiovascular complications. Current Pharmaceutical Biotechnology, 11, 819–836.
Watanabe, K., Thandavarayan, R. A., Harima, M., Sari, F. R., Gurusamy, N., Veeraveedu, P. T., et al. (2010). Role of differential signaling pathways and oxidative stress in diabetic cardiomyopathy. Current Cardiology Reviews, 6(4), 280–290.
Shen, E., Diao, X., Wang, X., Chen, R., & Hu, B. (2011). MicroRNAs involved in the mitogen-activated protein kinase cascades pathway during glucose-induced cardiomyocyte hypertrophy. American Journal of Pathology, 179, 639–650.
Diao, X., Shen, E., Wang, X., & Hu, B. (2011). Differentially expressed microRNAs and their target genes in the hearts of streptozotocin-induced diabetic mice. Molecular Medicine Reports, 4, 633–640.
Feng, B., Chen, S., George, B., Feng, Q., & Chakrabarti, S. (2010). miR133a regulates cardiomyocyte hypertrophy in diabetes. Diabetes Metabolism Research and Reviews, 26, 40–49.
Caporali, A., Meloni, M., Völlenkle, C., Bonci, D., Sala-Newby, G. B., Addis, R., et al. (2011). Deregulation of microRNA-503 contributes to diabetes mellitus-induced impairment of endothelial function and reparative angiogenesis after limb ischemia. Circulation, 123, 282–291.
Wang, X. H., Qian, R. Z., Zhang, W., Chen, S. F., Jin, H. M., & Hu, R. M. (2009). MicroRNA-320 expression in myocardial microvascular endothelial cells and its relationship with insulin-like growth factor-1 in type 2 diabetic rats. Clinical and Experimental Pharmacology and Physiology, 36, 181–188.
Mishra, P. K., Tyagi, N., Kumar, M., & Tyagi, S. C. (2009). MicroRNAs as a therapeutic target for cardiovascular diseases. Journal of Cellular and Molecular Medicine, 13(4), 778–789.
Catalucci, D., Gallo, P., & Condorelli, G. (2009). MicroRNAs in cardiovascular biology and heart disease. Circulation Cardiovascular Genetics, 2, 402–408.
Zampetaki, A., Kiechl, S., Drozdov, I., Willeit, P., Mayr, U., Prokopi, M., et al. (2010). Plasma microRNA profiling reveals loss of endothelial miR-126 and other microRNAs in type 2 diabetes. Circulation Research, 107, 810–817.
Kolfschoten, I. G., Roggli, E., Nesca, V., & Regazzi, R. (2009). Role and therapeutic potential of microRNAs in diabetes. Diabetes, Obesity and Metabolism, 4, 118–129.
Shantikumar, S., Caporali, A., & Emanueli, C. (2012). Role of microRNAs in diabetes and its cardiovascular complications. Cardiovascular Research, 93, 583–593.
Sebastiani, G., Vendrame, F., & Dotta, F. (2011). MicroRNAs as new tools for exploring type 1 diabetes: relevance for immunomodulation and transplantation therapy. Transplantation Proceedings, 43, 330–332.
Kuwabara, Y., Ono, K., Horie, T., Nishi, H., Nagao, K., Kinoshita, M., et al. (2011). Increased microRNA-1 and microRNA-133a levels in serum of patients with cardiovascular disease indicate myocardial damage. Circulation Cardiovascular Genetics, 4, 446–454.
Goren, Y., Kushnir, M., Zafrir, B., Tabak, S., Lewis, B. S., & Amir, O. (2012). Serum levels of microRNAs in patients with heart failure. European Journal of Heart Failure, 14, 147–154.
Elton, T. S., Martin, M. M., Sansom, S. E., Belevych, A. E., Györke, S., & Terentyev, D. (2011). miRNAs got rhythm. Life Sciences, 88, 373–383.
Sakakibara, M., Hirashiki, A., Cheng, X. W., Bando, Y., Ohshima, K., Okumura, T., et al. (2011). Association of diabetes mellitus with myocardial collagen accumulation and relaxation impairment in patients with dilated cardiomyopathy. Diabetes Research and Clinical Practice, 92, 348–355.
Martín-Gallán, P., Carrascosa, A., Gussinyé, M., & Domínguez, C. (2003). Biomarkers of diabetes-associated oxidative stress and antioxidant status in young diabetic patients with or without subclinical complications. Free Radical Biology and Medicine, 34, 1563–1574.
Aydemir-Koksoy, A., Bilginoglu, A., Sariahmetoglu, M., Schulz, R., & Turan, B. (2010). Antioxidant treatment protects diabetic rats from cardiac dysfunction by preserving contractile protein targets of oxidative stress. Journal of Nutritional Biochemistry, 21, 827–833.
Yaras, N., Ugur, M., Ozdemir, S., Gurdal, H., Purali, N., Lacampagne, A., et al. (2005). Effects of diabetes on ryanodine receptor Ca release channel (RyR2) and Ca2+ homeostasis in rat heart. Diabetes, 54, 3082–3088.
Yaras, N., Sariahmetoglu, M., Bilginoglu, A., Aydemir-Koksoy, A., Onay-Besikci, A., Turan, B., et al. (2008). Protective action of doxycycline against diabetic cardiomyopathy in rats. British Journal of Pharmacology, 155, 1174–1184.
Fiordaliso, F., Bianchi, R., Staszewsky, L., Cuccovillo, I., Doni, M., Laragione, T., et al. (2004). Antioxidant treatment attenuates hyperglycemia-induced cardiomyocyte death in rats. Journal of Molecular and Cellular Cardiology, 37, 959–968.
Treweeke, A. T., Winterburn, T. J., Mackenzie, I., Barrett, F., Barr, C., Rushworth, G. F., et al. (2012). N-Acetylcysteine inhibits platelet-monocyte conjugation in patients with type 2 diabetes with depleted intraplatelet glutathione: a randomised controlled trial. Diabetologia, 55, 2920–2928.
Calzadilla, P., Sapochnik, D., Cosentino, S., Diz, V., Dicelio, L., Calvo, J. C., et al. (2011). N-Acetylcysteine reduces markers of differentiation in 3T3-L1 adipocytes. International Journal of Molecular Sciences, 12, 6936–6951.
Hong, C. S., Kwon, S. J., Cho, M. C., Kwak, Y. G., Ha, K. C., Hong, B., et al. (2008). Overexpression of junctate induces cardiac hypertrophy and arrhythmia via altered calcium handling. Journal of Molecular and Cellular Cardiology, 44, 672–682.
Fan, G.-C., Yuan, Q., Zhao, W., & Chu, G. (2007). Junctin is a prominent regulator of contractility in cardiomyocytes. Biochemical and Biophysical Research Communications, 352, 617–622.
Evangelia, G., KraniasKirchhefer, U., Hanske, G., Jones, L. R., Justus, I., Kaestner, L., et al. (2006). Overexpression of junctin causes adaptive changes in cardiac myocyte Ca(2+) signaling. Cell Calcium, 39, 131–142.
Bartel, D. P. (2004). MicroRNAs: genomics, biogenesis, mechanism, and function. Cell, 116, 281–297.
Cheng, A. M., Byrom, M. W., Shelton, J., & Ford, L. P. (2005). Antisense inhibition of human miRNAs and indications for an involvement of miRNA in cell growth and apoptosis. Nucleic Acids Research, 33, 1290–1297.
Lu, H., Buchan, R. J., & Cook, S. A. (2010). MicroRNA-223 regulates Glut4 expression and cardiomyocyte glucose metabolism. Cardiovascular Research, 86, 410–420.
Thum, T., Gross, C., Fiedler, J., Fischer, T., Kissler, S., Bussen, M., et al. (2008). MicroRNA-21 contributes to myocardial disease by stimulating MAP kinase signalling in fibroblasts. Nature, 456, 980–984.
Suckau, L., Fechner, H., Chemaly, E., Krohn, S., Hadri, L., Kockskämper, J., et al. (2009). Long-term cardiac-targeted RNA interference for the treatment of heart failure restores cardiac function and reduces pathological hypertrophy. Circulation, 119, 1241–1252.
Norlin, S., Ahlgren, U., & Edlund, H. (2005). Nuclear factor-{kappa}B activity in {beta}-cells is required for glucose-stimulated insulin secretion. Diabetes, 54, 125–132.
Fu, J. D., Rushing, S. N., Lieu, D. K., Chan, C. W., Kong, C. W., Geng, L., et al. (2011). Distinct roles of microRNA-1 and -499 in ventricular specification and functional maturation of human embryonic stem cell-derived cardiomyocytes. PLoS One, 6, e27417.
Shieh, J. T., Huang, Y., Gilmore, J., & Srivastava, D. (2011). Elevated miR-499 levels blunt the cardiac stress response. PLoS One, 6, e19481.
Yu, X. Y., Song, Y. H., Geng, Y. J., Lin, Q. X., Shan, Z. X., Lin, S. G., et al. (2008). Glucose induces apoptosis of cardiomyocytes via microRNA-1 and IGF-1. Biochemical and Biophysical Research Communications, 376, 548–552.
Li, Q., Song, X. W., Zou, J., Wang, G. K., Kremneva, E., Li, X. Q., et al. (2010). Attenuation of microRNA-1 derepresses the cytoskeleton regulatory protein twinfilin-1 to provoke cardiac hypertrophy. Journal of Cell Science, 123, 2444–2452.
Terentyev, D., Viatchenko-Karpinski, S., Vedamoorthyrao, S., Oduru, S., Györke, I., Williams, S. C., et al. (2007). Protein protein interactions between triadin and calsequestrin are involved in modulation of sarcoplasmic reticulum calcium release in cardiac myocytes. Journal of Physiology, 583, 71–80.
Hong, C. S., Cho, M. C., Kwak, Y. G., Song, C. H., Lee, Y. H., Lim, J. S., et al. (2002). Cardiac remodeling and atrial fibrillation in transgenic mice overexpressing junctin. FASEB Journal, 16, 1310–1312.
Tijskens, P., Jones, L. R., & Franzini-Armstrong, C. (2003). Junctin and calsequestrin overexpression in cardiac muscle: the role of junctin and the synthetic and delivery pathways for the two proteins. Journal of Molecular and Cellular Cardiology, 35, 961–974.
Acknowledgments
This work has been supported by grants from TUBITAK SBAG-109S267 and SBAG-111S042. We are also grateful to N. Tekin and F. A. Cicek for their technical contributions.
Conflict of interest
No potential conflicts of interest relevant to this article were reported.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yildirim, S.S., Akman, D., Catalucci, D. et al. Relationship Between Downregulation of miRNAs and Increase of Oxidative Stress in the Development of Diabetic Cardiac Dysfunction: Junctin as a Target Protein of miR-1. Cell Biochem Biophys 67, 1397–1408 (2013). https://doi.org/10.1007/s12013-013-9672-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-013-9672-y