Abstract
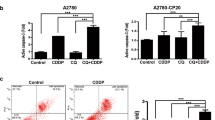
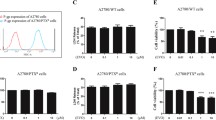
Cisplatin is a first-line chemotherapy drug against ovarian cancer. However, its strong toxic side effects and the development of cisplatin resistance in human cancer cells seriously influence the effects of chemotherapy and quality of life in patients. Noscapine (Nos), a non-toxic benzylisoquinoline alkaloid extracted from opium, has been recently reported to have anti-cancer activity, but the mechanism of that effect has not been clearly established. In the present study, we investigated cytotoxicity of Nos in combination with cisplatin (DDP) in drug-resistant human ovarian cancer cell line SKOV3/DDP in vitro and in vivo null mice xenograft model. Cell proliferation was measured by MTT assay, flow cytometry was used to analyze cell cycle and apoptosis, protein expression of several apoptotic factors was investigated by flow cytometry and immunohistochemical method, and their mRNA expression levels were determined by real-time PCR. In vitro experiments showed that Nos significantly inhibited proliferation of SKOV3/DDP cells. DDP/Nos-combined treatment notably enhanced DDP-induced inhibition of cell proliferation and increased the pro-apoptotic effect of DDP in SKOV3/DDP cells. DDP/Nos administration increased the proportion of G2/M cells, reduced both protein and mRNA expression of anti-apoptotic factors XIAP, surviving and NF-kB, and augmented protein and mRNA levels of pro-apoptotic caspase-3. In vivo experiments revealed that Nos/DDP treatment increased the apoptotic rate of xenograft tumors in null mice. Tumor volume decreased from 1.733 ± 0.155 g in mice treated with DDP alone to 1.191 ± 0.106 g in animals treated with Nos/DDP. These observations suggest that Nos increases the anti-cancer activity of DDP against the drug-resistant ovarian cancer cell line SKOV3/DDP by modulating the cell cycle and activating apoptotic pathways. The study provides a new chemotherapy strategy for the treatment of DDP-resistant human ovarian cancer.







Similar content being viewed by others
References
Jemal, A., Siegel, R., Ward, E., Hao, Y., Xu, J., & Thun, M. J. (2009). Cancer statistics, 2009. CA: A Cancer Journal for Clinicians, 59(4), 225–249.
Perez-Tomas, R. (2006). Multidrug-resistance: Retrospect and prospects in anti-cancer drug treatment. Current Medicinal Chemistry, 13(16), 1859–1876.
Parkin, D. M., Bray, F., Ferlay, J., & Pisani, P. (2001). Estimating the world cancer burden: Globocan 2000. International Journal of Cancer, 94(2), 153–156.
McGuire, W. P., Hoskins, W. J., Brady, M. F., Kucera, P. R., Partridge, E. E., Look, K. Y., et al. (1996). Cyclophosphamide and cisplatin compared with paclitaxel and cisplatin in patients with stage III and stage IV ovarian cancer. New England Journal of Medicine, 334(1), 1–6.
Hogberg, T. (2005). A systematic overview of chemotherapy effects in ovarian cancer. Acta Oncologica, 40(2), 340–360.
Takara, K., Sakaeda, T., & Okumura, K. (2006). An update on overcoming MDR1-mediated multidrug resistance in cancer chemotherapy. Current Pharmaceutical Design, 12(3), 273–286.
Bose, R. N. (2002). Biomolecular targets for platinum antitumor drugs. Mini-Reviews in Medicinal Chemistry, 2, 103–111.
Ye, K., Ke, Y., Keshava, N., Shanks, J., Kapp, J. A., Tekmal, R. R., et al. (1998). Opium alkaloid noscapine is an antitumor agent that arrests metaphase and induces apoptosis in dividing cells. Proceedings of the National Academy of Sciences of the United States of America, 95(4), 1601–1606.
Zhou, J., Panda, D., Landen, J. W., Wilson, L., & Joshi, H. C. (2002). Minor alteration of microtubule dynamics causes loss of tension across kinetochore pairs and activates the spindle checkpoint. Journal of Biological Chemistry, 277(19), 17200–17208.
Ke, Y., Ye, K., Grossniklaus, H. E., Archer, D. R., Joshi, H. C., & Kapp, J. A. (2000). Noscapine inhibits tumor growth with little toxicity to normal tissues or inhibition of immune responses. Cancer Immunology, Immunotherapy, 49(4–5), 217–225.
Tulpule, A., Lavine, A. M., Berman, N. E., Smith, L., Auerbach, A., & Douer, D. (2005). Phase I study of noscapine for patients with non-Hodgkin’s lymphoma or chronic lymphocytic leukemia refractory to chemotherapy. Blood, 106(11), 3341.
Jackson, T., Chougule, M. B., Ichite, N., Patlolla, R. R., & Singh, M. (2008). Antitumor activity of noscapine in human non-small cell lung cancer xenograft model. Cancer Chemotherapy and Pharmacology, 63(1), 117–126.
Mahmoudian, M., & Rahimi-Moghaddam, P. (2009). The anti-cancer activity of noscapine: A review. Recent Patents on Anti-Cancer Drug Discovery, 4(1), 92–97.
Soslow, R. A., Dannenberg, A. J., Rush, D., Woerner, B. M., Khan, K. N., Masferrer, J., & Koki, A. T. (2000). Cox-2 is expressed in human pulmonary, colonic, and mammary tumors. Cancer, 89(12), 2637–2645.
Guo, Z., Ni, H., Li, B., Xing, W. G., Liu, F., Yu, H. P., et al. (2006). The effect of the MDM2-p53 loop on the sensitivity of ovarian cancer cells to cisplatin. Chinese Journal of Clinical Oncology, 3(2), 87–91.
Rauch, C. (2011). The “Multi” of drug resistance explained by oscillating drug transporters, drug–Membrane physical interactions and spatial dimensionality. Cell Biochemistry and Biophysics, 61(1), 103–113.
Harries, M., & Gore, M. (2002). Part 1: Chemotherapy for epithelial ovarian cancer-treatment at first diagnosis. Lancet Oncology, 3(9), 529–536.
Lowe, S. W., & Lin, A. W. (2000). Apoptosis in cancer. Carcinogenesis, 21(3), 485–495.
Goyal, L. (2001). Cell death inhibition: Keeping caspases in check. Cell, 104(6), 805–808.
Chougule, M. B., Patel, A. R., & Jackson, T. (2011). Antitumor activity of noscapine in combination with doxorubicin in triple negative breast cancer. PLoS One, 6(3), e17733.
Zhou, J., Gupta, K., Yao, J., Ye, K., Panda, D., Giannakakou, P., & Joshi, H. C. (2002). Paclitaxel-resistant human ovarial cancer cells undergo c-Jun NH2-terminal kinase-mediated apoptosis in response to noscapine. The Journal of Biological Chemistry, 277(42), 39777–39785.
Aneja, R., Ghaleb, A. M., Zhou, J., Yang, V. W., & Joshi, H. C. (2007). p53 and p21 determine the sensitivity of noscapine-induced apoptosis in colon cancer cells. Cancer Research, 67(8), 3862–3870.
Newcomb, E. W., Lukyanov, Y., Smirnova, I., Schnee, T., & Zagzag, D. (2008). Newcomb noscapine induces apoptosis in human glioma cells by an apoptosis-inducing factor-dependent pathway. Anti-Cancer Drugs, 19, 553–563.
Mitsios, N., Gaffney, J., Krupinski, J., Mathias, R., Wang, Q., Hayward, S., et al. (2007). Expression of signaling molecules associated with apoptosis in human ischemic stroke tissue. Cell Biochemistry and Biophysics, 47(1), 73–85.
Fulda, S., & Vucic, D. (2012). Targeting IAP proteins for therapeutic intervention in cancer. Nature Reviews Drug Discovery, 11(2), 109–124.
Kluger, H. M., McCarthy, M. M., Alvero, A. B., Sznol, M., Ariyan, S., Camp, R. L., et al. (2007). The X-linked inhibitor of apoptosis protein(XIAP)is up- regulated in metastatic melanoma, and XIAP cleavage by phenoxodiol is associated with carboplatin sensitization. Journal of Translational Medicine, 5, 6.
Schimmer, A. D., Dalili, S., Batey, R. A., & Riedl, S. J. (2006). Targeting XIAP for the treatment of malignancy. Cell Death Differentiation, 13(2), 179–188.
Kashkar, H. (2010). X-linked inhibitor of apoptosis: A chemoresistance factor or a hollow promise. Clinical Cancer Research, 16(18), 4496–4502.
Yang, L., Cao, Z., Yan, H., & Wood, W. C. (2003). Coexistence of high levels of apoptotic signaling and inhibitor of apoptosis proteins in human tumor cells: Implication for cancer specific therapy. Cancer Research, 63, 6815–6824.
Tong, Q. S., Zheng, L. D., Chen, F. M., Zeng, F. Q., Wang, L., Dong, J. H., & Lu, G. C. (2005). Selection of optimal antisense accessible sites of survivin and its application in treatment of gastric cancer. World Journal of Gastroenterology, 11(5), 634–640.
Dohi, T., Okada, K., Xia, F., Wilford, C. E., Samuel, T., Welsh, K., & Marusawa, H. (2004). An IAP–IAP complex inhibits apoptosis. Journal of Biological Chemistry, 279(33), 34087–34090.
Yang, X. H., Sladek, T. L., Liu, X., Butler, B. R., Froelich, C. J., & Thor, A. D. (2001). Reconstitution of caspase-3 sensitizes MCF-7 breast cancer cells to doxorubicin- and etoposide-induced apoptosis. Cancer Research, 61(1), 348–354.
Tsang, W. P., & Kwok, T. T. (2008). Let-7a microRNA suppresses therapeutics-induced cancer cell death by targeting caspase-3. Apoptosis, 13(10), 1215–1222.
McGonigal, K., Tanha, J., Palazov, E., Li, S. H., Gueorguieva-Owens, D., & Pandey, S. (2009). Isolation and functional characterization of single domain antibody modulators of Caspase-3 and apoptosis. Applied Biochemistry and Biotechnology, 157(2), 226–236.
Kim, Y., Kang, H., & Jang, S. W. (2011). Celastrol inhibits breast cancer cell invasion via suppression of NF-kappa B-mediated matrix metalloproteinase-9 expression. Cellular Physiology and Biochemistry, 28(2), 175–184.
Shishodia, S., Potdar, P., Gairola, C. G., & Aggarwal, C. G. (2003). Curcumin (diferuloylmethane) down-regulates cigarette smoke-induced NF-κB activation through inhibition of IκBα kinase in human lung epithelial cells: Correlation with suppression of COX-2, MMP-9 and cyclin D1. Carcinogenesis, 24(7), 1269–1279.
Chougule, M., Patel, A. R., Sachdeva, P., Jackson, T., & Singh, M. (2011). Anticancer activity of noscapine, an opioid alkaloid in combination with cisplatin in human non-small cell lung cancer. Lung Cancer, 71(3), 271–282.
Sung, B., Seok, K. S., & Aggarwal, B. (2010). Noscapine, a benzylisoquinoline alkaloid, sensitizes leukemic cells to chemotherapeutic agents and cytokines by modulating the NF-κB signaling pathway. Cancer Research, 70(8), 3259–3268.
Acknowledgments
This work was supported by the Key Project of Medical Scientific Research of Hebei Province, China (NO. 20110477).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, W., Liang, B., Yin, J. et al. Noscapine Increases the Sensitivity of Drug-Resistant Ovarian Cancer Cell Line SKOV3/DDP to Cisplatin by Regulating Cell Cycle and Activating Apoptotic Pathways. Cell Biochem Biophys 72, 203–213 (2015). https://doi.org/10.1007/s12013-014-0438-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12013-014-0438-y