Abstract
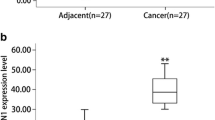
Programmed cell death 4 (PDCD4) is a tumor suppressor that can inhibit tumorigenesis by suppressing activator protein (AP)-1 activation and protein translation. Lost or decreased PDCD4 expression has been found in multiple types of human cancers, which was also associated with progression and metastasis of the tumors. However, the status and significance of PDCD4 in gastrointestinal stromal tumors have not been evaluated. In the present study, we examined the PDCD4 expression in a total of 63 gastrointestinal stromal tumor samples at both mRNA and protein levels by RT-PCR, western blot, and immunohistochemistry. We demonstrated that the expression of PDCD4 mRNA was diminished in 68% (17/25) of the tumor samples, and the level of PDCD4 protein appeared to be decreased in 66.7% (42/63) of the samples, as compared to adjacent normal gastrointestinal tissues, which expressed high levels of PDCD4 mRNA and protein. In addition, altered expression of PDCD4 was associated with clinicopathological parameters including risk group, tumor size, and mitosis. Moreover, PDCD4 expression had a negative correlation with the Ki-67 labeling index (r = −0.6059, P < 0.0001). All these results suggest that downregulation of PDCD4 expression may have an essential role in the progression and malignant proliferation of human gastrointestinal stromal tumors.






Similar content being viewed by others
References
Paral J, Slaninka I, Kalabova H, Hadzi-Nikolov D. Gastrointestinal stromal tumors: review on morphology, molecular pathology, diagnostics, prognosis and treatment options. Acta Gastroenterol Belg. 2010;73:349–59.
Bayraktar UD, Bayraktar S, Rocha-Lima CM. Molecular basis and management of gastrointestinal stromal tumors. World J Gastroenterol. 2010;16:2726–34.
Liegl-Atzwanger B, Fletcher JA, Fletcher CD. Gastrointestinal stromal tumors. Virchows Arch. 2010;456:111–27.
Lankat-Buttgereit B, Goke R. The tumour suppressor Pdcd4: recent advances in the elucidation of function and regulation. Biol Cell. 2009;101:309–17.
Cmarik JL, Min H, Hegamyer G, Zhan S, Kulesz-Martin M, Yoshinaga H, et al. Differentially expressed protein Pdcd4 inhibits tumor promoter-induced neoplastic transformation. Proc Natl Acad Sci USA. 1999;96:14037–42.
Dhar A, Young MR, Colburn NH. The role of AP-1, NF-kappaB and ROS/NOS in skin carcinogenesis: the JB6 model is predictive. Mol Cell Biochem. 2002;234–235:185–93.
Yang HS, Knies JL, Stark C, Colburn NH. Pdcd4 suppresses tumor phenotype in JB6 cells by inhibiting AP-1 transactivation. Oncogene. 2003;22:3712–20.
Hilliard A, Hilliard B, Zheng SJ, Sun H, Miwa T, Song W, et al. Translational regulation of autoimmune inflammation and lymphoma genesis by programmed cell death 4. J Immunol. 2006;177:8095–9102.
Leupold JH, Yang HS, Colburn NH, Asangani I, Post S, Allgayer H. Tumor suppressor Pdcd4 inhibits invasion/intravasation and regulates urokinase receptor (u-PAR) gene expression via Sp-transcription factors. Oncogene. 2007;26(31):4550–62.
Wang Q, Sun Z, Yang HS. Downregulation of tumor suppressor Pdcd4 promotes invasion and activates both beta-catenin/Tcf and AP-1-dependent transcription in colon carcinoma cells. Oncogene. 2008;27:1527–35.
Chen Y, Knosel T, Kristiansen G, Pietas A, Garber ME, Matsuhashi S, et al. Loss of PDCD4 expression in human lung cancer correlates with tumour progression and prognosis. J Pathol. 2003;200:640–6.
Afonja O, Juste D, Das S, Matsuhashi S, Samuels HH. Induction of PDCD4 tumor suppressor gene expression by RAR agonists, antiestrogen and HER-2/neu antagonist in breast cancer cells. Evidence for a role in apoptosis. Oncogene. 2004;23:8135–45.
Mudduluru G, Medved F, Grobholz R, Jost C, Gruber A, Leupold JH, et al. Loss of programmed cell death 4 expression marks adenoma-carcinoma transition, correlates inversely with phosphorylated protein kinase B, and is an independent prognostic factor in resected colorectal cancer. Cancer. 2007;110:1697–707.
Zhang H, Ozaki I, Mizuta T, Hamajima H, Yasutake T, Eguchi Y, et al. Involvement of programmed cell death 4 in transforming growth factor-beta1-induced apoptosis in human hepatocellular carcinoma. Oncogene. 2006;25:6101–12.
Ma G, Guo KJ, Zhang H, Ozaki I, Matsuhashi S, Zheng XY, et al. Expression of programmed cell death 4 and its clinicopathological significance in human pancreatic cancer. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2005;27:597–600.
Wei ZT, Zhang X, Wang XY, Gao F, Zhou CJ, Zhu FL, et al. PDCD4 inhibits the malignant phenotype of ovarian cancer cells. Cancer Sci. 2009;100:1408–13.
Gao F, Zhang P, Zhou C, Li J, Wang Q, Zhu F, et al. Frequent loss of PDCD4 expression in human glioma: possible role in the tumorigenesis of glioma. Oncol Rep. 2007;17:123–8.
Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Int J Surg Pathol. 2002;10:81–9.
Chadderton T, Wilson C, Bewick M, Gluck S. Evaluation of three rapid RNA extraction reagents: relevance for use in RT-PCR’s and measurement of low level gene expression in clinical samples. Cell Mol Biol (Noisy-le-grand). 1997;43:1227–34.
Culley DE, Kovacik WP Jr, Brockman FJ, Zhang W. Optimization of RNA isolation from the archaebacterium Methanosarcina barkeri and validation for oligonucleotide microarray analysis. J Microbiol Methods. 2006;67:36–43.
Scholzen T, Gerdes J. The Ki-67 protein: from the known and the unknown. J Cell Physiol. 2000;182:311–22.
Nishida T, Takahashi T, Miyazaki Y. Gastrointestinal stromal tumor: a bridge between bench and bedside. Gastric Cancer. 2009;12:175–88.
Belev B, Vrbanec D, Kralik M, Plestina S, Petricević B, Sirotković-Skerlev M. Gastrointestinal stromal tumors (GIST)—a paradigm of successful targeted therapy of solid tumors. Acta Med Croatica. 2006;60:471–5.
Lankat-Buttgereit B, Gregel C, Knolle A, Hasilik A, Arnold R, Goke R. PDCD4 inhibits growth of tumor cells by suppression of carbonic anhydrase type II. Mol Cell Endocrinol. 2004;214:149–53.
Stalberg P, Lopez-Egido JR, Wang S, Gobl A, Oberg K, Skogseid B. Differentially expressed cDNAs in PLCbeta3-induced tumor suppression in a human endocrine pancreatic tumor cell line: activation of the human mismatch repair protein 3 gene. Biochem Biophys Res Commun. 2001;281:227–31.
Yoshinaga H, Matsuhashi S, Fujiyama C, Masaki Z. Novel human PDCD4 (H731) gene expression in proliferative cells is expressed in the small duct epithelial cells of the breast as revealed by an anti-H731 antibody. Int Pathol. 1997;49:1067–77.
Goke A, Goke R, Knolle A, Trusheim H, Schmidt H, Wilmen A, et al. DUG is a novel homologue of translation initiation factor 4G that binds eIF4A. Biochem Biophys Res Commun. 2002;297:78–82.
Goke R, Gregel C, Goke A, Arnold R, Schmidt H, Lankat-Buttgereit B. Programmed cell death protein 4 (PDCD4) acts as a tumor suppressor in neuroendocrine tumor cells. Ann NY Acad Sci. 2004;1014:220–1.
Jansen AP, Camalier CE, Colburn NH. Epidermal expression of the translation inhibitor programmed cell death 4 suppresses tumorigenesis. Cancer Res. 2005;65:6034–41.
Bitomsky N, Bohm M, Klempnauer KH. Transformation suppressor protein PDCD4 interferes with JNK-mediated phosphorylation of c-Jun and recruitment of the coactivator p300 by c-Jun. Oncogene. 2004;23:7484–93.
Dorrello NV, Peschiaroli A, Guardavaccaro D, Colburn NH, Sherman NE, Pagano M. S6K1- and betaTRCP-mediated degradation of PDCD4 promotes protein translation and cell growth. Science. 2006;314:467–71.
Gao F, Wang X, Zhu F, Wang Q, Zhang X, Guo C, et al. PDCD4 gene silencing in gliomas is associated with 5′CpG island methylation and unfavorable prognosis. J Cell Mol Med. 2009;13:4257–67.
Young MR, Santhanam AN, Yoshikawa N, Colburn NH. Have tumor suppressor PDCD4 and its counteragent oncogenic miR-21 gone rogue? Mol Interv. 2010;10:76–9.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No. 81000126), grants from the Specialized Research Fund for the Doctoral Program of Higher Education of China (No. 20090131120066), and grants from the Natural Science Foundation of Shandong Province, China (No. ZR2009CM021-2009ZRB01192).
Conflict of interest
The authors have declared that no conflict of interest exists.
Author information
Authors and Affiliations
Corresponding author
Additional information
Lingling Ding, Xia Zhang, and Miaoqing Zhao contributed equally to this article.
Rights and permissions
About this article
Cite this article
Ding, L., Zhang, X., Zhao, M. et al. An essential role of PDCD4 in progression and malignant proliferation of gastrointestinal stromal tumors. Med Oncol 29, 1758–1764 (2012). https://doi.org/10.1007/s12032-011-0042-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-011-0042-6