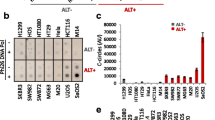
Abstract
Despite a standard of care combining surgery, radiotherapy (RT), and temozolomide chemotherapy, the average overall survival (OS) of glioblastoma patients is only 15 months, and even far lower when the patient cannot benefit from this combination. Therefore, there is a strong need for new treatments, such as new irradiation techniques. Against this background, carbon ion hadrontherapy, a new kind of irradiation, leads to a greater biological response of the tumor, while minimizing adverse effects on healthy tissues in comparison with RT. As carbon ion hadrontherapy is restricted to RT-resistant patients, photon irradiation resistance biomarkers are needed. Long telomeres and high telomerase activity have been widely associated with photon radioresistance in other cancers. Moreover, telomere protection, telomere function, and telomere length (TL) also depend on the shelterin protein complex (TRF1, TRF2, TPP1, POT1, TIN2, and hRAP1). We thus decided to evaluate an enlarged telomeric status (TL, telomerase catalytic subunit, and the shelterin component expression level) as a potential radioresistance biomarker in vitro using cellular models and ex vivo using patient tumor biopsies. In addition, nothing was known about the role of telomeres in carbon ion response. We thus evaluated telomeric status after both types of irradiation. We report here a significant correlation between TL and the basal POT1 expression level and photon radioresistance, in vitro, and a significant increase in the OS of patients with long telomeres or a high POT1 level, in vivo. POT1 expression was predictive of patient response irrespective of the TL. Strikingly, these correlations were lost, in vitro, when considering carbon irradiation. We thus propose (1) a model of the implications of telomeric damage in the cell response to both types of irradiation and (2) assessment of the POT1 expression level and TL using patient tumor biopsies to identify radioresistant patients who could benefit from carbon hadrontherapy.








Similar content being viewed by others
References
Preusser M, de Ribaupierre S, Wohrer A, Erridge SC, Hegi M, Weller M, Stupp R (2011) Current concepts and management of glioblastoma. Ann Neurol 70(1):9–21. doi:10.1002/ana.22425
Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y, Wilkerson MD, Miller CR, Ding L, Golub T, Mesirov JP, Alexe G, Lawrence M, O’Kelly M, Tamayo P, Weir BA, Gabriel S, Winckler W, Gupta S, Jakkula L, Feiler HS, Hodgson JG, James CD, Sarkaria JN, Brennan C, Kahn A, Spellman PT, Wilson RK, Speed TP, Gray JW, Meyerson M, Getz G, Perou CM, Hayes DN (2010) Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 17(1):98–110. doi:10.1016/j.ccr.2009.12.020
Hegi ME, Diserens AC, Gorlia T, Hamou MF, de Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, Bromberg JE, Hau P, Mirimanoff RO, Cairncross JG, Janzer RC, Stupp R (2005) MGMT gene silencing and benefit from temozolomide in glioblastoma. N Engl J Med 352(10):997–1003. doi:10.1056/NEJMoa043331
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352(10):987–996. doi:10.1056/NEJMoa043330
Amelio D, Lorentini S, Schwarz M, Amichetti M (2010) Intensity-modulated radiation therapy in newly diagnosed glioblastoma: a systematic review on clinical and technical issues. Radiother Oncol 97(3):361–369. doi:10.1016/j.radonc.2010.08.018
Li L, Quang TS, Gracely EJ, Kim JH, Emrich JG, Yaeger TE, Jenrette JM, Cohen SC, Black P, Brady LW (2010) A phase II study of anti-epidermal growth factor receptor radioimmunotherapy in the treatment of glioblastoma multiforme. J Neurosurg 113(2):192–198. doi:10.3171/2010.2.JNS091211
Mizoe JE, Tsujii H, Hasegawa A, Yanagi T, Takagi R, Kamada T, Tsuji H, Takakura K (2007) Phase I/II clinical trial of carbon ion radiotherapy for malignant gliomas: combined X-ray radiotherapy, chemotherapy, and carbon ion radiotherapy. Int J Radiat Oncol Biol Phys 69(2):390–396
Allen C, Borak TB, Tsujii H, Nickoloff JA (2011) Heavy charged particle radiobiology: using enhanced biological effectiveness and improved beam focusing to advance cancer therapy. Mutat Res 711(1–2):150–157. doi:10.1016/j.mrfmmm.2011.02.012
Maucort-Boulch D, Baron MH, Pommier P, Weber DC, Mizoe JE, Rochat J, Boissel JP, Balosso J, Tsujii H, Amsallem E (2010) Rationale for carbon ion therapy in high-grade glioma based on a review and a meta-analysis of neutron beam trials. Cancer Radiother 14(1):34–41. doi:10.1016/j.canrad.2009.08.141
Boldrini L, Pistolesi S, Gisfredi S, Ursino S, Ali G, Pieracci N, Basolo F, Parenti G, Fontanini G (2006) Telomerase activity and hTERT mRNA expression in glial tumors. Int J Oncol 28(6):1555–1560
Akiyama M, Yamada O, Kanda N, Akita S, Kawano T, Ohno T, Mizoguchi H, Eto Y, Anderson KC, Yamada H (2002) Telomerase overexpression in K562 leukemia cells protects against apoptosis by serum deprivation and double-stranded DNA break inducing agents, but not against DNA synthesis inhibitors. Cancer Lett 178(2):187–197
Serakinci N, Christensen R, Graakjaer J, Cairney CJ, Keith WN, Alsner J, Saretzki G, Kolvraa S (2007) Ectopically hTERT expressing adult human mesenchymal stem cells are less radiosensitive than their telomerase negative counterpart. Exp Cell Res 313(5):1056–1067
Gomez-Millan J, Goldblatt EM, Gryaznov SM, Mendonca MS, Herbert BS (2007) Specific telomere dysfunction induced by GRN163L increases radiation sensitivity in breast cancer cells. Int J Radiat Oncol Biol Phys 67(3):897–905
Wesbuer S, Lanvers-Kaminsky C, Duran-Seuberth I, Bolling T, Schafer KL, Braun Y, Willich N, Greve B (2010) Association of telomerase activity with radio- and chemosensitivity of neuroblastomas. Radiat Oncol 5:66. doi:10.1186/1748-717X-5-66
Papanikolaou V, Athanassiou E, Dubos S, Dimou I, Papathanasiou I, Kitsiou-Tzeli S, Kappas C, Tsezou A (2011) hTERT regulation by NF-kappaB and c-myc in irradiated HER2-positive breast cancer cells. Int J Radiat Biol 87(6):609–621. doi:10.3109/09553002.2011.572112
Ram R, Uziel O, Eldan O, Fenig E, Beery E, Lichtenberg S, Nordenberg Y, Lahav M (2009) Ionizing radiation up-regulates telomerase activity in cancer cell lines by post-translational mechanism via ras/phosphatidylinositol 3-kinase/Akt pathway. Clin Cancer Res 15(3):914–923. doi:10.1158/1078-0432.CCR-08-0792
Kurvinen K, Rantanen V, Syrjanen S, Johansson B (2006) Radiation-induced effects on telomerase in gynecological cancer cell lines with different radiosensitivity and repair capacity. Int J Radiat Biol 82(12):859–867
Zhong YH, Liao ZK, Zhou FX, Xie CH, Xiao CY, Pan DF, Luo ZG, Liu SQ, Zhou YF (2008) Telomere length inversely correlates with radiosensitivity in human carcinoma cells with the same tissue background. Biochem Biophys Res Commun 367(1):84–89. doi:10.1016/j.bbrc.2007.12.078
Komuro Y, Watanabe T, Tsurita G, Muto T, Nagawa H (2005) Expression pattern of telomerase reverse transcriptase in rectal carcinoma predicts tumor radiosensitivity, local recurrence and disease-free survival. Hepato-Gastroenterology 52(64):985–989
Ogawa Y, Nishioka A, Hamada N, Terashima M, Inomata T, Yoshida S, Seguchi H, Kishimoto S (1998) Changes in telomerase activity of advanced cancers of oral cavity and oropharynx during radiation therapy: correlation with clinical outcome. Int J Mol Med 2(3):301–307
Tchirkov A, Rolhion C, Kemeny JL, Irthum B, Puget S, Khalil T, Chinot O, Kwiatkowski F, Perissel B, Vago P, Verrelle P (2003) Clinical implications of quantitative real-time RT-PCR analysis of hTERT gene expression in human gliomas. Br J Cancer 88(4):516–520
Hakin-Smith V, Jellinek DA, Levy D, Carroll T, Teo M, Timperley WR, McKay MJ, Reddel RR, Royds JA (2003) Alternative lengthening of telomeres and survival in patients with glioblastoma multiforme. Lancet 361(9360):836–838
Martinez P, Blasco MA (2011) Telomeric and extra-telomeric roles for telomerase and the telomere-binding proteins. Nat Rev Cancer 11(3):161–176. doi:10.1038/nrc3025
Du G, Drexler GA, Friedland W, Greubel C, Hable V, Krucken R, Kugler A, Tonelli L, Friedl AA, Dollinger G (2011) Spatial dynamics of DNA damage response protein foci along the ion trajectory of high-LET particles. Radiat Res 176(6):706–715. doi:10.1667/RR2592.1
Iwadate Y, Mizoe J, Osaka Y, Yamaura A, Tsujii H (2001) High linear energy transfer carbon radiation effectively kills cultured glioma cells with either mutant or wild-type p53. Int J Radiat Oncol Biol Phys 50(3):803–808
Kamlah F, Hanze J, Arenz A, Seay U, Hasan D, Juricko J, Bischoff B, Gottschald OR, Fournier C, Taucher-Scholz G, Scholz M, Seeger W, Engenhart-Cabillic R, Rose F (2011) Comparison of the effects of carbon ion and photon irradiation on the angiogenic response in human lung adenocarcinoma cells. Int J Radiat Oncol Biol Phys 80(5):1541–1549. doi:10.1016/j.ijrobp.2011.03.033
Cui X, Oonishi K, Tsujii H, Yasuda T, Matsumoto Y, Furusawa Y, Akashi M, Kamada T, Okayasu R (2011) Effects of carbon ion beam on putative colon cancer stem cells and its comparison with X-rays. Cancer Res 71(10):3676–3687. doi:10.1158/0008-5472.CAN-10-2926
Rieken S, Habermehl D, Wuerth L, Brons S, Mohr A, Lindel K, Weber K, Haberer T, Debus J, Combs SE (2011) Carbon ion irradiation inhibits glioma cell migration through downregulation of integrin expression. Int J Radiat Oncol Biol Phys. doi:10.1016/j.ijrobp.2011.06.2004
Alphonse G, Aloy MT, Broquet P, Gerard JP, Louisot P, Rousson R, Rodriguez-Lafrasse C (2002) Ceramide induces activation of the mitochondrial/caspases pathway in Jurkat and SCC61 cells sensitive to gamma-radiation but activation of this sequence is defective in radioresistant SQ20B cells. Int J Radiat Biol 78(9):821–835
Beuve M, Alphonse G, Maalouf M, Colliaux A, Battiston-Montagne P, Jalade P, Balanzat E, Demeyer A, Bajard M, Rodriguez-Lafrasse C (2008) Radiobiologic parameters and local effect model predictions for head-and-neck squamous cell carcinomas exposed to high linear energy transfer ions. Int J Radiat Oncol Biol Phys 71(2):635–642
Poncet D, Belleville A, t’kint de Roodenbeke C, Roborel de Climens A, Ben Simon E, Merle-Beral H, Callet-Bauchu E, Salles G, Sabatier L, Delic J, Gilson E (2008) Changes in the expression of telomere maintenance genes suggest global telomere dysfunction in B-chronic lymphocytic leukemia. Blood 111(4):2388–2391
Leteurtre F, Li X, Gluckman E, Carosella ED (1997) Telomerase activity during the cell cycle and in gamma-irradiated hematopoietic cells. Leukemia 11(10):1681–1689
Neuhof D, Ruess A, Wenz F, Weber KJ (2001) Induction of telomerase activity by irradiation in human lymphoblasts. Radiat Res 155(5):693–697
Satra M, Tsougos I, Papanikolaou V, Theodorou K, Kappas C, Tsezou A (2006) Correlation between radiation-induced telomerase activity and human telomerase reverse transcriptase mRNA expression in HeLa cells. Int J Radiat Biol 82(6):401–409
Hyeon Joo O, Hande MP, Lansdorp PM, Natarajan AT (1998) Induction of telomerase activity and chromosome aberrations in human tumour cell lines following X-irradiation. Mutat Res 401(1–2):121–131
He X, Qiao Q, Ge N, Nan J, Shen S, Wang Z, Yang Y, Bao G (2010) Irradiation-induced telomerase activity and gastric cancer risk: a case–control analysis in a Chinese Han population. BMC Cancer 10:312. doi:10.1186/1471-2407-10-312
Neuhof D, Bischof M, Debus J, Weber KJ (2007) Telomerase activity in peripheral blood mononuclear cells after whole body irradiation. Oncol Rep 17(4):865–869
Yajima T, Yagihashi A, Kameshima H, Kobayashi D, Hirata K, Watanabe N (2001) Telomerase reverse transcriptase and telomeric-repeat binding factor protein 1 as regulators of telomerase activity in pancreatic cancer cells. Br J Cancer 85(5):752–757
Loayza D, De Lange T (2003) POT1 as a terminal transducer of TRF1 telomere length control. Nature 423(6943):1013–1018
Chen YJ, Hakin-Smith V, Teo M, Xinarianos GE, Jellinek DA, Carroll T, McDowell D, MacFarlane MR, Boet R, Baguley BC, Braithwaite AW, Reddel RR, Royds JA (2006) Association of mutant TP53 with alternative lengthening of telomeres and favorable prognosis in glioma. Cancer Res 66(13):6473–6476
McDonald KL, McDonnell J, Muntoni A, Henson JD, Hegi ME, von Deimling A, Wheeler HR, Cook RJ, Biggs MT, Little NS, Robinson BG, Reddel RR, Royds JA (2010) Presence of alternative lengthening of telomeres mechanism in patients with glioblastoma identifies a less aggressive tumor type with longer survival. J Neuropathol Exp Neurol 69(7):729–736. doi:10.1097/NEN.0b013e3181e576cf
Sampl S, Pramhas S, Stern C, Preusser M, Marosi C, Holzmann K (2012) Expression of telomeres in astrocytoma WHO grade 2 to 4: TERRA level correlates with telomere length, telomerase activity, and advanced clinical grade. Transl Oncol 5(1):56–65
Nakagami Y, Ito M, Hara T, Inoue T (2002) Loss of TRF2 by radiation-induced apoptosis in HL60 cells. Radiat Med 20(3):121–129
Ning H, Li T, Zhao L, Li T, Li J, Liu J, Liu Z, Fan D (2006) TRF2 promotes multidrug resistance in gastric cancer cells. Cancer Biol Ther 5(8):950–956
Wu L, Multani AS, He H, Cosme-Blanco W, Deng Y, Deng JM, Bachilo O, Pathak S, Tahara H, Bailey SM, Deng Y, Behringer RR, Chang S (2006) Pot1 deficiency initiates DNA damage checkpoint activation and aberrant homologous recombination at telomeres. Cell 126(1):49–62
Perera SA, Maser RS, Xia H, McNamara K, Protopopov A, Chen L, Hezel AF, Kim CF, Bronson RT, Castrillon DH, Chin L, Bardeesy N, Depinho RA, Wong KK (2008) Telomere dysfunction promotes genome instability and metastatic potential in a K-ras p53 mouse model of lung cancer. Carcinogenesis 29(4):747–753. doi:10.1093/carcin/bgn050
Artandi SE, Chang S, Lee SL, Alson S, Gottlieb GJ, Chin L, DePinho RA (2000) Telomere dysfunction promotes non-reciprocal translocations and epithelial cancers in mice. Nature 406(6796):641–645
Begus-Nahrmann Y, Hartmann D, Kraus J, Eshraghi P, Scheffold A, Grieb M, Rasche V, Schirmacher P, Lee HW, Kestler HA, Lechel A, Rudolph KL (2012) Transient telomere dysfunction induces chromosomal instability and promotes carcinogenesis. J Clin Invest 122(6):2283–2288. doi:10.1172/JCI61745
Salhab M, Jiang WG, Newbold RF, Mokbel K (2007) The expression of gene transcripts of telomere-associated genes in human breast cancer: correlation with clinico-pathological parameters and clinical outcome. Breast Cancer Res Treat 109:35–46
Fujii K, Sasahira T, Moriwaka Y, Oue N, Yasui W, Kuniyasu H (2008) Protection of telomeres 1 protein levels are associated with telomere length in gastric cancer. Int J Mol Med 21(5):599–604
Tang T, Zhou FX, Lei H, Yu HJ, Xie CH, Zhou YF, Liu SQ (2009) Increased expression of telomere-related proteins correlates with resistance to radiation in human laryngeal cancer cell lines. Oncol Rep 21(6):1505–1509
Drissi R, Wu J, Hu Y, Bockhold C, Dome JS (2011) Telomere shortening alters the kinetics of the DNA damage response after ionizing radiation in human cells. Cancer Prev Res (Phila) 4(12):1973–1981. doi:10.1158/1940-6207.CAPR-11-0069
Soler D, Pampalona J, Tusell L, Genesca A (2009) Radiation sensitivity increases with proliferation-associated telomere dysfunction in nontransformed human epithelial cells. Aging Cell 8(4):414–425. doi:10.1111/j.1474-9726.2009.00488.x
Goytisolo FA, Samper E, Martin-Caballero J, Finnon P, Herrera E, Flores JM, Bouffler SD, Blasco MA (2000) Short telomeres result in organismal hypersensitivity to ionizing radiation in mammals. J Exp Med 192(11):1625–1636
Latre L, Tusell L, Martin M, Miro R, Egozcue J, Blasco MA, Genesca A (2003) Shortened telomeres join to DNA breaks interfering with their correct repair. Exp Cell Res 287(2):282–288
Wong KK, Chang S, Weiler SR, Ganesan S, Chaudhuri J, Zhu C, Artandi SE, Rudolph KL, Gottlieb GJ, Chin L, Alt FW, DePinho RA (2000) Telomere dysfunction impairs DNA repair and enhances sensitivity to ionizing radiation. Nat Genet 26(1):85–88
Bhattacharyya S, Sandy A, Groden J (2010) Unwinding protein complexes in ALTernative telomere maintenance. J Cell Biochem 109(1):7–15. doi:10.1002/jcb.22388
Opresko PL, Mason PA, Podell ER, Lei M, Hickson ID, Cech TR, Bohr VA (2005) POT1 stimulates RecQ helicases WRN and BLM to unwind telomeric DNA substrates. J Biol Chem 280(37):32069–32080. doi:10.1074/jbc.M505211200
Flynn RL, Centore RC, O’Sullivan RJ, Rai R, Tse A, Songyang Z, Chang S, Karlseder J, Zou L (2011) TERRA and hnRNPA1 orchestrate an RPA-to-POT1 switch on telomeric single-stranded DNA. Nature 471(7339):532–536. doi:10.1038/nature09772
Nakano T, Suzuki Y, Ohno T, Kato S, Suzuki M, Morita S, Sato S, Oka K, Tsujii H (2006) Carbon beam therapy overcomes the radiation resistance of uterine cervical cancer originating from hypoxia. Clin Cancer Res 12(7 Pt 1):2185–2190. doi:10.1158/1078-0432.CCR-05-1907
Oikawa S, Kawanishi S (1999) Site-specific DNA damage at GGG sequence by oxidative stress may accelerate telomere shortening. FEBS Lett 453(3):365–368
Chuang TC, Moshir S, Garini Y, Chuang AY, Young IT, Vermolen B, van den Doel R, Mougey V, Perrin M, Braun M, Kerr PD, Fest T, Boukamp P, Mai S (2004) The three-dimensional organization of telomeres in the nucleus of mammalian cells. BMC Biol 2:12. doi:10.1186/1741-7007-2-121741-7007-2-12
R Development Core Team (2005) R: A language and environment for statistical computing, reference index version 2.2.1. R Foundation for Statistical Computing, Vienna, Austria. ISBN: 3-900051-07-0. Available at http://www.R-project.org
Acknowledgments
This work was supported by “La Ligue Nationale contre le Cancer” (comité de la Saône), the “Association pour la Recherche contre le Cancer” (ARC), the “Association des Neuro-Oncologues d’Expression Française” (ANOCEF), and Geron Corp. We thank Mrs. Helen Morrin and Mr. Martin McFarlane of The Cancer Society Tissue Bank, The University of Otago Christchurch (New Zealand) for the glioma tissue samples and related information. We thank the GRRAAL (Gliomas Radiobiology Rhône-Alpes Auvergne Labs) project for sharing the GBM-derived cell lines, Guillaume Belz for his valuable help in “R” manipulation, and Sergei Gryaznov for his accurate corrections. Pr. Rodriguez-Lafrasse Laboratory is part of the Labex PRIMES (“Physique Radiobiologie Imagerie Médicale et Simulation”) and is supported by the PRRH (Programme Régional de Recherches en Hadronthérapie).
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 177 kb)
Rights and permissions
About this article
Cite this article
Ferrandon, S., Saultier, P., Carras, J. et al. Telomere Profiling: Toward Glioblastoma Personalized Medicine. Mol Neurobiol 47, 64–76 (2013). https://doi.org/10.1007/s12035-012-8363-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-012-8363-9